Marchantia polymorpha, L.
|
publication ID |
https://doi.org/10.1016/j.phytochem.2020.112533 |
|
DOI |
https://doi.org/10.5281/zenodo.8302135 |
|
persistent identifier |
https://treatment.plazi.org/id/24402D47-4364-FFEE-0B28-9420FD9BF8CC |
|
treatment provided by |
Felipe |
|
scientific name |
Marchantia polymorpha |
| status |
|
2.2. Oxylipin profiles of M. polymorpha View in CoL View at ENA
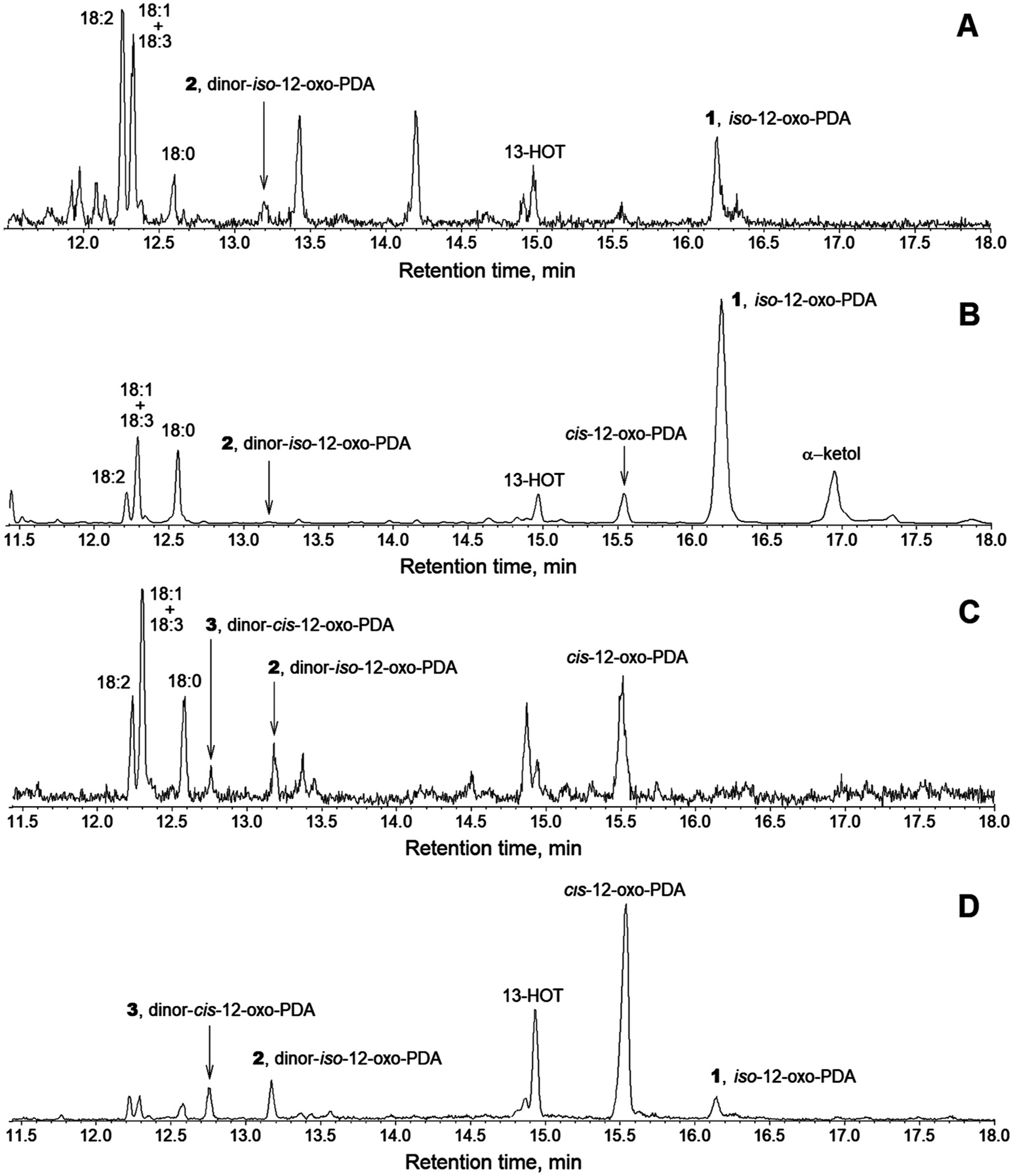
The GC-MS profiles of M. polymorpha oxylipins ( Fig. 1C and D View Fig ) possessed a general similarity to those registered for P. patens ( Fig. 1A and B View Fig ). The AOS products, namely the cyclopentenones, constituted a majority. At the same time, the product patterns were not identical. Firstly, the endogenous M. polymorpha oxylipins ( Fig. 1C View Fig ) possessed much lower content of iso -12-OPDA ( 1) than P. patens . The same thing concerned the M. polymorpha oxylipins biosynthesized in vitro from the exogenous α linolenic acid ( Fig. 1D View Fig ). The major oxylipin in M. polymorpha profiles ( Fig. 1C and D View Fig ) was the cis -12-OPDA. While the iso -12-OPDA ( 1) content was significantly lower than in P. patens , the shorter chain homologue dinor- iso -12-OPDA ( 2) was more abundant ( Fig. 1C and D View Fig ). One more peculiarity of M. polymorpha oxylipin profiles was the presence of product 3, missing in P. patens . The mass spectrum of compound 3 is presented in Fig, 2C. It possessed M + at m/z 278 (10%), as well as the isomer 2. The spectrum well matched that described before for 2,3-dinor-12-OPDA ( Weber et al., 1997). Both cyclopentenones 2 and 3 were present in M. polymorpha endogenously along with the 12-OPDA, in full agreement with the recent data ( Monte et al., 2018).
Besides with the above-described products, M. polymorpha possessed some other oxylipins, for instance, the hydroxy fatty acids 13-HOT and 11-HHT (ca. 4:1, as estimated by the total ion current chromatogram integration). The hydroxy derivatives of arachidonic and eicosapentaenoic acids were present only at trace levels. The HPL products were significantly less abundant than in P. patens .
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |