Characidium tatama, Agudelo‐Zamora & Tavera & Murillo & Ortega‐Lara, 2020
|
publication ID |
https://doi.org/10.1111/jfb.14527 |
|
DOI |
https://doi.org/10.5281/zenodo.4324229 |
|
persistent identifier |
https://treatment.plazi.org/id/03F287B4-FF93-FFDD-D639-FD2A7CD8FBEA |
|
treatment provided by |
Plazi |
|
scientific name |
Characidium tatama |
| status |
sp. nov. |
3.2 | Characidium tatama new species
Figures 3 View FIGURE 3 , 4 View FIGURE 4 and 5 View FIGURE 5 , and Table 1 View TABLE 1 .
urn:lsid:zoobank.org:act:4170703E-80D7-4170-9212-10B5918D3E79.
Characidium sp. 2 “ Pacifico ” – Agudelo-Zamora et al., 2020, p. 14 (material examined).
3.3 | Holotype
IMCN 8925 , 38.1 mm L S, Colombia, Chocó , San José Del Palmar, río Ingará by the bridge, 04 55 ' 13.39 '' N, 76 12 ' 21.48 '' W, A. Ortega-Lara.
09 September 2018.
GoogleMaps3.4 | Paratypes
All from Colombia. IMCN 8924 , 1 C&S female, 36.0 mm L S; CHIFF 0011 , 2, 27.9 – 36.3 mm L S, all collected with the holotype . IMCN 8926 , 1 ( AOL 138 tissue), 36 mm L S, Colombia, Chocó, San José Del Palmar, río Cruces tributary of río Ingará , A. Ortega-Lara. 09 September 2018 ; IMCN 8927 , 1 ( AOL 139 tissue), 32.5 mm L S, Colombia, Chocó, San José Del Palmar, quebrada Corcobado , 4 54 ' 54.9 '' N, 76 13 ' 3.6 '' W, A. Ortega-Lara, 09 September 2018 GoogleMaps ; CIRUV 010-19 View Materials , 1, 30.5 mm L S, Colombia, Valle del Cauca, Buenaventura, quebrada La Brea, tributary of río Calima , 3 59 ' 25.7 '' N, 76 58 ' 23.3 '' W. Díaz & Serna. 16 May 2010 GoogleMaps .
3.5 | Diagnosis
Characidium tatama is distinguished from all Characidium , except C. alipioi Travassos 1955 , C. amaila Lujan et al., 2013 , C. boavistae Steindachner 1915 , C. bolivianum Pearson 1924 , C. crandellii Steindachner 1915 , C. declivirostre Steindachner 1915 , C. gomesi Travassos 1956 , C. grajahuense Travassos 1944 , C. japuhybense Travassos 1949 , C. kamakan Zanata & Camelier, 2015 , C. lauroi Travassos 1949 , C. pterostictum Gomes 1947 , C. purpuratum Steindachner 1882 , C. timbuiense Travassos, 1946 , C. travassosi Melo et al., 2016 , C. vidali Travassos 1967 and the clade members of C. fasciatum or clade C1 ( sensu Buckup, 1992) for having the isthmus without scales (Figure 3). Also, C. tatama is distinguished from C. helmeri Zanata , Sarmento- Soares & Martins-Pinheiro 2015, C. oiticicai Travassos 1967 and C. travassossi Melo et al., 2016 by the presence of an adipose fin ( vs. adipose fin absence). Additionally, C. tatama n. sp. is distinguished among clade C1 congeners by having a rounded point in the midline of the upper scales of the lateral line ( Figure 4 View FIGURE 4 ); it is distinguished from C. amaila by having a lower number of scales on the lateral line (32 – 33 vs. 34 – 36) and by the shape of its swim bladder (wide bilobed anterior chamber and small posterior thin chamber vs. anterior chamber as wide as the posterior, and the posterior chamber long and thin); from C. crandellii and C. declivirostre by having free branchiostegal rays ( vs. joined branchiostegal rays [see Lujan et al., 2013, p. 455); from C. boavistae , C. bolivianum , C. gomesi , C. grajahuensis , C. japuhybense , C. kamakan , C. lauroi , C. purpuratum , C. pterostictum , C. schubarti and C. timbuiense for having the caudal and anal fins without bands on the rays ( vs. caudal and anal fins with dark pigment or with some type of band) (see Lujan et al., 2013, p. 458; Zanata & Camelier, 2015, p. 488).
3.6 | Description
Morphometric data can be seen in Table 1 View TABLE 1 , the largest known specimen not exceeding 45 mm L S Body slightly compressed laterally, strongly convex dorsal profile from snout to supraoccipital fontanelle, ascending slightly convex arc to dorsal-fin origin; descending through base of dorsal fin, and to first caudal-fin procurrent ray. Ventral profile concave from dentary to pelvic fin, vertex located from this point to caudal peduncle straight. Greatest body depth at, or just above, dorsal-fin origin. Snout strongly rounded tip, at level of ventral margin of orbit. Maxillary long and extending beyond anterior margin of orbit. Circular orbit larger than snout length, smaller than maxilla. Iris round.
Dorsal-fin rays ii8(1), 9(5), 10*(1); anal-fin rays ii6*(7); pectoral-fin rays ii6(1), 7*(2) ii8(4); pelvic-fin rays i7(6), 8*(1); principal caudal-fin rays in dorsal lobe 8(1), 9*(5), in ventral lobe 8*(7).
Scales cycloid. Lateral line complete; pored lateral-line scales 32(2), 33*(5). Scales above lateral line 3(1), 4*(6), below 2*(7). Predorsal scales 11*(7). Circumpeduncular scales 10(1), 11*(4). Isthmus and area between pectoral fins without scales.
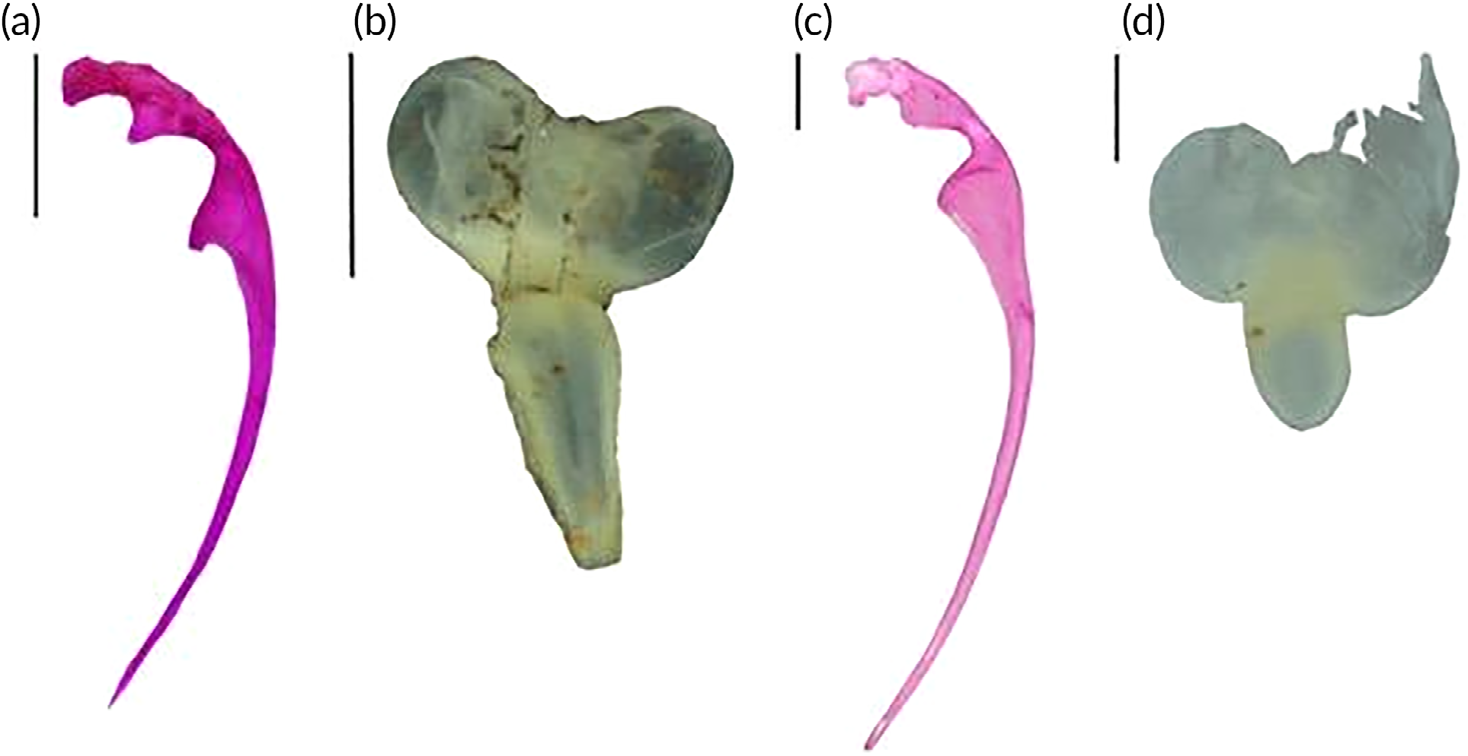
Mesethmoid bone separated from frontal, parietal fontanel absent, postcleitrum (PC) I absent, PC II and III present. Medial process of first rib with ventral concave curvature in hook shape decreasing in width to the tip of belly, dorsal upper process triangular with concave space between process and base to vertebral joint ( Figure 8a View FIGURE 8 ). Swimbladder with two chambers, anterior chamber bilobate and short, posterior chamber slim, its base wider than the tip ( Figure 8b View FIGURE 8 ).
3.7 | Colour in alcohol
Dark colouration on head due to the high concentration of chromatophores divided longitudinally by a black lateral band that crosses the eye. Ventral region silver and divided by the lateral line. Body crossed by longitudinal band that ends in the caudal region with a dark bar on the base of the caudal fin, basicaudal spot in medial rays of the caudal fin. Dorsal region brown with the edges of the scales black, forming a pattern of chainrings, which extend beyond the longitudinal band. Pectoral, pelvic and anal fins hyaline; caudal fin in smaller specimens (< 28 mm L S,) also hyaline, but larger specimens with scattered chromatophores not clustered or forming bands. Dorsal-fin base with dark black stripe. Chromatophores forming a band at the base of the dorsal fin. Unpigmented belly, except on caudal peduncle, which has a last dark transversal body band.
3.8 | Ecology
The Ingará River ( type locality) has U-shaped banks ( sensu Parsons et al., 2002) with abundant riparian coverage on both sides, and a predominance of a rocky substrate and some large ridges, which makes it a torrential river with few pools or backwaters ( Figure 5 View FIGURE 5 ).
3.9 | Distribution
Characidium tatama is endemic to the Chocó biogeographic region and inhabits rivers tributaries of the upper San Juan and Calima river (Figure 1).
3.10 | Etymology
A noun in apposition to the Tatamá National Natural Park created in 1987 and the place where most of the type specimens were collected. The word Tatamá in Embera-Chamí language means the grandfather of the rivers.
3.11 | Conservation assessment
Characidium tatama is so far known from two localities in tributaries of the San Juan and Calima Rivers. The EOO estimated for the species is 5.419. 3 km 2, and the AOO estimated is 74 km 2 ( sensu Bachman et al., 2011). Additional collecting efforts should be conducted in the region to better understand the geographic distribution of the species and the existent threats. From the points assessed here for the distribution of C. tatama , the species is tentatively categorized as Vulnerable (VU) D2 under the geospatial aspect because the population is restricted to less than 20 km 2 and the number of localities is less than five (IUCN Standards and Petitions Subcommittee, 2019), with accelerated deforestation inside Colombian national parks ( Clerici et al., 2020).
TABLE 1 Morphometric data from C. tatama n. sp. (n = 7)
| Holotype | Range | Mean | S.D. | |
|---|---|---|---|---|
| TL | 44.2 | 33.6 – 44.2 | 39.5 | |
| SL | 38.1 | 27.9 – 38.1 | 33.2 | |
| % SL | ||||
| Head length | 21.5 | 22.5 – 25 | 23.8 | 0.9 |
| Pectoral distance | 20.3 | 20.8 – 26.1 | 22.8 | 2.0 |
| Predorsal distance | 45.5 | 44.2 – 50.6 | 47.2 | 2.3 |
| Prepelvic distance | 51.2 | 48.3 – 59.1 | 53.4 | 4.0 |
| Preanal distance | 76.2 | 71.1 – 83.1 | 76.6 | 3.9 |
| Anal-apex distance | 84.0 | 82.1 – 90.2 | 85.8 | 3.4 |
| Body width | 10.5 | 9.4 – 11.3 | 10.2 | 0.7 |
| Body depth at DO | 17.4 | 15.2 – 19.3 | 18.0 | 1.6 |
| Body depth at AO | 12.9 | 12.7 – 16.6 | 14.4 | 1.3 |
| Caudal peduncle depth | 11.0 | 9.4 – 11.5 | 10.5 | 0.7 |
| % HL | ||||
| Snout length | 23.0 | 17.7 – 22.8 | 19.8 | 2.1 |
| Snout-maxillary tip | 27.5 | 21.8 – 28 | 25.7 | 2.3 |
| Anterior naris-eye | 8.2 | 3.7 – 10.2 | 6.0 | 2.3 |
| Posterior naris - eye | 1,7 | 1.3 – 3.8 | 2.6 | 1.0 |
| Cheek | 23.4 | 18.5 – 25.8 | 21.3 | 2.8 |
| Orbital diameter | 26.6 | 24.7 – 31.4 | 28.0 | 2.3 |
| Interorbital distance | 10.8 | 8.7 – 17.7 | 13.1 | 3.3 |
Abbreviations: DO, dorsal origin; AO, anal origin; HL, head length; S.D; standard deviation; LS, standard length; TL, total length.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |