Toxicodryas vexator, Greenbaum & Allen & Vaughan & Pauwels & Wallach & Kusamba & Muninga & Aris- Tote & Mali & Badjedjea & Penner & Rödel & Rivera & Sterkhova & Johnson & Tapondjou & Brown, 2021
|
publication ID |
https://doi.org/10.11646/zootaxa.4965.1.1 |
|
publication LSID |
lsid:zoobank.org:pub:67F8DED5-EBDB-46F8-9EAE-88B37446C9FA |
|
DOI |
https://doi.org/10.5281/zenodo.4741195 |
|
persistent identifier |
https://treatment.plazi.org/id/103C375C-4013-4029-831C-B24DFBA31614 |
|
taxon LSID |
lsid:zoobank.org:act:103C375C-4013-4029-831C-B24DFBA31614 |
|
treatment provided by |
Plazi |
|
scientific name |
Toxicodryas vexator |
| status |
sp. nov. |
Toxicodryas vexator sp. nov.
( Table 1, Figs. 1 View FIGURE 1 , 8–10 View FIGURE 8 View FIGURE 9 View FIGURE 10 )
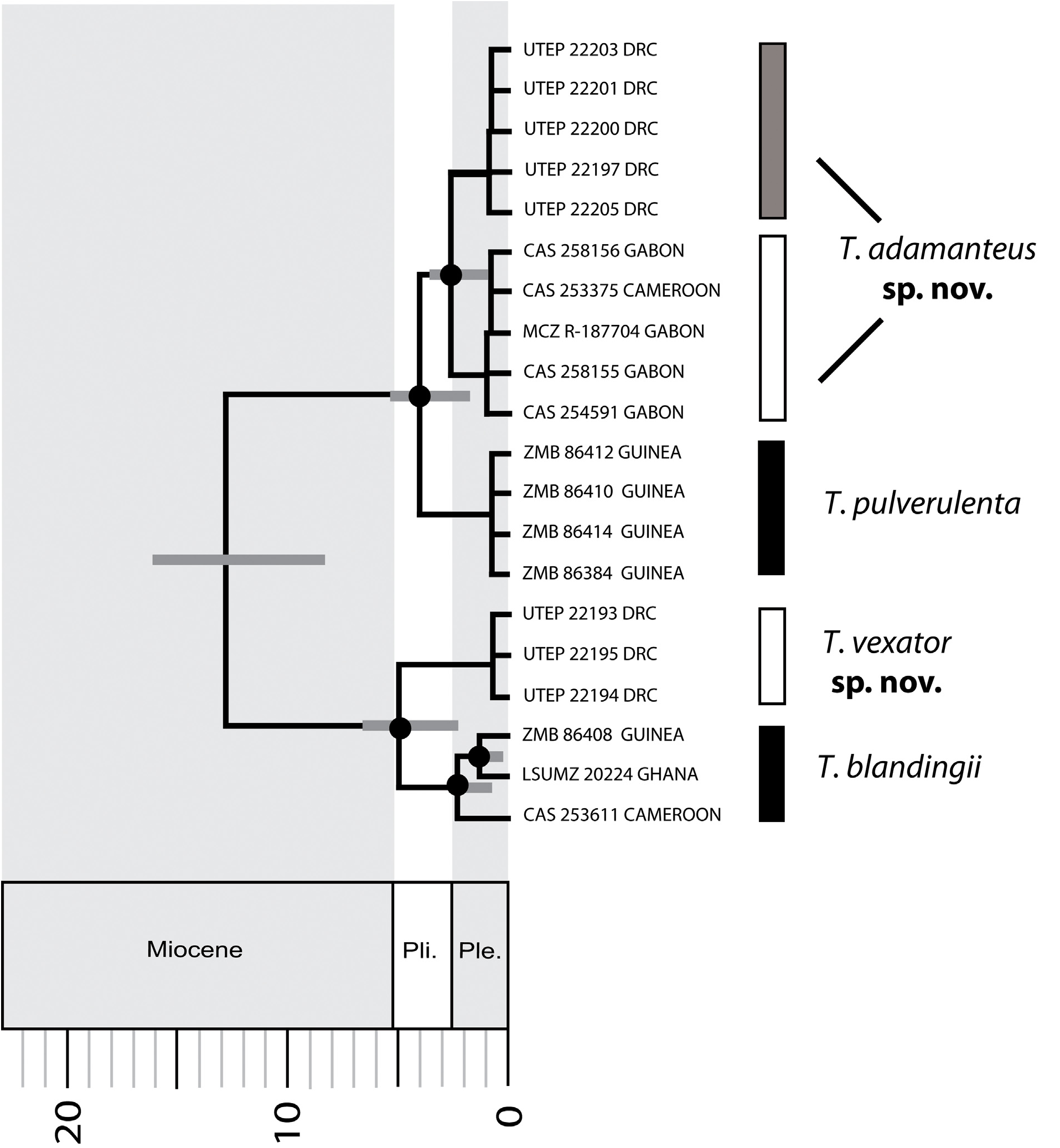
We hypothesize that this new, cryptic species occurs east of the confluence of the Congo and Ubangi rivers, and it has been considered to be conspecific with Toxicodryas blandingii since it was first documented to occur east of these rivers in DRC ( Boulenger 1919). The recognition of this new species is supported by molecular data ( Fig. 2 View FIGURE 2 ), several significant differences in scale counts (see Results), and less toxic venom in comparison to its sister taxon T. blandingii (see Venom).
Diagnosis. A species of Toxicodryas restricted to east-central and East Africa (east of the confluence of the Congo and Ubangi rivers), defined by the following combination of characters: maximum SVL> 1 meter (vs. maximum SVL < 1 meter in T. pulverulenta and T. adamanteus sp. nov.); DSRN 23–29 (vs. 19–21 in T. pulverulenta and 18–23 in T. adamanteus sp. nov.); DSRM 21–25 (vs. 19–21 in T. pulverulenta and 18–21 in T. adamanteus sp. nov.); cloacal plate divided or undivided (vs. usually divided in T. blandingii , and always undivided in T. pulverulenta and T. adamanteus sp. nov.); adult males glossy or velvety black with a yellow venter, and adult females light brown, gray, or yellowish-brown with light-brown or cream cross-bars on the flanks, with yellowish-brown venters (vs. both sexes brown to pink with darker cross-bars that often enclose a whitish spot, and the dorsum and venter sprinkled with fine dark brown or black spots in T. pulverulenta and T. adamanteus sp. nov.); hemipenis relatively short and massive (i.e., broad), proximal third covered with spines, distal two-thirds dimpled with a flattened apex (vs. relatively long with long spines mid-way along the shaft that decrease in size towards the apex and base, and with a domed apex in T. pulverulenta and T. adamanteus sp. nov.); venom toxicity LD 50 = 4.88 mg /kg in mice (vs. venom toxicity LD 50 = 2.85–3.55 mg /kg in mice for T. blandingii ).
Holotype. UTEP 22196 (field number MUSE 10341; Fig. 10 View FIGURE 10 ), adult male collected in Mulisi , Nzovu Est , Kahuzi-Biega National Park ( 02.447291° S, 28.2825378° E, 1101 m), South Kivu Province, DRC, collected by Guillain M. Mitamba, Deo Kujirakwinja, Emmanuel Muhindo, Radar Nushili, Wandege M. Muninga, and Andrew J. Plumptre on 13 November 2015. GoogleMaps
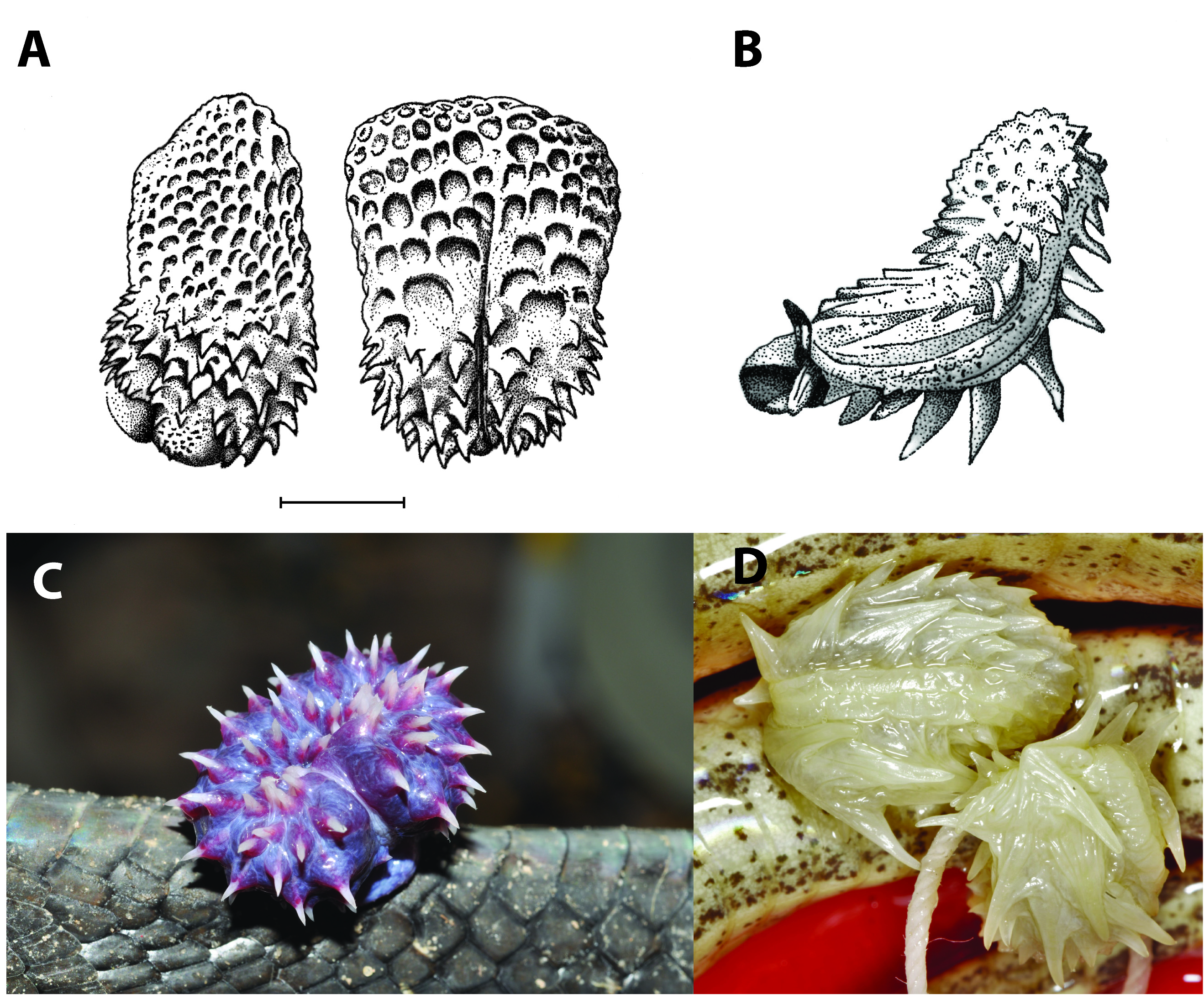
Paratypes. UTEP 22195 (field number EBG 1362; Figs. 1B View FIGURE 1 , 9C View FIGURE 9 ), adult female collected in the vicinity of Irangi ( 01.8780° S, 28.4524° E, 811 m), South Kivu Province , DRC, collected from a tree by a resident and brought to Maurice Luhumyo, Chifundera Kusamba, Mwenebatu M. Aristote, Wandege M. Muninga, and Eli Greenbaum on 30 August 2007 GoogleMaps ; RBINS 2698 (formerly RBINS 8621) (field no. Leloup #27), adult male from Bunyakiri ( 2.075630° S, 28.573194° E, 1000 m), South Kivu Province , DRC, collected by Paul Leloup on 6 March 1958 GoogleMaps .
Description of the holotype. Adult male, 1438 mm SVL; head strongly triangular and distinct from the neck, 2.1% of SVL ( 30.9 mm); interocular distance 21.7 mm, pupil elliptical, maximum horizontal eye diameter 7.5 mm; loreal near-rectangular, longer (3.0 mm) than high ( 2.4 mm), slightly tapering superiorly; body triangular; tail moderately long (32.1% of SVL). Supralabials 9/9, 4 th, 5 th, and 6 th /4 th, 5 th, and 6 th contacting orbit; infralabials 14/14, 1 st on each side in contact behind mental, 1 st –4 th /1 st –4 th contacting anterior chin shields, 5 th –8 th /5 th –8 th contacting posterior chin shields, and 1 cuneate between the 5 th and 6 th left infralabials; 2/2 preoculars; 2/2 postoculars; temporals 2 + 2/2 + 2; 2 internasals; nasal divided on both sides; frontal slightly wider ( 10.1 mm) than long ( 9.2 mm); dorsal scale rows 23 one head length posterior to jaw rictus, 21 at midbody, 15 one head length anterior to vent, smooth and oblique with apical pits visible on the neck, vertebral scales broad and apically flattened; ventrals 261 (standard), 260 (Dowling); cloacal plate undivided; paired subcaudals 136 (terminal spine missing); both hemipenes everted.
Coloration (in life) of the holotype. Solid black over the entire length of the dorsum, and black ventrally except for the anterior half of the body, where the ventral scales are pale yellow with a black posterior edge that increases in thickness in correlation with increased distance from the head. The head is black dorsally and laterally except for yellow on the lower, anterior region of the 5 th –9 th supralabials, and yellow ventrally except for thin black rims on the posterior edges of the infralabials.
Coloration (in preservative) of the holotype. The specimen’s coloration in preservative is nearly the same as in life, although the ventral yellow coloration has dulled to a yellowish off-white; some scales also seem cloudy under close lighting, indicative of some stage of ecdysis in life.
Variation. For years, the size record for this species was attributed to Goodman (1985), who recorded a total length of 2515 mm for an adult female collected in Uganda. However, Hedges (1983:20) measured a specimen from Kenya that was 2707 mm total length, and said it “is reputed to exceed 3300 mm.” Weinstein et al. (2011) noted the species may exceed 3.5 meters. Spawls & Branch (1995, 2020) listed the maximum size as “about 2.8 m ” (no specific location provided), but Weinstein (pers. comm.) estimated a captive specimen from Kakamega was approximately 3.0 meters total length.
Schmidt (1923:104) remarked that two specimens from DRC had fused prefrontals, creating a single transverse scale. Five of his 20 DRC specimens had a divided cloacal plate, “with indications of a groove in two others,” and as a result, he stated that the condition of the cloacal plate is “obviously useless” as a diagnostic character. Laurent (1956) noted that, with the exception of DRC specimens from Mayombe (north of the Congo River in Kongo Central Province and thus attributable to T. blandingii as recognized herein) with consistently divided cloacal plates, 13 of 31 snakes from other areas of DRC had an undivided cloacal plate. Our examined specimens confirm this sentiment, because 17/ 92 specimens have a divided cloacal plate, and an additional five specimens have a partially divided cloacal plate. Hellmich (1957b) noted maximum sizes of Angolan males ( 1740 mm SVL; 523 mm TL) and females ( 1730 mm SVL; 505 mm TL), and aberrant individuals that had 11 supralabials and 9–14 infralabials. De Witte (1966) noted that the species has 17 scale rows at midbody, but this datum is so aberrant that it is likely an error ( Pitman 1974; Table 1). Skinner (1973) provided ranges of scale counts (161–274 ventrals, 86–147 subcaudals) that are so aberrant that they are clearly erroneous (at least for lower counts), and noted a maximum length of about 3 meters. For Uganda snakes, Pitman (1974) listed ventral scale counts from 240–260 (males) and 240–259 (females). De Witte (1975) noted a single individual with 25 scale rows at midbody, and Broadley et al. (2003) also noted 25 scale rows as the maximum amount of variation. Based on two specimens from Lukolela, Belgian Congo (AMNH 45907) and Akenge, Belgian Congo (AMNH 12243), Bogert (1940:61) described their maxillary teeth as “ten in number, followed after a short diastema by three enlarged, grooved fangs, the posterior one of which is smaller than the other two.”
Schmidt (1923:105) described two color phases of DRC specimens, including (1) a black phase with the anterior portion of the venter yellow, each ventral scale bordered with black on its posterior edge, the border increasing in width until the yellow color disappears on the posterior two-thirds of the venter; and (2) a brownish phase, “with more or less distinct wide dark cross-bars, confluent anteriorly, alternate posteriorly on the vertebral line.” Pitman (1938:211) described an unsexed individual from Uganda as “darkish gunmetal with a purplish mottling towards the tail, belly pale yellow from head to tail but with slight brown markings on the posterior half increasing in occurrence towards and on the tail.” A female from Uganda was described as rich chestnut dorsally and ventrally, with handsome chocolate blotches on the flanks, each one containing a small white spot. On the posterior part of the body, including most of the tail, these blotches were confluent with pale edging. The ventral aspect of the head and the first 50 ventrals were “paly yellowish tinged green.” The dorsum of the head was dark brown, and the posterior supralabials were dull greenish grey. The supralabials had black edging posteriorly, except for the last one. There were two elongated dark brown blotches behind the eye, which was iridescent hazel.
Bogert (1940:61) provided data for a specimen (AMNH 45907; determined to be a subadult male based on photographs examined by EG) that had been described by James P. Chapin as “olive-brown, with dark brown patches; below grayish brown.” Hellmich (1957b) described the coloration of several individuals from Angola, including an adult male that had a blue-reddish-black dorsum with a venter that was pale yellow in the front half of the body, with an ever-widening blue-black border that eventually enclosed the entire ventral scales towards the posterior side. Another adult male had a dark black-brown dorsum, and light yellow venter on the first third of the body, but starting on the 5 th ventral, there was a dark gray-blue spot that increased in size posteriorly until it covered the entire venter towards the posterior end of the body. An adult female’s dorsum was described as clay yellow with mostly alternating dark transverse bars and a pale yellow venter. A second adult female had similar coloration to the latter specimen, but the transverse bands were only faintly visible on the posterior sixth of the body and tail. A juvenile dorsum was described as gray-brown with transverse black-brown spots that converge in the anterior part of the body, but then alternate posteriorly, with a light yellowish-white spot in the lower center of the spots.
Laurent (1956:195) listed a section about possible sexual dimorphism for this species, but stated that nothing glaring appears in his data. Pitman (1974:125) described several individuals from Uganda that were similar to the descriptions above, and added that both color phases “exhibit a handsome suede effect.” He noted that although subadult males might retain the brown phase, black females are unknown, and the black and brown color phases have “sexual significance.” The dark blotches of juveniles were noted to vary from “blackish, chocolate, reddishbrown or dark or bright chestnut,” with interspaces ranging from light gray, pale brown or chestnut, sometimes with a pinkish tinge. Goodman (1985:56) described two adult females from Uganda as “dingy olive-brown.” Hughes (2000) noted that Laurent (1964) seemed to imply that his largest male specimen from Dundo, Angola was not black, but this is not clear, because the latter author only mentioned in passing that a smaller male from Andrada, Angola represented the black phase. Spawls & Branch (1995, 2020) remarked that the yellow ventral pigmentation might form a stripe in the middle of the venter. In the brown color morph, the skin between the scales is bluish gray, and especially visible when the snake inflates its body during a threat display. The eye can be yellowish or brown. The black phase snakes are usually male, whereas the brown phase ones are usually female. Based on photos of an adult male from Banalia, DRC (individual shown in Fig. 9A View FIGURE 9 ), the base of the tongue is bluish black, and the forked tip is silvery gray.
Hemipenis. Bogert (1940:61) described the hemipenis of an individual, presumably from DRC, as “not bifurcate, extending to the tenth caudal, the undivided sulcus deeply buried between two fleshy fold [sic]. The basal half is heavily armed with stout spines but distally the organ is calyculate, the edges of the calyces crenulated.” Hemipenes of our examined specimens had a simple, subcylindrical shape, simple sulcus spermaticus, and spinose ornamentation with a rough apical structure, as also noted for T. blandingii . Because only two specimens with everted hemipenes were available for both T. blandingii and T. vexator sp. nov., any finer-scale differences cannot be attributed to species-level differentiation ( Dowling & Savage 1960; Doucet 1963).
Diet. Laurent (1964) noted a juvenile snake contained the remains of a bird. In snakes from Uganda, Pitman (1974) noted the diet included weaver finches, robber birds’ eggs, small rodents, bats, frogs, large agamids, and chameleons. Hedges (1983) described a Kenyan snake that regurgitated a Nectarinia famosa sunbird. Spawls & Branch (1995, 2020) described the diet as birds, bird eggs, arboreal lizards, frogs, arboreal rodents, and bats. Nagy et al. (2011) documented an adult female from DRC that predated a Short-palated Fruit Bat ( Casinycteris argynnis ) from a bat net in secondary forest. Another male specimen from DRC was captured with a nestling bushshrike ( Laniarius sp. ) in its stomach.
Parasites. Loveridge (1937) noted a tick on the scales of a specimen (ANSP 20504) from Belgian Congo. Parasites of Ugandan snakes included internal nematodes ( Kalicephalus sp. ) and cestodes, ectoparasites (ticks, including Aponomma latum ), and captive specimens had bacterial infections, including mouth rot ( Pitman, 1974). Goodman (1985:56) documented dozens of ticks, “probably Aponomma sp. ,” and linguatulid worms from the posterior saccular lung cavity of an adult female from Uganda.
Behavior. Spawls & Branch (1995, 2020) described the snake as arboreal, sometimes climbing to heights of 30 meters in trees, but also descending to the ground to cross open spaces and roads. It is mostly nocturnal, sheltering in leaf clumps and tree hollows during the day. When threatened, it will open its mouth widely and expose the pink lining inside, inflate its body, flatten the head, lift the anterior part of its body off the ground and into C-shaped coils, and strike at perceived threats. It is adept at smelling sleeping birds in nests at night, and will make a slow, deliberate approach to attack them.
Reproduction. A personal communication from “Leakey” (most likely Richard Leakey) to Pitman (1974) noted that a female from Kakamega ( Kenya) laid 9 eggs that were approximately 20 x 40 mm on 9 January. Spawls & Branch (1995) reported an average of 7– 14 eggs in this species ( 3–14 in Spawls & Branch 2020), presumably based mostly on East African data, which is substantially more than clutch sizes reported for T. blandingii by Luiselli et al. (1998a).
Habitat. Laurent (1954) remarked that an adult female from Dundo, Angola was captured after it fell out of a mango tree. Laurent (1960) stated that his colleague M. Leloup often encountered the species in eastern DRC. De Witte (1962) listed the habitats for DRC snakes as equatorial forest and gallery forest, a classification that was repeated by Thys van den Audenaerde (1965). Pitman (1974) also limited the species to forest in Uganda, but he remarked that the species frequently enters houses (where it hunts for bats), and one individual was killed in a tree over 20 meters above the ground. Broadley & Cotterill (2004) stated the species inhabits forests and wooded savannas. Spawls & Branch (1995, 2020) noted the species from forest, woodland, forest-savanna mosaic, riverine woodland, and human habitations from sea level to about 2,200 m elevation. The holotype was found basking on a shrub (ca. 4 meters above ground) in a clearing created by a fallen tree in primary forest. The Irangi paratype was found in secondary forest near a road.
Geographic distribution. Based on molecular data from Allen et al. (in press) and patterns of our morphometric data ( Table 1), we hypothesize that this species occurs east of the Congo and Ubangi Rivers in CAR, DRC, Angola, Zambia, South Sudan, Uganda, Kenya, and Tanzania. Although Boulenger (1896) listed a specimen from “ Zanzibar,” the locality likely referred to mainland records ( Parker et al. 1940; Loveridge 1957; Pitman 1974).
Venom. Wakeman (1966) documented a 2.59 m (presumably total length) captive adult from Uganda that struck an adult rat, which died after 8.5 minutes. Pitman (1974:127) described the case of a young European who was bitten on the thumb three times in rapid succession by a small ( 305 mm) individual (erroneously reported as 350 mm by Goodman 1985); only “initial smarting” at the puncture sites occurred. Spawls (1979) described the bite of a captive 2.1 meter snake on a man, including three fang punctures on his thumb, which resulted in mild swelling of his hand. Anecdotal observations of a bite by a captive snake on a mouse resulted in death in under a minute. Hedges (1983:20) was bitten, “and although it drew a considerable amount of blood, I felt no effects whatsoever.” He also remarked that this species “chews” when biting, ostensibly to ensure the rear fangs penetrate the flesh to inject venom. Based on venom collected from a snake originating from Kenya, Weinstein & Smith (1993) estimated the LD 50 value as 4.88 mg /kg in mice. Weinstein & Smith (1993:88) also noted “the differences in lethal potencies between the two samples of B. blandingi [compare with LD 50 = 2.85–3.55 mg /kg in T. blandingii from West Africa] demonstrate variation of secretion toxicity in this species,” suggesting possible species-level differences in venom composition of these sister taxa ( Weinstein & Kardong 1994). Immunological cross reactivity with elapid venom antisera was demonstrated, but Weinstein & Smith (1993) did not distinguish between samples originating from West Africa (i.e., T. blandingii ) or East Africa (i.e., T. vexator sp. nov.). Branch (2005) suggested bites cause nausea and headaches, but fatalities are unknown. Dashevsky et al. (2018) included a Toxicodryas from Tanzania in their proteomics study, and they noted its venom is dominated by three-finger toxins.
Scott Weinstein (pers. comm., October 2020) clarified that a snakebite mentioned by Weinstein & Smith (1993) and a second one that occurred one year after this publication were caused by snakes originating from Kakamega forest, Kenya. “The bite mentioned in the paper was delivered by [an approximately] 3.0 m jet black male that was a favourite member of my collection for nearly 7 years. He typically performed the whole gamut of open mouthed posturing and mock strikes, but was also a very aggressive feeder, and although I tried to condition him to receiving food (freeze-thawed chicks and rodents) out of his enclosure, he still occasionally would ‘pour’ out when he detected food nearby. The bite occurred just after I opened the front [of] the cage (a tall arboreal habitat); he ‘poured’ out and delivered what was clearly a feeding strike with firm attachment on the ulnar zone, dorsal aspect [of the right] wrist. Attachment lasted approximately 30–40 seconds with attempted jaw advancement; I was able to detach him by gently [maneuvering] his head. Pain was akin to several punctures with a sewing needle; immediate bleeding was proportional to the wounds and transient (<3 min). I developed some very slight edema also proportional to the extent of physical trauma, but did have some persistent erythema (lasting approximately 36 hr), and some slight pruritus that also lasted a couple of days. The second bite was from an approximately 45 cm male and was [a] quick bite and release delivered to the lateral aspect of the [right] index finger, consistent with a brief defensive bite. I only experienced some sharp punctures, very brief, slight bleeding (lasting barely a minute) and a few minor lacerations from a few of the teeth inflicting a ‘dragging’ effect; there were no other effects. It should be noted that although most of the minor effects of the first bite (the second was too brief to assess) could be assigned to local effects of low volume venom inoculation, some contribution of local hypersensitivity is certainly possible. I was sensitised in the 1970s during the beginning of my venom research.”
Etymology. The specific epithet is a noun in apposition, invariable, from the Latin noun vexator , meaning harasser or stalker, in reference to the fact that this snake stalks prey when they are sleeping, and to its aggressiveness when disturbed.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |