Anacardium occidentale, L.
|
publication ID |
https://doi.org/10.1016/j.phytochem.2020.112527 |
|
DOI |
https://doi.org/10.5281/zenodo.8302079 |
|
persistent identifier |
https://treatment.plazi.org/id/743187C0-FF84-5152-FF9C-1D6EFB40FAB8 |
|
treatment provided by |
Felipe |
|
scientific name |
Anacardium occidentale |
| status |
|
2.4. A molecular mechanism for the chitinolytic and antifungal properties of A. occidentale View in CoL View at ENA class VI chitinase
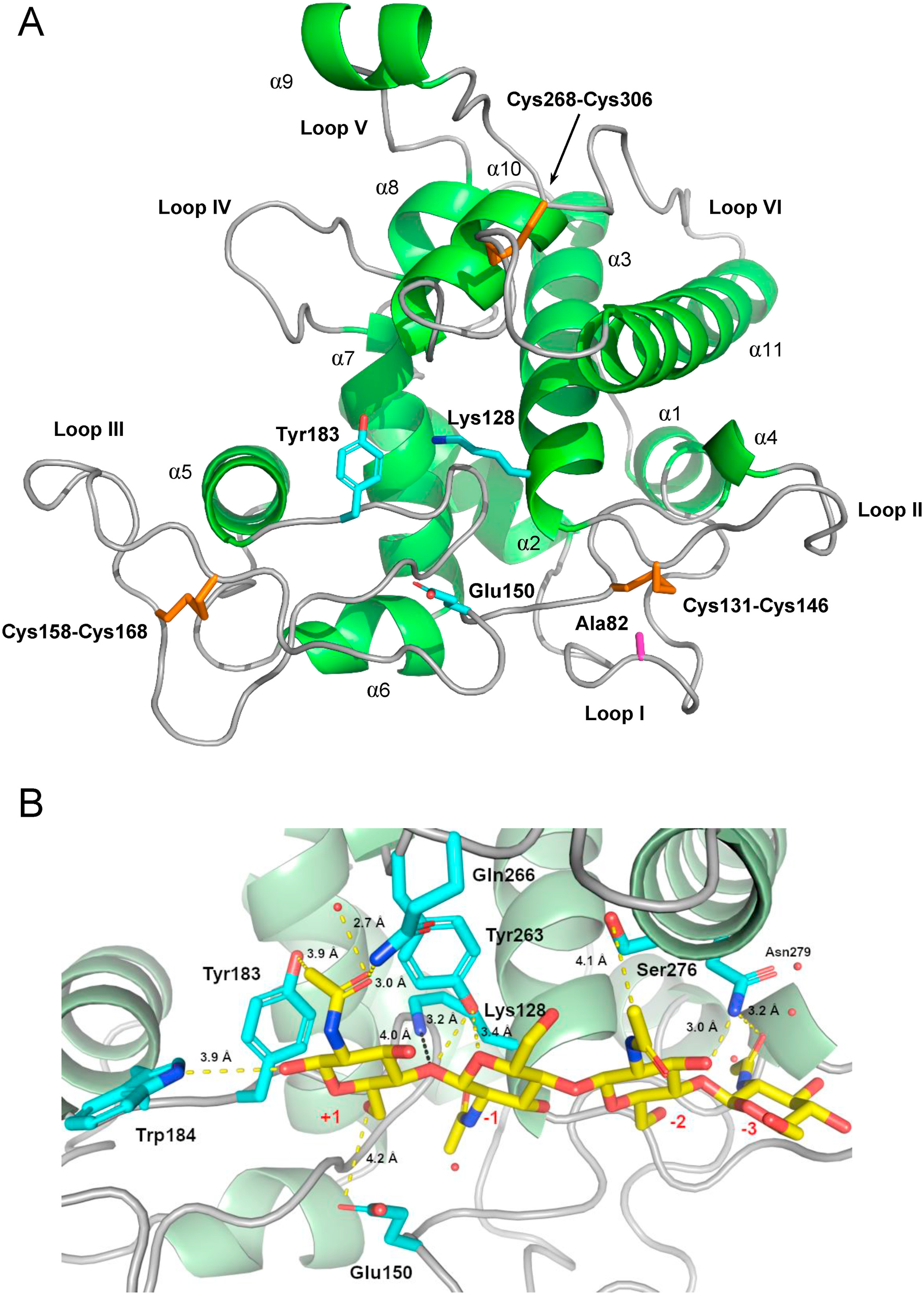
To investigate a probable mechanism for the chitinolytic and antifungal properties of cashew class VI chitinase, a three-dimensional molecular model of its GH19 domain was generated by homology modeling (Fig. S21), the model was validated (Fig. S22 and Table S3) and molecular docking calculations were performed, in which a (GlcNAc) 4 oligosaccharide was docked in the substrate-binding cleft of AoChi ( Fig. 7 View Fig ). The three-dimensional model of AoChi showed the typical features of GH19 structures, with 11 α- helixes, several loop regions connecting the helical segments and 3 disulfide bonds ( Fig. 7A View Fig ). The replacement of a conserved Cys residue by Ala (Ala 82) was compensated by the emergence of a new Cys residue (Cys 131), and this new Cys residue was predicted to establish a disulfide bond with Cys 146, thus maintaining the conserved number of 3 disulfide linkages, a typical structural feature of plant GH19 chitinases ( Fig. 7A View Fig ).
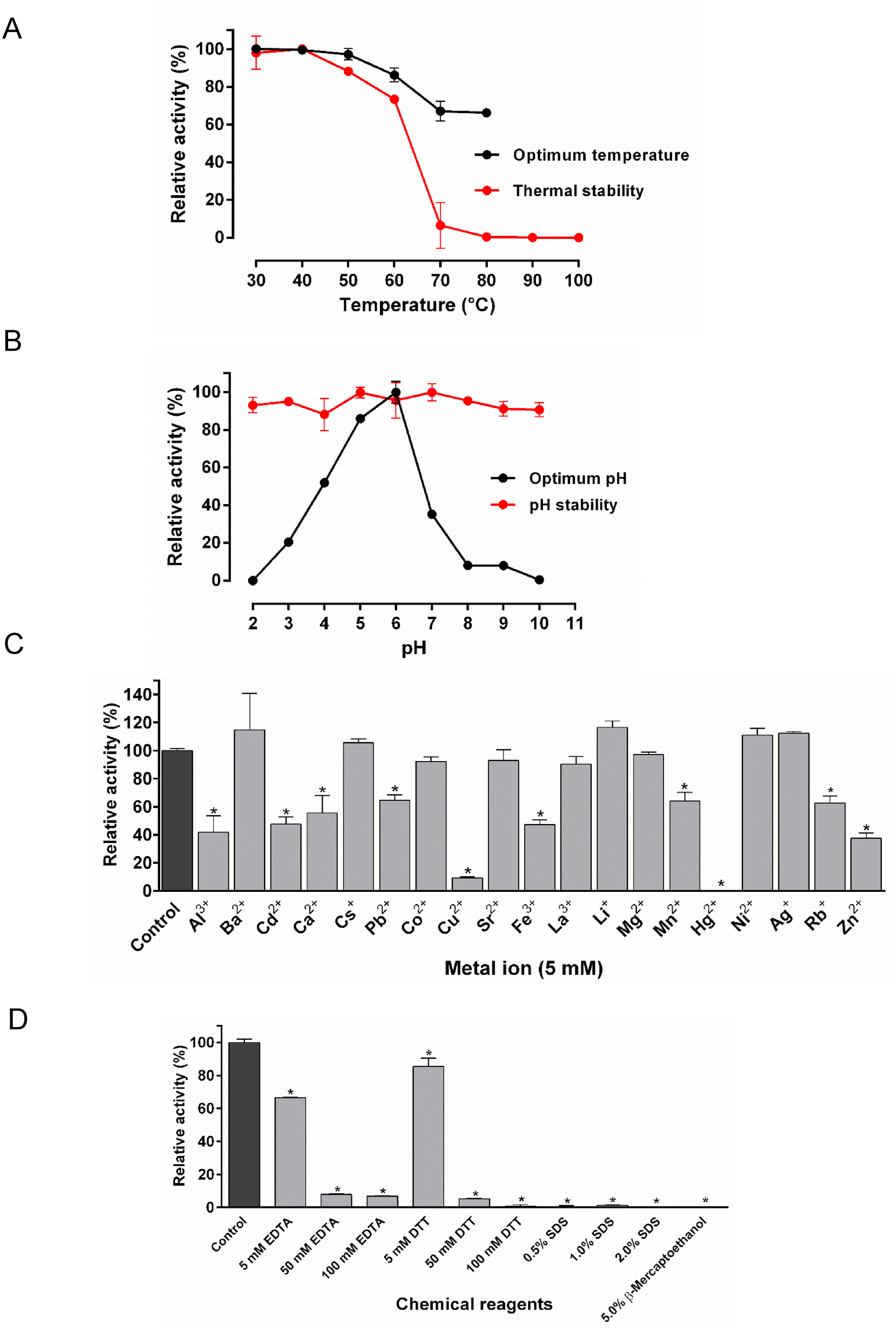
Once the overall characteristics of the AoChi three-dimensional model were analyzed as well as the quality of its local and global stereo-chemical parameters were assessed, a chito-oligosaccharide with 4 units of GlcNAc was docked in the substrate-binding cleft of the validated model. The complex protein-carbohydrate was predicted to be stabilized by a network of hydrogen bonds, involving O and N atoms of the side chains of certain AoChi residues and O atoms of hydroxyl and carbonyl groups of the sugar units as well as the O atoms of the glycosidic bonds, linking the GlcNAc units of the oligosaccharide ( Fig. 7B View Fig ). The side chain of Lys 128, which replaces the conserved Glu that acts as the proton donor in classical chitinases, was directed towards the ligand. The side chain of Glu 150, the putative general base, was on the opposite side of the substrate-binding cleft. The distances between the Nζ atom of Lys 128 and the atoms Oε1 and Oε2 of Glu 150 were 8.0 Å and 7.9 Å. The relative positions of the side chains of Lys 128 and Glu 150 as well as the average distance between their Nζ and Oε atoms agree to what is usually observed for the positions and average distances between the catalytic carboxyl groups in several structures of inverting GHs ( Mhlongo et al., 2014). Furthermore, the distance between the Nζ atom of Lys 128 and the most probable scissile O -glycosidic bond in the oligosaccharide docked in the substrate-binding groove of AoChi (approximately 4 Å), suggests that Lys 128 could act as the proton donor during catalysis. Supporting this assumption, the theoretical p K a value of the ζNH 3 + group of Lys 128, as predicted using the software PROPKA3, was 6.01, whereas the p K a for the δCOOH group of the general base Glu 150 was calculated as 4.48 (Table S4). These numbers corroborate the enzymatic activity profile of AoChi as a function of pH values ( Fig. 5B View Fig ). A rapid, exponential-like increase in AoChi activity was observed from pH 3.0 to pH 5.0. This could be due to the continuous increase in the deprotonated form of the carboxyl group of Glu 150, acting as a base, and the concomitant deprotonation of the ζNH 3 + group of Lys 128, acting as the proton donor. On the other hand, AoChi activity rapidly decreased beyond pH 6.0, reaching negligible values at pH 8.0. At pH 8.0, most (~99%) ζNH 3 + groups of Lys 128 residues would be in a deprotonated form, and thus could no longer act as proton donors to sustain catalysis.
The CatD of AoChi has, besides Lys 128, another 11 Lys residues, all of them with theoretical p K a values of their ζNH 3 + groups ranging from 10.18 (Lys 76) to 10.75 (Lys 111). The average experimental p K a values for Lys residues, measured in 157 proteins, have been determined to be 10.68 ( Pahari et al., 2019), which is close to the intrinsic p K a of the ζNH 3 + group of Lys in bulk water, which is 10.4 ( Nozaki and Tanford, 1967). However, Lys and other ionizable residues buried in the hydrophobic environments of certain proteins might have anomalous p K a values, with large deviations from the intrinsic p K a values in water. Indeed, buried Lys residues with experimental p K a values as low as 6.2 have been experimentally determined ( Kougentakis et al., 2018). Therefore, this analysis supports the assumption that Lys 128 of AoChi can act as the proton donor during catalysis, explaining the ability of A. occidentale class VI chitinase to degrade colloidal chitin and to cause damages in the cell walls of pathogenic fungi.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |