Thecadactylus solimoensis, Bergmann & Russell, 2007, Bergmann & Russell, 2007
|
publication ID |
https://doi.org/10.1111/j.1096-3642.2007.00251.x |
|
persistent identifier |
https://treatment.plazi.org/id/422087C2-B22A-602F-FCD8-4488FBC1FB6A |
|
treatment provided by |
Felipe |
|
scientific name |
Thecadactylus solimoensis |
| status |
|
DESCRIPTION OF THECADACTYLUS SOLIMOENSIS
SP. NOV.
Synonymy: See Russell & Bauer (2002a) for complete list.
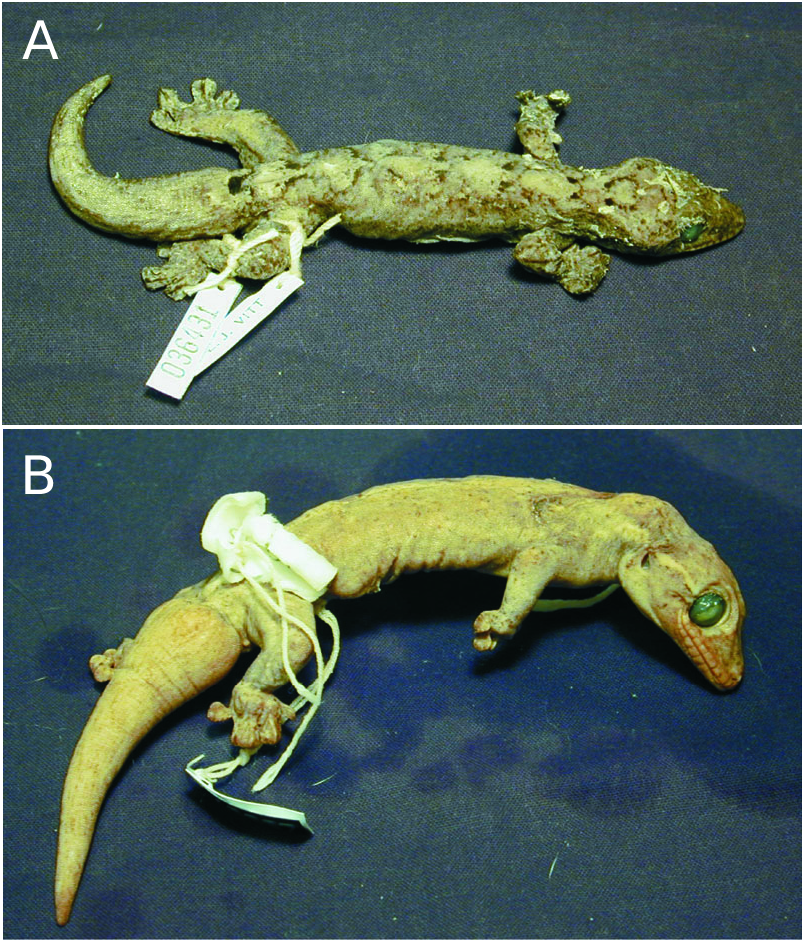
Material examined: Holotype: QCAZ-6691 (= OMNH- 36431). Male, collected by L. J. Vitt in 1994 from the type locality of Reserva Faunistica Cuyabeno ( Estacion Biologia da Universidad Catolica ), Sucumbios, Ecuador. Description ( Fig. 7A View Figure 7 ): 116 mm SVL, 76 mm TL, 29 mm HL, 24 mm HW, 6 mm IND; regenerated tail broader than more proximal stump and relatively unpatterned; 4 nasal granules, dorsalmost enlarged; internasals with reduced contact medially; 11 supralabials, 6 anterior to the eye; 10 infralabials; rostral partly divided; 11 spinose supraciliaries; postmetals stubby; 3 post-postmetals, medial one enlarged; 9 post-infralabials; dorsal forearm and ventral neck scales granular; hindlimb web and tail base scales imbricate; 1 single-apexed, pointy cloacal spur; cloacal sacs present; total manual lamellae on digits I, IV and V, 15, 21 and 19, respectively; total pedal lamellae on digits I, IV and V, 18, 22, 20, respectively; scansors on manual and pedal digits IV, 12 and 13, respectively; subdigital sulcus divides all lamellae on manual digit II, but only some on digit I; lateral stripe absent; dorsal colour and pattern light, unmottled, consisting of 5 diamonds; 7 paravertebral pairs of spots, of varying definition, some interconnected across the midline; nuchal spot absent; postocular stripes project posterodorsally, but do not connect middorsally; dark caudal blotches present; 13 teeth occluded by rostral; anterolateral process of parietal swept back; otic capsule anterolateral to occipital complex; 26 presacral vertebrae, 3 cervical vertebrae without ribs; 22 ribs, 2 anterior to medial level of clavicles, 6 shortened; cloacal bones absent; paraphalanges not visible in radiographs.
Paratypes: All paratypes were collected by L. J. Vitt in 1994 from the type locality, but from Neotropic Turis, instead of Estacion Biologia. Five paratypes are defined: OMNH-36427: male, original tail; OMNH- 36428: female, original tail; OMNH-36429: male, regenerated tail; OMNH-36430: male, regenerated tail; and OMNH-36433: female, regenerated tail.
Referred specimens: All those listed in Appendix 1 that originate from Bolivia, Peru and Ecuador. Also, those from the Brazilian states of Amazonas and Rondonia. Tentatively, the single specimen from the southern Colombian state of Caqueta is also referred.
Variation: Presented only for characters that do not span the entire range for Thecadactylus . 2–5 nasal granules; 8–14 supralabials, 4–7 anterior to the eye; 8–12 infralabials; rostral never fully divided; up to 12 spinose supraciliaries; 2–7 post-postmentals; 4–11 post-infralabials; 1–3 cloacal spurs; total manual lamellae on digits I, IV and V, 13–20, 17–25, 16–24, respectively; total pedal lamellae on digits I, IV and V, 14–21, 19–25, 18–23, respectively; scansors on manual and pedal digits IV, 9–14 and 10–14, respectively; 0–7 dorsal diamonds; 0–8 paravertebral spots; 0–5 dark caudal blotches; 0–10 caudal bands; 11–15 teeth occluded by rostral; 26–27 presacral vertebrae, 2–3 cervicals without ribs; 21–24 pairs of ribs, 0–3 anterior to medial level of clavicles, 5–6 shortened.
Although both species of Thecadacytlus are highly variable in general, T. solimoensis displays less variability than does T. rapicauda . This is especially true in terms of maximal SVL ( T. solimoensis specimens are relatively large) and in terms of pattern ( T. solimoensis specimens tend to have a dorsally directed postocular stripe and a dorsal diamond pattern, see below).
Diagnosis: Thecadactylus solimoensis invariably possesses strongly dilated subdigital pads, well-developed subdigital sulci that house the claws and divide the lamellae into two series, and interdigital webs, uniting it with T. rapicauda within the genus. T. solimoensis also possesses morphological, morphometric and molecular autapomorphies that distinguish it from T. rapicauda . Morphological and morphometric divergence is less pronounced than molecular divergence, which includes sequence divergences of 23.0–26.9% between the two species ( Kronauer et al., 2005).
Morphological characters that diagnose T. solimoensis are never fixed, but are possessed by the majority of specimens. This is unsurprising for a cryptic species, and similar characters have been used as diagnostic by other authors ( Branch, Bauer & Good, 1996; Wiens & Penkrot, 2002). Four morphological characters are here denoted as diagnostic. The strongest of these is that 78% of specimens examined have a dorsally directed postocular stripe, compared with <0.01% of specimens of T. rapicauda . Seventy-seven per cent of those specimens examined have a dorsal diamond pattern (39% of T. rapicauda ), and 69% possess stubby postmental scales (53% of T. rapicauda ). Finally, 65% of specimens assigned to T. solimoensis have granular scales on the gular region (25% in T. rapicauda ).
All morphometric characters examined have a high degree of range overlap between T. solimoensis and T. rapicauda . However, significant differences occur in some, when size-removed (residual data set) species means are compared using two-sample t -tests or Mann–Whitney U -tests, as appropriate ( Table 4). Mean values for T. solimoensis are significantly greater for internasal distance, interorbital distance, ear–eye distance, axilla–groin distance, metatarsus length, fourth toe width and first toe length than for T. rapicauda . Means are significantly less for eye– naris distance, orbital diameter, upper arm length and crus length than for T. rapicauda ( Table 4).
Cytochrome b sequences between the two species are highly distinct ( Kronauer et al., 2005; see above). As molecular evidence provides very strong support for the recognition of a second species ( Seberg, 2004) and broad geographical samplings of numerous specimens for each species were included, diagnostic molecular characters are detailed ( Table 5). A smaller cyt b sample size would tend to inflate these differences and negate the use of this approach. Of 584 bp sequenced, 26 are fixed and exclusive to T. solimoensis . A further 9 bp are fixed and almost exclusive to T. solimoensis in that only a single specimen sequenced of T. rapicauda shares the same nucleotide. Within the sequenced fragment, a highly diagnostic region, located between nucleotide positions 489 and 532, contains 12 of the 35 fixed sites, ten of which are exclusive ( Table 5).
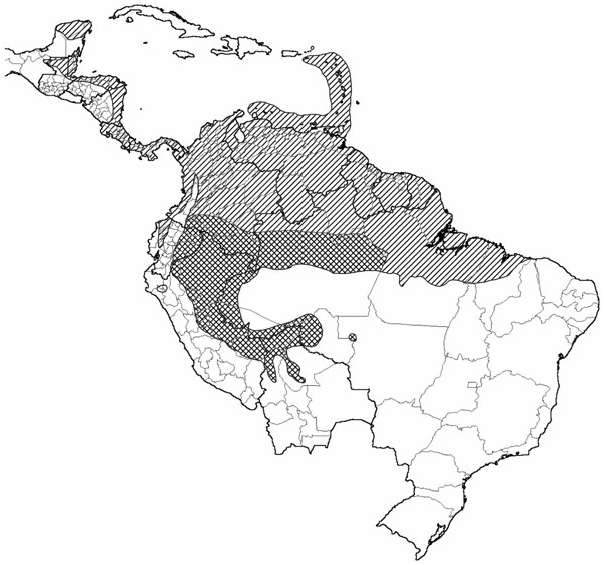
Distribution: T. solimoensis occurs throughout Ecuador, Peru and Bolivia, only east of the Andes, in Brazilian Amazonas and Rondonia, and in southern Colombia ( Fig. 1 View Figure 1 ). The western and southern extent of its range is absolutely delimited by the edge of the range of Thecadactylus , with the western edge bounded by the Andes, and the south-eastern extent of the range defined by the edge of mesic Amazonia. The northern extent of the range is difficult to determine with current specimen availability, but may be defined by the Colombian Cordillera Oriental. The eastern range boundary tentatively approximates the political boundary between the Brazilian states of Amazonas and Pará, as inferred from molecular sampling ( Kronauer et al., 2005). More intensive sampling is required to determine the northern and eastern boundaries more definitively.
Etymology: The specific epithet, solimoensis , is a locative adjective referring to the drainage of the Solimões River, representing the headwaters of the Amazon River, and draining much of the area in which Thecadactylus solimoensis occurs.
| IND |
Indiana University |
| V |
Royal British Columbia Museum - Herbarium |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.