Huffmanela longa, Justine, Jean-Lou, 2007
|
publication ID |
https://doi.org/10.5281/zenodo.179332 |
|
DOI |
https://doi.org/10.5281/zenodo.6247500 |
|
persistent identifier |
https://treatment.plazi.org/id/0E737405-5155-FFB6-D5FD-FB3CFAA814C2 |
|
treatment provided by |
Plazi |
|
scientific name |
Huffmanela longa |
| status |
sp. nov. |
Huffmanela longa n. sp.
( Figs 4–5)
Type-host: Gymnocranius grandoculis (Valenciennes) (Lethrinidae)
Hosts: Three infected hosts were found: JNC1726, 24.i.2006, FL 570, W 3500; JNC2166, 15.v.2007, FL 595, W 3800; JNC2174, 5.vi.2007, FL 365, W 1050. All from near Récif Toombo, off Nouméa, New Caledonia, 22°32’S, 166°28’E.
Type-locality: Lagoon off Nouméa, New Caledonia.
Material examined: a single incomplete female ( holotype); laid eggs.
Deposition of material: Holotype, MNHN, JNC 2174; vouchers of eggs, MNHN, JNC 1726, JNC 2166, JNC 2174; vouchers of eggs from fish JNC 2166, HCIP, N-881; BMNH, 2007.7.12.2; USNPC, 99978; SAMA, AHC 34776.
Site of infection: Eggs laid within mesentery, external mucosa of swimbladder, and mucosa of dorsal wall of abdominal body cavity.
Remarks on infection and prevalence: In fish JNC1726, the eggs formed a dark spot, about 1 cm in diameter, on the mesentery attached to the gonad; no other organ was infected. In fish JNC2166 and JNC2174, the eggs were found on the surface of two organs: the outer mucosa of the swimbladder and the mucosa of the dorsal wall of the abdominal cavity; no other organ was infected. Prevalence was 3/12 (25%).
Etymology: longa , long in Latin, for the length of body.
Comparative material examined: syntypes of Huffmanela filamentosa Justine, 2004 , MNHN JNC 892A. A slide with eggs kept in lactophenol for more than 3 years was examined using the same methods and microscope as the new species, and measurements were consistent with those of the original publication ( Table 4 View TABLE 4 ).
Female [from holotype, immature]. Body length (incomplete) 20730, maximum width 32, width of cephalic end 11. Length of entire oesophagus 6720, length of muscular oesophagus 181, length of posterior part (broken) 14010. Muscular oesophagus without differentiation between cuticularised and non-cuticularised part. Length of stichosome 6510, approximately 35 stichocytes; long stichocytes 217 in length and 22 in width. Lateral bacillary bands 5–6 in width. Vulva situated slightly posterior to oesophago-intestinal junction, 40 from end of oesophagus; vulva not elevated, no vulvar appendage. No immature eggs seen in uterus.
Eggs in tissue. Mature eggs yellow or yellow-brown, with visible larvae; no dark-brown or black eggs found. Eggs elongate, with slightly protruding plugs. Size of eggs including polar plug, from swimbladder, JNC2166, 66.0 ± 2.6 (58–72) × 26.6 ± 1.7 (23–32) (n = 121); egg wall 2–4. For measurements of eggs from other fish and organs, see Table 3 View TABLE 3 . Fully developed larvae in most eggs. Surface of eggs apparently smooth. Superficial envelope of eggs: presence of filaments at both extremities; filaments often tightly packed at the level of plugs; when released, filaments form groups at both extremities and along length of eggs. Few unembryonated eggs, yellow-brown, showed strong longitudinal folds on egg surface; considered as aborted eggs. Aborted eggs, in which extremities do not show plugs, also showed longitudinal folds. Eggs after 24h did not change in length ( Table 3 View TABLE 3 ).
L 1 Larvae. Larvae in eggs, length 151 (140–168, n = 11), maximum width 4–5.
Mobility of larvae and hatching. Larvae in fresh laid eggs were immobile in JNC1726 and JNC2166; eggs kept in saline or seawater for four weeks (fish JNC2166) had immobile larvae and no hatching was detected. By contrast, in fish JNC2174, a few mobile larvae were seen within fresh eggs observed in saline; 24h later, the same slides kept in a humid chamber at room temperature showed 10–20% of eggs with motile larvae. Motile larvae were seen in eggs of various colours, i.e. darker eggs did not have more motile larvae than lighter eggs. These experiments show that differences existed between eggs from different individual fish. No hatching was observed.
Differential diagnosis of adults. H. longa is characterised by the longest body of all species in which the adult is known (incomplete body of immature specimen 20.7 mm vs adult females 4.9–7.5 in H. huffmani , 7.7–8.2 in H. canadensis and 11.1 in H. moraveci ).
Differential diagnosis of eggs. Eggs of H. longa are distinctly lighter (yellow to yellow-brown) than other species, in which darker eggs are dark-brown or black. Three species have egg measurements comparable to that of H. longa ( Table 1): these are H. mexicana , H. schouteni and H. balista .
H. schouteni has eggs of similar size (69–75 × 27–30 vs 66 × 27) but the eggs have superficial protuberances and no filaments as in H. longa .
H. mexicana has eggs with filaments as in H. longa but the eggs are shorter (57 × 30, this publication).
H. balista has eggs slightly longer and wider (70 × 34), the eggs have no filaments, the wall is thicker (5– 6 vs 2–4).
H. schouteni and H. mexicana have been reported from the inner layer of the swimbladder, in contrast to H. balista and H. longa , both from the outer layer.
H. filamentosa , another species from the same host, is known only from eggs. H. longa and H. filamentosa share a common morphological characteristic, the presence of thin filaments on the eggs, but have very different measurements ( Table 4 View TABLE 4 ) and were not found in the same organ. For this reason, the author considered several hypotheses concerning these two infections.
—Hypothesis 1. There is a single species of Huffmanela in G. grandoculis , which inhabits the gills, on the one hand, and the internal abdominal organs, in the other hand. Differences of eggs measurements can be attributed to different trophic sites, with much smaller eggs in the gills.
—Hypothesis 2. Similar to hypothesis 1 with a single species of Huffmanela in various organs, but the difference in egg size is related to the parasite life-cycle. Eggs from the gill mucosa are constantly dispersed in the external milieu through natural renewal of the gill mucosa, and thus have a short life-cycle; this is consistent with the presence of small eggs. In contrast, eggs from the internal organs are available for continuation of the life-cycle only at the death of the fish, and thus are longer-lasting eggs; this is consistent with the occurrence of large eggs.
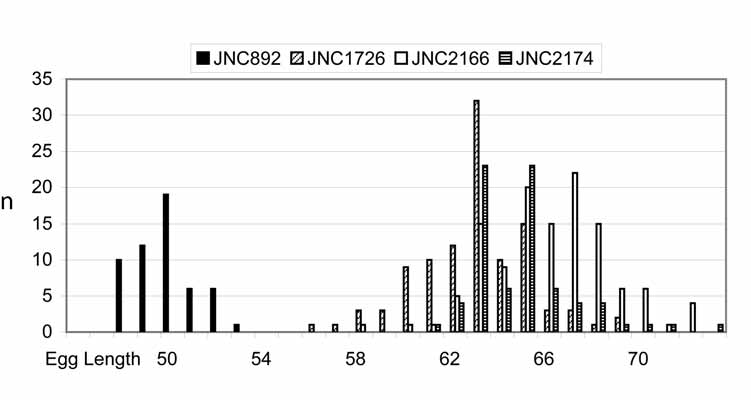
—Hypothesis 3. Two different species are present, one in the gills, H. filamentosa , and one in the internal abdominal organs, H. longa . The species are differentiated by distinct eggs measurements, with lengths not overlapping ( Figure 5 View FIGURE 5 ).
Hypothesis 1 is contradicted by observations on H. ossicola , which show that egg measurements vary only subtly in various parts of the skeletal system ( Justine 2004; this paper); however, it might be argued that the different parts of the skeletal system are not as different as between the gills and the internal abdominal organs.
Hypothesis 2 is supported by comparison with other trichinelloid nematodes in which two life cycles are possible, i.e. Calodium hepaticum (Bancroft) and Paracapillaria philippinensis (Chitwood, Velasquez & Salazar) (see Moravec 2001). However, the absence of observations on the adults of the species studied here precludes the choice of this hypothesis.
Hypothesis 3 would have been chosen without hesitation had the two species been found in two different fish species, because the eggs are extremely different. A possible argument against Hypothesis 3 is the presence in both species of thin filaments on the eggs, apparently a shared character. However, observation of similar thin filaments on eggs of H. mexicana , and on H. ossicola , probably means that such filaments are widespread in Huffmanela eggs and cannot be considered of great differential importance. This hypothesis, the presence of two distinct species, was finally preferred as the most probable, and the one which does not imply speculations on the life cycle which cannot be verified. A consequence is that G. grandoculis is the first fish in which two different species of Huffmanela have been found. H. filamentosa has not been found since its description, meaning that it is possibly rarer than H. longa , found in three fish.
More than 15 specimens of the closely related species Gymnocranius euanus were examined and none had Huffmanela infection. Several species of Lethrinus were examined (lists in Justine 2007; Rascalou & Justine 2007) and none had Huffmanela infection. This suggests that H. longa is specific to G. grandoculis .
TABLE 4. Measurements of eggs of Huffmanela filamentosa and H. longa, both from Gymnocranius grandoculis. See also histogram in Figure 5.
| Fish, organ and methods | Length | Width | n |
|---|---|---|---|
| Huffmanela filamentosa | |||
| JNC892, gills, original publication, ‘fresh eggs’, syntypes | 49.8 ± 1.3 (48–53) | 27.3 ± 1.5 (25–30) | 54 |
| JNC892, gills, old slide, ‘fresh eggs’, syntypes | 48.9 ± 1.6 (45–52) | 25.3 ± 1.3 (23–27) | 20 |
| Huffmanela longa | |||
| JNC2166, swimbladder, ‘fresh eggs’ | 66.0 ± 2.6 (58–72) | 26.6 ± 1.7 (23–32) | 121 |
| JNC2166, body wall, ‘fresh eggs’ | 65.4 ± 2.8 (58–72) | 25.5 ± 1.7 (22–29) | 58 |
| JNC2174, Swimbladder ‘fresh eggs’ | 64.7 ± 2.2 (61–73) | 24.1 ± 1.3 (22–27) | 75 |
| JNC2174, Body wall ‘fresh eggs’ | 66.1 ± 2.4 (62–73) | 25.4 ± 1.3 (22–28) | 52 |
| JNC1726, mesentery, ‘fresh eggs’ | 62.8 ± 2.4 (56–69) | 28 ± 2.2 (23–34) | 105 |
TABLE 3. Egg measurements of Huffmanela longa, JNC 2166, from swimbladder and body wall.
| Methods and characteristics of eggs | Length | Width | n |
|---|---|---|---|
| Swimbladder ‘fresh eggs’ | 66.0 ± 2.6 (58–72) | 26.6 ± 1.7 (23–32) | 121 |
| Body wall ‘fresh eggs’ | 65.4 ± 2.8 (58–72) | 25.5 ± 1.7 (22–29) | 58 |
| Swimbladder ‘ethanol-preserved eggs’ | 64.8 ± 1.8 (62–69) | 27.7 ± 1.9 (24–33) | 44 |
| Body wall ‘ethanol-preserved eggs’ | 64.3 ± 1.6 (61–68) | 28.5 ± 2.4 (24–32) | 36 |
| 24h saline body wall ‘fresh eggs’ | 65.3 ± 2.6 (60–71) | 27.3 ± 2.1 (24–34) | 97 |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |