Axianassa heardi, Anker, Arthur, 2011
|
publication ID |
https://doi.org/10.5281/zenodo.202972 |
|
DOI |
https://doi.org/10.5281/zenodo.6186746 |
|
persistent identifier |
https://treatment.plazi.org/id/667AC75A-174C-FF9A-66E0-B17447AFC88A |
|
treatment provided by |
Plazi |
|
scientific name |
Axianassa heardi |
| status |
sp. nov. |
Axianassa heardi View in CoL n. sp.
( Figs 9–13 View FIGURE 9 View FIGURE 10 View FIGURE 11 View FIGURE 12 View FIGURE 13 )
Type material. Holotype: male (CL 4.65 mm), QM W29049, Australia, Queensland, Great Barrier Reef, Lizard Island, Casuarina Beach near Lizard Island marine station, 14°40’31.4”S, 145°26’39.5”E, sand flat with corals and coral rubble, depth 1 m, suction (yabby) pump, from burrow, coll. A. Anker, 24 February 2009, fcn AUST-1797.
Additional material. 1 female (CL 5.3 mm), FLMNH UF Arthropoda 23957, French Polynesia, Society Islands, Moorea, channel between NW Motu islands, 17°29’21.5”S, 149°54’47.5”W, sand flat with some large and small rocks and coral “bommies”, 0–2 m, hand, A. Anker, 10 November 2009, fcn BMOO 8770.
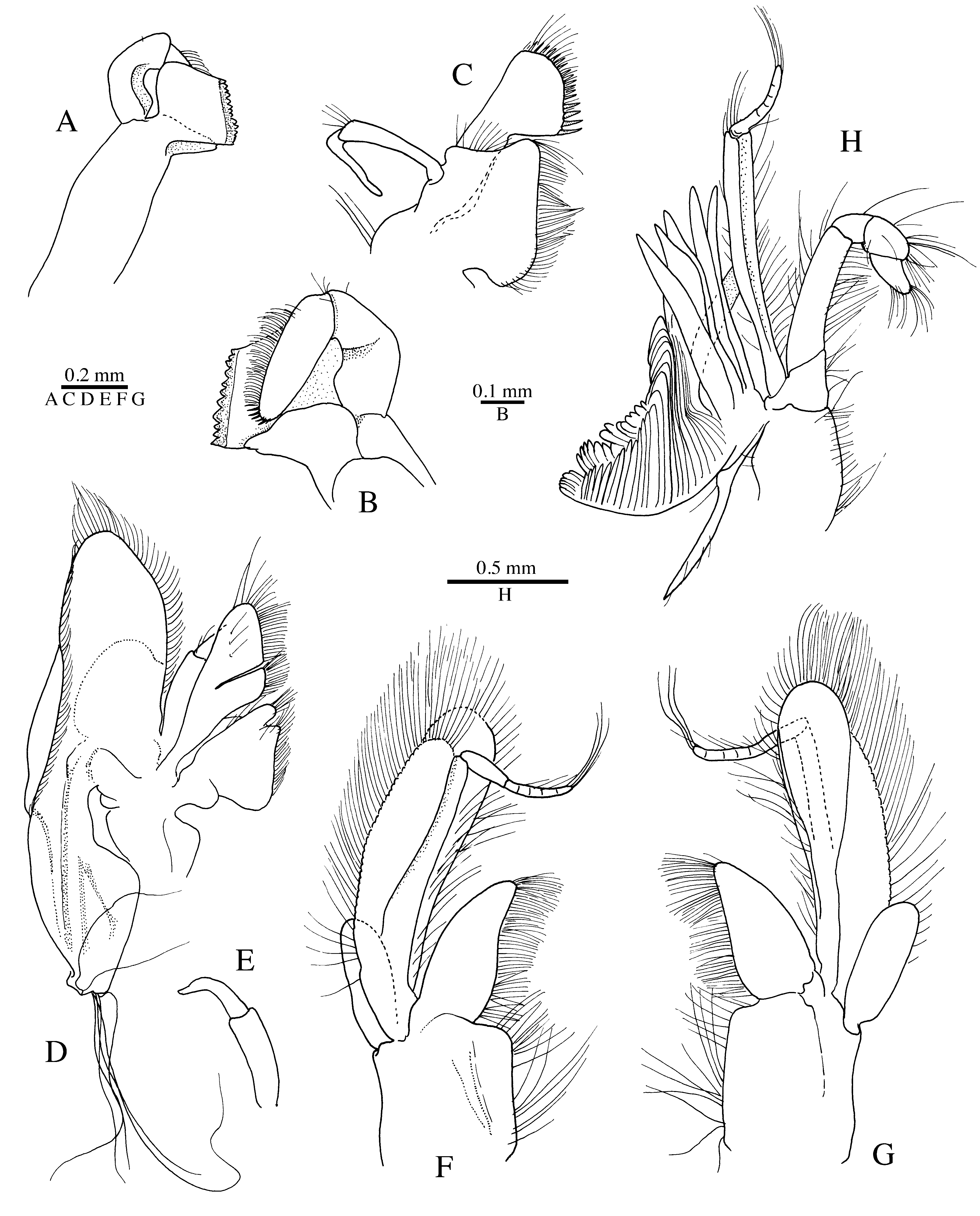
Description. [based on male holotype] Body and appendages sparsely covered with conspicuous, long, erect setae ( Fig. 9 View FIGURE 9 A, 12). Carapace with straight linea thalassinica and well-defined, crescent-shaped cervical groove; pterygostomial region broadly rounded, fringed with setae ( Fig. 9 View FIGURE 9 A, C). Rostrum about as long as broad at base, reaching far beyond anterior margin of eyestalks, with rounded apical tooth and three teeth on each lateral margin, most posterior lateral tooth being much less than conspicuous than most anterior lateral tooth ( Fig. 9 View FIGURE 9 B, E).
Abdomen thinly sclerotised, smooth; first abdominal somite with pleuron ventrally produced into fairly strong blunt process ( Fig. 9 View FIGURE 9 A); posterior somites without dense lateral fringes of plumose setae. Telson broad, tapering posteriorly, without armature, posterior margin broadly rounded ( Fig. 9 View FIGURE 9 I).
Eyestalks rounded, with large cornea occupying entire terminal portion ( Fig. 9 View FIGURE 9 D), pigmented blackish in alcohol-preserved specimen, white-yellowish in life ( Fig. 12 View FIGURE 12 ).
Antennular peduncle with third article subcylindrical, slender, not reaching half-length of fourth article of antennal peduncle ( Fig. 9 View FIGURE 9 A, B). Antennal acicle dagger-like, tapering distally, reaching to about 0.3 length of third article of antennular peduncle; fourth article subcylindrical, very long, slender, furnished with elongate setae on lateral and ventrolateral surface ( Fig. 9 View FIGURE 9 A); flagellum well developed, reaching well beyond chelipeds.
Mouthparts more or less typical for genus (cf. Kensley & Heard 1990: fig. 2D–I; Rodrigues & Shimizu 1992: figs. 4–8; Anker 2010: fig. 2). Mandible with triarticulated palp (first two articles possibly partly fused); distal article densely furnished with setae, most distal setae brown-blackish in colour; molar and incisor processes partly fused; molar edge without teeth (except for one blunt distal bump); incisor edge with 11 small subacute teeth ( Fig. 10 View FIGURE 10 A, B). Maxillule with biarticulated palp, coxal endite very setose; basal endite with stout spine-like setae in addition to more slender setae ( Fig. 10 View FIGURE 10 C). Maxilla with broad scaphognathite, bearing at least six long, flexible setae on ventral (= posterior) margin; endopod biarticulated, distal article slightly narrower than proximal, tapering ( Fig. 10 View FIGURE 10 D, E). First maxilliped with broad, paddle-shaped endopod; exopod triarticulated, basal article broad, paddle-shaped, distal articles much narrower; epipod present as large oval-shaped lobe ( Fig. 10 View FIGURE 10 F, G). Second maxilliped typical for Axianassa , with elongate epipod bearing well-developed podobranch; endopod robust, with apical “head” formed by propodus and dactylus; exopod triarticulated ( Fig. 10 View FIGURE 10 H). Third maxilliped pediform; epipod bifid, with well-developed podobranch; ischium with crista dentata bearing at least 14 teeth; merus with small acute tooth distolaterally; bands of fusiform setae grouped in well-delimited areas present on mesial side of merus, carpus, propodus and dactylus, ventral margin of these articles with long simple setae, latter especially dense on dactylus ( Fig. 11 View FIGURE 11 A, B).
First pereiopods (chelipeds) stout, unequal in size by about 20%, slightly asymmetrical in shape. Major cheliped very robust; ischium with about eight small teeth on ventral margin, most distal tooth largest and most conspicuous; merus with ventral and dorsal margins inflated, ventral margin with at least eight small teeth proximally and one larger projecting tooth at about mid-length; carpus cup-shaped, distally widening, unarmed; chela ovate, compressed laterally, smooth, with fingers about 0.7 palm length, stout, with crossing tips; cutting edge of pollex with two large subtriangular teeth and a series of smaller teeth proximally; cutting edge of dactylus with at least one larger tooth ( Fig. 11 View FIGURE 11 C–E). Minor cheliped generally similar to major cheliped, with slightly smaller, more slender chela; teeth on cutting edges of pollex less pronounced ( Fig. 11 View FIGURE 11 F).
Second pereiopod pediform, robust; merus, carpus and propodus with long flexible setae along ventral margin; dactylus about half as long as propodus, simple ( Fig. 11 View FIGURE 11 G). Third pereiopod moderately stout; ischium, merus and carpus smooth, moderately setose; propodus with distoventral brush(es) of stiff setae; dactylus slightly shorter than propodus, somewhat sickle-shaped; dorsal margin bearing at least six stout corneous spines; distoventral margin slightly expanded, carrying row of short spiniform setae ( Fig. 11 View FIGURE 11 H). Fourth pereiopod similar to third pereiopod, slightly shorter and more slender. Fifth pereiopod much more slender than third or fourth pereiopods; propodus subchelate, ending in short subacute tooth (obscured by setae); distal half of propodus with brush of stiff setae ( Fig. 11 View FIGURE 11 I); dactylus subspatulate, with deep ventroproximal excavation opposed to subacute tooth of propodus.
First pleopod absent. Second to fifth pleopods similar, biramous, typical for genus. Uropods with broadly ovoid exopod and endopod; exopod with blunt distolateral tooth adjacent to movable spiniform seta, sometimes with a second much smaller tooth on lateral margin above distolateral tooth, and with distinct transverse suture furnished with about 12 small denticles; endopod with one strong subacute tooth on lateral margin and with several subacute teeth on dorsal surface as illustrated ( Fig. 9 View FIGURE 9 G, H).
Gill/exopod formula possibly* as described for genus (cf. Kensley & Heard 1990), except for the presence of well-developed (vs. rudimentary) epipod on the first maxilliped (*note: some arthrobranchs were detached during dissection).
Colour pattern. Uniform hyaline white; ovary orange ( Figs. 12 View FIGURE 12 , 13 View FIGURE 13 ).
Etymology. Named after Dr. Richard Heard (Gulf Coast Research Laboratory, University of Southern Mississippi, Ocean Springs, MS, USA), for his contributions to decapod taxonomy, including the co-authorship in the revision of the American species of Axianassa ( Kensley & Heard 1990).
Habitat. Lizard Island: sand flat with large coral heads and coral rubble, at depth of about 1 m, in burrows (same collection site as for Athanopsis saurus n. sp.); Moorea: shallow channel between two small islands, with large areas of sand between rocks and coral “bommies”.
Type locality. Lizard Island, Great Barrier Reef off Queensland, Australia.
Distribution. Lizard Island, northern section of the Great Barrier Reef (type locality), and Moorea, Society Islands, French Polynesia.
Variation. The female specimen from Moorea generally agrees with the male holotype, except for having a stouter major cheliped, with a more pronounced tooth on the cutting edge of the dactylus ( Fig. 13 View FIGURE 13 ), and bearing a pair of slender uniramous pleopods on the first abdominal somite, as is typical for females of Axianassa .
Remarks. Axianassa heardi n. sp. is presently the only species in the genus Axianassa with the uropodal exopod bearing a conspicuous transverse suture furnished with row of small subacute denticles ( Fig. 9 View FIGURE 9 G, H). The presence of a dentate transverse suture immediately separates the new species from all the other species of the genus Axianassa (cf. Kensley & Heard 1990; Rodrigues & Shimizu 1992; Anker 2010; Liu & Liu 2010). Axianassa heardi n. sp. is also the third member of Axianassa reported from the Indo-West Pacific, differing from the recently described A. ngochoae Anker, 2010 and A. sinica Liu & Liu 2010 in a number of other characters, such as the marginally dentate rostrum and a much shorter antennal acicle (cf. Anker 2010: fig. 1d; Liu & Liu 2010: fig. 2A). All American species of Axianassa differ from the new species by at least two morphological characters, in addition to the toothed diaeresis on the uropodal exopod (cf. Kensley & Heard 1990; Rodrigues & Shimizu 1992). Noteworthy, A. heardi n. sp. appears to be confined to sandy reef patches, whereas all the other species of Axianassa inhabit muddier bottoms (cf. Kensley & Heard 1990; Anker 2010; Liu & Liu 2010).
Several morphological features of A. heardi n. sp. disagree with the generic diagnosis of Axianassa provided by Kensley & Heard (1990). For example, the eyestalks were described as “short, cornea poorly defined, eyes poorly pigmented ...”. In A. heardi n. sp., the eyes have a relatively large, globular cornea occupying most of the eyestalk’s apical area; the cornea seems to be relatively well-pigmented, although in life its colour is yellowish white ( Fig. 12 View FIGURE 12 ), becoming blackish in alcohol ( Fig. 9 View FIGURE 9 D). More importantly, in all other species of Axianassa the uropodal exopod and endopod are “lacking sutures”, with the exopod bearing a “variable weak spination” ( Kensley & Heard 1990). As emphasised above, in A. heardi n. sp., the distal area of the exopod has a distinct suture furnished with small acute denticles. Such a suture is absent from the endopod, which only bears a few acute or subacute denticles dorsally and one marginal tooth laterally. In addition, the first maxilliped of A. heardi n. sp. bears a fairly large epipod ( Fig. 10 View FIGURE 10 E, F), which seems to be rudimentary in all other species of Axianassa (cf. Kensley & Heard 1990: fig. 2H; Anker 2010: fig. 2e). Other possible particularities in the mouthparts of A. heardi n. sp. include the absence of teeth on the molar process and a biarticulated endopod of the maxilla. With more Indo-West Pacific species discovered in the future, Axianassa may eventually require a redefinition, which, however, is beyond the scope of the present study.
| FLMNH |
Florida Museum of Natural History |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |