Amphiscolops blumi, Achatz, Johannes G., Hooge, Matthew D. & Tyler, Seth, 2007
|
publication ID |
https://doi.org/10.5281/zenodo.176820 |
|
DOI |
https://doi.org/10.5281/zenodo.5665126 |
|
persistent identifier |
https://treatment.plazi.org/id/945B87AC-FFB1-FFE3-77BF-B7506FD4B866 |
|
treatment provided by |
Plazi |
|
scientific name |
Amphiscolops blumi |
| status |
sp. nov. |
Amphiscolops blumi View in CoL sp. nov.
( Figs. 1–3 View FIGURE 1 View FIGURE 2 View FIGURE 3 )
Amphiscolops sp.: Hooge & Tyler 2005 (p. 102).
Diagnosis. Amphiscolops with seminal bursa filled with dense parenchymal tissue, 2–4 curved bursal nozzles, each measuring 80–120 µm in length. Male copulatory organ consists of a ciliated, glandular male antrum and a ciliated ejaculatory duct. A penis sac is absent, but numerous parenchymal muscles run through dense parenchymal tissue, which surrounds the antrum and the ejaculatory duct, and attach to the body wall. A caudal fold, or sperm guide (sensu Hyman 1937) covers the opening of the ejaculatory duct to the antrum. Setting the total body length to 100 units (100 U), the distance from the anterior tip of the body are as follows: st 11 U, m 48 U, fgp 73 U, mgp 77 U.
Type Material. Holotype: USNM 1096735, one set of 1.5-µm-thick serial sagittal sections of epoxyembedded specimen stained with toluidine blue. Paratype: USNM 1096736, one set of 1.5-µm-thick serial sagittal sections of epoxy-embedded specimen stained with toluidine blue.
Type Locality. In medium grained sand at the waterline on the northeast side of Carrie Bow Cay, Belize ( 16°48’9.4” N, 88°04’54.1” W).
Other Material Examined. Living specimens in squeeze preparations, two complete sets of serial sections of epoxy-embedded specimens stained with toluidine blue, one partial set of epoxy-embedded specimen stained with Heidenhain’s hematoxylin, one whole-mount for fluorescence microscopy.
Etymology. The species is named in honor and memory of Adolf Blum, Austria.
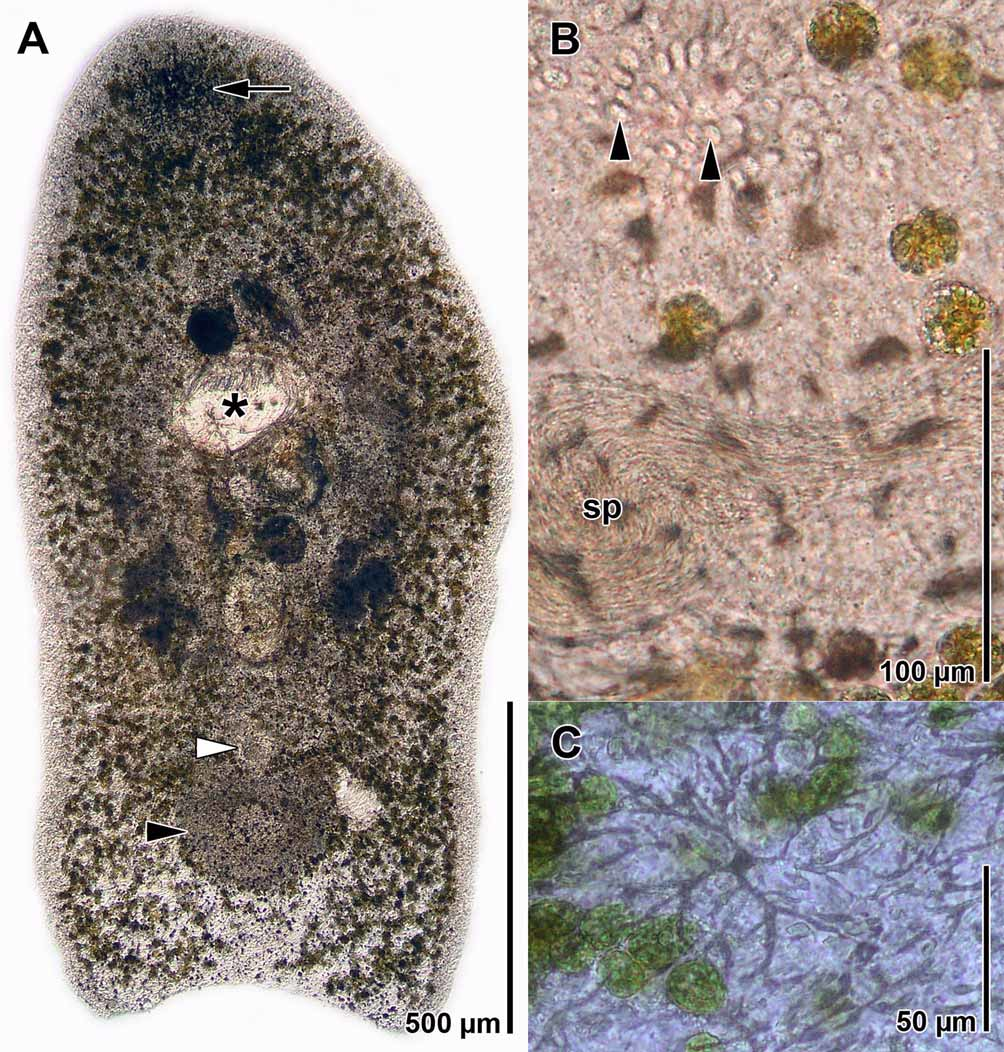
Description. Mature animals are ~ 2 mm long and ~ 0.5 mm wide ( Fig. 1 View FIGURE 1 A).
Cilia, ~7 µm long, occur on the entire body surface. The dorsal epidermis contains concrements that appear black in transmitted light, white in reflected light. The concrements are arranged in anastomosing starshaped patterns ( Fig. 1 View FIGURE 1 C) over the whole surface and form a spot in front the statocyst ( Fig. 1 View FIGURE 1 A). The epidermal nuclei are sunken beneath the body-wall musculature, which consists of outer circular muscles, crossover muscles, and longitudinal muscles.
Body color is green-brown due to the presence of numerous, 20–24 µm wide zooxanthellae, which are scattered throughout the parenchyma ( Fig. 1 View FIGURE 1 B). Rhabdoid gland cells and mucous gland cells occur on the entire surface, rhabdoid gland cells most numerous dorsally and in the anterior part of the body, mucous gland cells most numerous ventrally. A frontal organ is absent.
The nervous system consists of a brain positioned ventral to the statocyst, one ventro-lateral pair and two dorsal pairs of prominent longitudinal nerve cords. The statocyst lies ~200 µm behind the anterior tip. Two red ocelli lie lateral to it.
The mouth lies slightly behind the middle of the body. The digestive syncytium is often filled with crustaceans ( Fig. 2 View FIGURE 2 A).
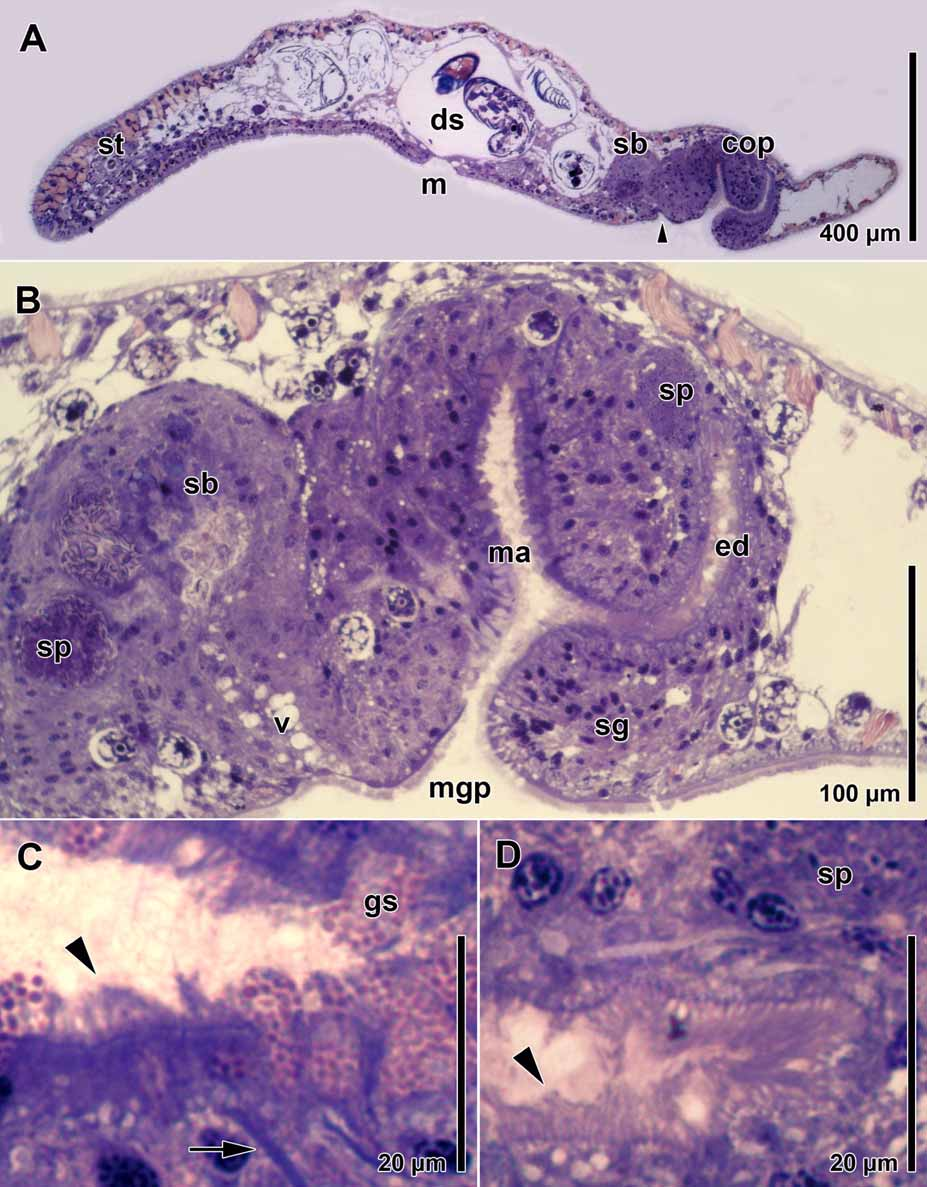
The paired testes lie lateral and dorsal to the paired ovaries. Testes and ovaries originate behind the statocyst. The germative zone of the testes reaches back to the edge of the mouth. Sperm migrate to the copulatory organ and accumulate lateral and posterior to it ( Fig. 1 View FIGURE 1 B). Female follicles develop into oocytes or abortive eggs. Oocytes are flat, lobulated, and incorporate abortive eggs and algae.
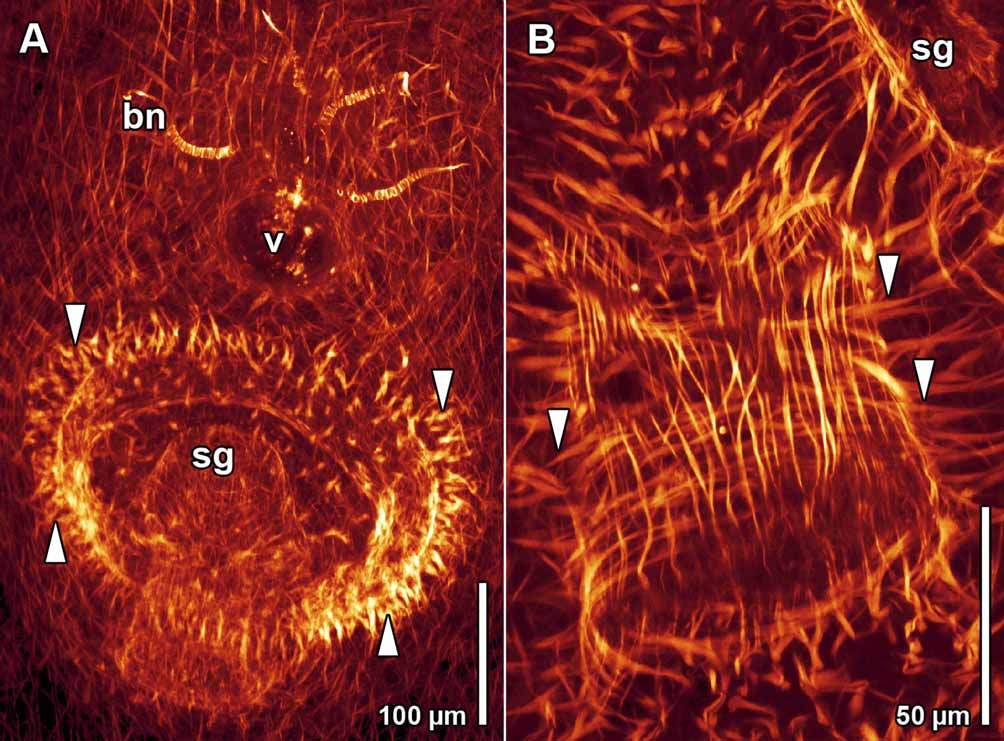
The female gonopore lies ~500 µm behind the mouth and ~100 µm in front of the male gonopore ( Fig. 2 View FIGURE 2 A). The short vagina opens into the seminal bursa caudally. Both are lined with parenchymal tissue. The seminal bursa is filled with dense parenchymal tissue and clusters of sperm ( Fig. 2 View FIGURE 2 B), which accumulate at the proximal end of the bursal nozzles. In mature animals, 2–4 curved bursal nozzles are present, each measuring 80–120 µm in length ( Fig. 3 View FIGURE 3 A).
The male gonopore is positioned 250–300 µm in front of the posterior end ( Fig. 2 View FIGURE 2 A). It opens into a glandular male antrum dorsally and an ejaculatory duct caudally ( Fig. 2 View FIGURE 2 B). The antrum is a ~220 µm-wide, transverse slit, lined with the distal necks of gland cells, which contain granular cyanophilic secretions ( Figs. 1 View FIGURE 1 B, 2B, C, 3A). The antrum and the ejaculatory duct are both ciliated ( Figs. 2 View FIGURE 2 C, D). A caudal fold, or sperm guide (sensu Hyman 1937) covers the opening of the ejaculatory duct to the antrum ( Figs. 2 View FIGURE 2 B, 3A). In some specimens sperm could be seen protruding into the proximal part of the ejaculatory duct. Beneath the ciliated epithelium of the ejaculatory duct lies a layer of circular muscles, which is constituted by parenchymal muscles and beneath this a layer of longitudinal muscles ( Fig. 3 View FIGURE 3 B). Numerous parenchymal muscles run through the dense parenchymal tissue, which surrounds the antrum and the ejaculatory duct, and attach to the body wall ( Fig. 3 View FIGURE 3 A).
Remarks. Of the 12 known species of Amphiscolops (see Tyler et al. 2006), Amphiscolops blumi is most similar to Amphiscolops langerhansi ( Graff, 1882) and Amphiscolops carvalhoi Marcus, 1952 . All three species have a spherical seminal bursa filled with parenchymal cells, multiple bursal nozzles, which are long and curved, and a male copulatory organ with a large glandular antrum and an ejaculatory duct. We found tissue identical with the frontal gland cells (Stirndrüsen) shown by Graff (1904) in figures 1, 2 and 3 of plate 12. Contrary to his interpretation we found this tissue to be nervous tissue. Consequently the three species are additionally united by the absence of a frontal organ.
Like A. carvalhoi , A. blumi is about half the size of A. langerhansi ( 2 mm in length vs. 4–5 mm in length of A. langerhansi ) and has similar number of bursal nozzles (2–4 compared to 2–6 in A. carvalhoi ), fewer than in A. langerhansi (which has 6–11).
Patterns of coloration are somewhat different in these three species as well. The epidermal concrements are white in both, A. blumi and A. langerhansi , golden in A. carvalhoi . The pattern of their distribution in A. blumi is similar to that of A. langerhansi (compare figure 1A with Graff’s (1904) figure 6, plate 11 of A. langerhansi ), including a dense aggregation of concrements in front of the statocyst and, in some specimens, aggregations into longitudinal stripes as has been described also for some specimens of A. langerhansi ( Graff 1904) . A. carvalhoi lacks these aggregations, and it also has two post-cerebral spots bare of concrements. Graff (1904) did not discern in A. langerhansi the star-like shape of concrements Marcus (1952) and we noted in A. blumi and A. carvalhoi , respectively, perhaps because he used lower magnifications in his investigation.
A. blumi and A. langerhansi are oviparous, A. carvalhoi is viviparous.
In having a ciliated epithelium lining both the male antrum and the ejaculatory duct, A. blumi is similar to A. langerhansi as Graff (1904) described it. Hyman’s (1937) account of copulation in specimens of A. langerhansi from Bermuda, however, describes her specimens as lacking ciliation in both the antrum and the duct; confounding identification of these specimens, however, is that they had fewer bursal nozzles (2–8) than originally described for A. langerhansi . Marcus (1952) reported cilia in only the male antrum of A. carvalhoi . Conceivably, however, the numerous gland cells of the male antrum in A. carvalhoi could have obscured ciliation here.
From several masses of floating Sargassum sp. gathered near Carrie Bow Cay, we collected acoels similar in appearance to A. blumi . Although some of these specimens had eggs, none had male copulatory organs, and as such, we were unable to determine if they were conspecific to A. blumi .
| USNM |
Smithsonian Institution, National Museum of Natural History |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |