Thenea muricata ( Bowerbank, 1858 )
|
publication ID |
https://doi.org/ 10.5281/zenodo.280590 |
|
DOI |
https://doi.org/10.5281/zenodo.6166257 |
|
persistent identifier |
https://treatment.plazi.org/id/A61787B7-DC4F-FF9D-FF33-FE5DFB98FE79 |
|
treatment provided by |
Plazi |
|
scientific name |
Thenea muricata ( Bowerbank, 1858 ) |
| status |
|
Thenea muricata ( Bowerbank, 1858)
( Figures 18 View FIGURE 18 E–F, 21–23, Table 5 View TABLE 5 )
Synonymy (modified from Maldonado (2002)).
Tethea muricata Bowerbank, 1858 : Bowerbank 1858, p. 308, pl. 25. fig. 18; Bowerbank 1872, p. 115, pl. V, figs. 1-6.
Thenea muricata: Gray 1867 , p. 541; Sollas 1888, p. 95; Topsent 1892, p. 37; Topsent 1894, p. 375; Lambe 1896, p. 202; Lambe 1900, p. 26; Topsent 1913a; Stephens 1915, p. 11; Ferrer-Hernández 1922, p. 2; Sarà 1958, p. 216; Gamulin-Brida 1969, p. 89; Pulitzer-Finali 1972, p. 348; Uriz 1981, p. 48; Steenstrup & Tendal 1982, p. 259; Pulitzer-Finali 1983, p. 474; Pansini 1987b, p. 43; Uriz & Rosell 1990, p. 48; Pansini & Musso 1991, Table 2; Boury-Esnault et al. 1994, p. 51; Maldonado 2002, p. 156; Voultsiadou 2005, Table 1 View TABLE 1 ; van Soest et al. 2007, Table 2; Cárdenas et al. 2011, Table S1.
Ancorina (Thenea) muricata : von Lendenfeld 1903, p. 53.
? Ancorina (Thenea) muricata: Babiç 1914 , p. 152.
Wyvillethomsonia wallichii Wright, 1870 : Wright 1870, p. 8.
Thenea wallichii: Sollas 1882 , p. 427.
Dorvillia agariciformis Kent, 1870 : Kent 1870, p. 293.
Tisiphonia agariciformis: Thomson 1873 , p. 167.
Clavellomorpha minima Hansen, 1885 : Hansen 1885, p. 19.
? Thenea ( muricata ) schmidti Sollas, 1886 : Babiç 1915, p. 389; Babiç 1922, p. 282.
Thenea intermedia Sollas, 1888 : Sollas 1888, p. 97.
Not Thenea schmidti Sollas, 1886 : Sollas 1886, p. 183; Sollas 1888, p. 67 (this study)
Material. ZMBN 85232, Brattholmen, Hjeltefjord, western Norway, 100 m; ZMBN 85252(a/b), Bømlafjorden, Midtvikøy, western Norway, 59°40'N, 5°24'E, 230- 90 m, triangular dredge; ZMBN 85231, west of Marstein, western Norway, 60°8'18''N, 4°50'47''E, 300 m, Agassiz trawl; ZMBN 87920, Sognefjorden deep basin, off Lånefjorden, Western Norway, 61°08.3'N, 6°10'E, 1232- 1225 m, Agassiz trawl; ZMBN 85253, mid Norway, 63°08.54'N, 8°13.37'E, 165 m, Møre 2006 cruise; NTNU-VM 54908, Trollsteinen, mid Norway, 64°35'59.9"N, 11°1'30"E, 420 m; NTNU-VM 54909, Kinebbneset, Trondheimsfjorden western Norway, 63°32'28"N, 9°49'47.2"E; NTNU-VM 55655, Tautra, Trondheimsfjorden, mid Norway, 63°32'17.99"N, 10°30'33.99"E, 160 m; NTNU-VM 55622, north of Smøla Island, mid Norway, 63°37'50"N, 7°59'14.9"E, 300 m; NTNU-VM 55843, Haltenbanken, mid Norway, 64°14'56''N, 9°0'3.7''E.
Comparative material.
Thenea muricata , ZMAPOR 19481, Rockall Bank, 55°31'7.9"N, 15°48'27.54"W, 844-857 m; MNHN- DCL3436, Banuyls, France, 800 m, Ecomarge 1985; MNHN-DCL4083, off Cape Santa Maria di Leuca, Italy, 39°34.01'N, 18°26.16'E, 585 m, MEDECO 2007 cruise; ZMBN 85254, Gulf of Cadiz, Balgim campaign, St. CP25, 36°41'5''N, 7°19'4''W, 544 m.
Thenea schmidti , ZMAPOR 18036, Gulf of Cadiz, 35°47'59.9"N, 7°47'59.9"W, 443 m.
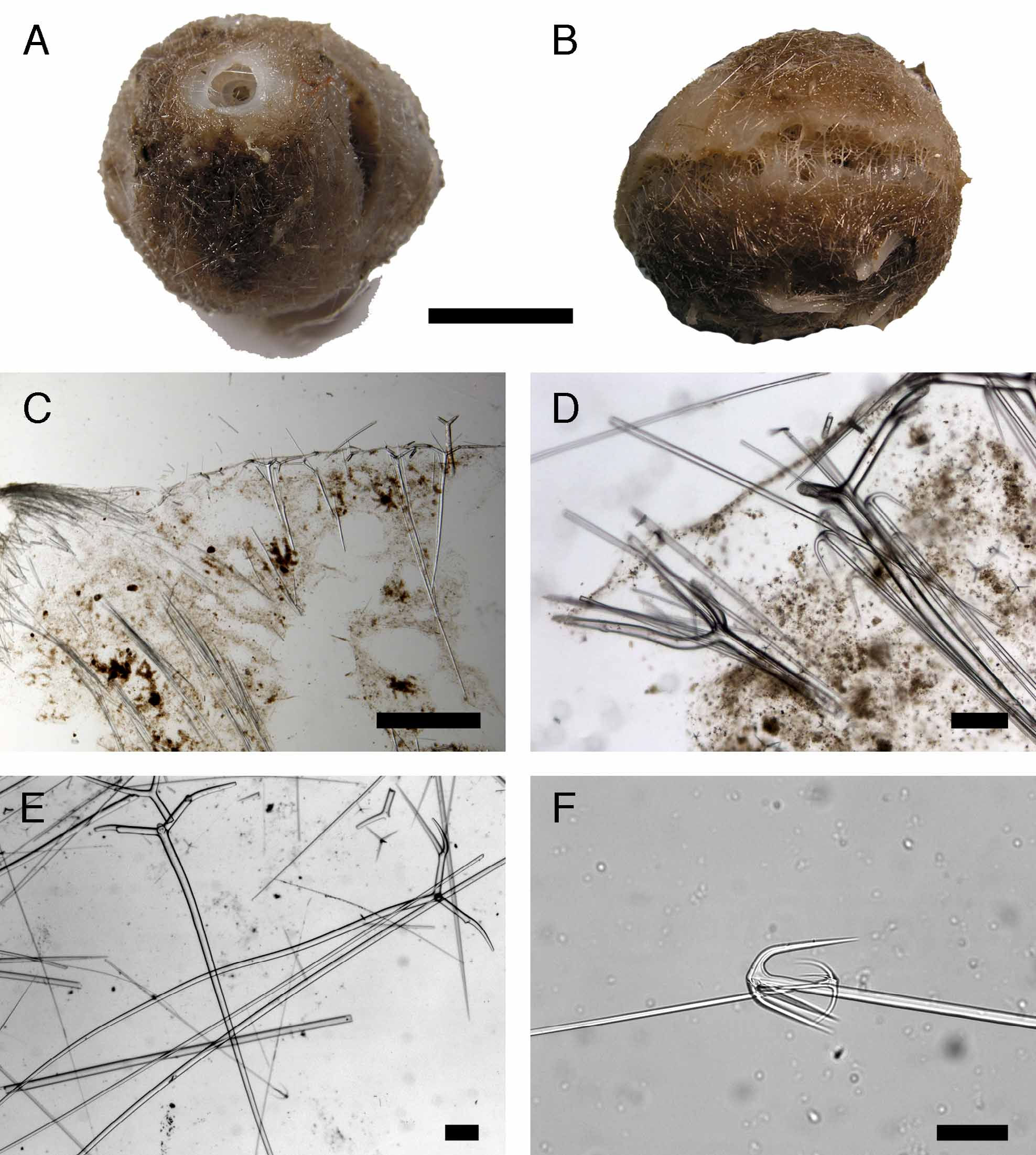
Outer morphology ( Fig. 21 View FIGURE 21 A-B). Massive sub-spherical sponge. ZMBN 85231 is 2.8 cm long and 2.6 cm high. ZMBN 85252(a/b) are made of “fused specimens” (4-8 cm of diameter). These larger specimens have many stringlike buds on its top surface, these can reach 2 cm long. ZMBN 85232 is a half damaged specimen. All specimens are slightly compressible. Surface is minutely hispid, like sand-paper. Color alive and in ethanol is brown. Choanosome is whitish to brownish (alive and in ethanol). Single, circular to ellipsoid oscule situated in the top (3 mm in ZMBN 85231, 1- 4 mm in ZMBN 85252); a conspicuous whitish sphincter surrounds it and there is no sieve. The sieved pore area runs along the equatorial side of the specimen ( ZMBN 85231) or can extend itself in the entire lower part of the specimen ( ZMBM 85252). All specimens have roots.
Skeleton ( Fig. 21 View FIGURE 21 C-D). Like T. abyssorum . Depending on where the section is made, plesiasters are rare (especially in the choanosome) to very abundant (especially under the ectosome). Protriaenes are found under the ectosome as well ( Fig. 21 View FIGURE 21 D), they can be rare to common depending on the specimen. Anatriaenes were observed in the choanosome associated in bundles with triaenes ( Fig. 21 View FIGURE 21 D); they were also observed coming out of the top surface.
Spicules ( ZMBN 85231) ( Figs. 18 View FIGURE 18 E-F, 21D-F). (a) oxeas, slightly bent, length: 4000- 5196.4 -5925 µm (N=7); width: 30- 41.4 -55 µm (N=7). (b) dichotriaenes ( Fig. 21 View FIGURE 21 D-E), rhabdome is straight to bent, or even flexuous, length: 1925- 2972 -3875 µm (N=9); width: 23- 49.6 -80 µm; protoclad length: 190- 284.5 -370 µm; deuteroclad length: 350- 564 -900 µm. (c) anatriaenes ( Fig. 21 View FIGURE 21 F), common, straight, bent or flexuous rhabdome, length: 1550- 3976.1 -4675 µm (N=23); rhabdome width: 5- 11.8 -22 µm; two shapes, the smaller ones, probably younger forms (with clad length: 60-85 µm and rhabdome width: 5-9 µm) have a regular umbrella-shape, the larger more common ones have bent clads, clad length: 60- 157.1 -242 µm. (d) protriaenes, rare, straight, rhabdome length>1000 µm; rhabdome width: 50 µm (N=1); clad length:>600 µm (N=1). (e) spirasters (and few metasters) ( Fig. 18 View FIGURE 18 F), spined, length: 13- 23.2 -32.5 µm. (f) plesiasters ( Fig. 18 View FIGURE 18 E), 3-7 actines, faintly spined (hardly visible with an optical microscope), actine length: 16.6- 37.0 -70.3 actine width: 1.7- 3.5 -10
Reproduction. We observed oocytes in vitellogenetic phase (A. Ereskovsky, pers. comm.) in the thick sections of T. schmidti (ZMAPOR 18036, collected at the end of August 2004) and T. muricata ( ZMBN 85231, collected mid-June 2008). Spermatogenesis has been observed in T. muricata from Norway, possibly collected in May ( Sollas 1882). Oocytes and spermatocytes have already been observed simultaneously in T. muricata from the Adriatic Sea indicating that this species can be hermaphrodite ( Babiç 1915). Elongated thread-like buds are common on the upper surface of large Norwegian specimens (e.g. ZMBN 85252) whereas they seem to be more subglobular in Mediterranean samples ( Uriz 1981, Fig. 28).
Distribution ( Fig. 22 View FIGURE 22 ). Greenland, Denmark Strait and southern Iceland ( Steenstrup & Tendal 1982); Norway ( Bowerbank 1872; Sollas 1882; Topsent 1913a; Steenstrup & Tendal 1982); Rockall Bank (van Soest et al. 2007); northern Spain ( Ferrer-Hernández 1922); Azores ( Topsent 1892; 1904); Gulf of Cadiz ( Boury-Esnault et al. 1994); Mediterranean Sea ( Sollas 1888; Sarà 1958; Gamulin-Brida 1969; Pulitzer-Finali 1972; Uriz 1981; Pulitzer-Finali 1983; Uriz & Rosell 1990; Pansini & Musso 1991; Boury-Esnault et al. 1994; Voultsiadou, 2005);? Canada ( Topsent 1892; Lambe 1896).
As T. schmidti : Puerto Rico ( Sollas 1888); Azores ( Topsent 1892; 1904); Gulf of Cadiz ( Sollas 1888; this study); Cape Verde (van Soest 1993); Mediterranean Sea ( Babiç 1915; 1922; Pansini 1987b).
Depth. For T. muricata : 60-2940 m ( Steenstrup & Tendal 1982; Voultsiadou 2005); for T. schmidti in the Azores: 349–4020 m ( Topsent 1904).
Discussion. The Norwegian specimens fully agree with the redescription of this species ( Steenstrup & Tendal 1982). It is the most widespread Thenea species ( Fig. 22 View FIGURE 22 ): its northernmost localities are at the moment in Eastern Greenland and the Denmark Strait while its southernmost localities are off Morocco. We have never identified T. muricata in northern Norway or the Barents Sea. We re-examined the T. muricata specimens (ZMAPOR 2385) collected by Vosmaer (1885) in the Barents Sea, and re-identified them as T. valdiviae based on the presence of sieves covering their oscules (vs. “naked” oscules with a conspicuous whitish sphincter in T. muricata ). We therefore agree with Steenstrup and Tendal (1982) that other arctic records of T. muricata are likely to be T. valdiviae (e.g. Hansen 1885; Koltun 1966). T. muricata from the Zanzibar area is suspicious and probably a misidentification ( Burton 1959a).
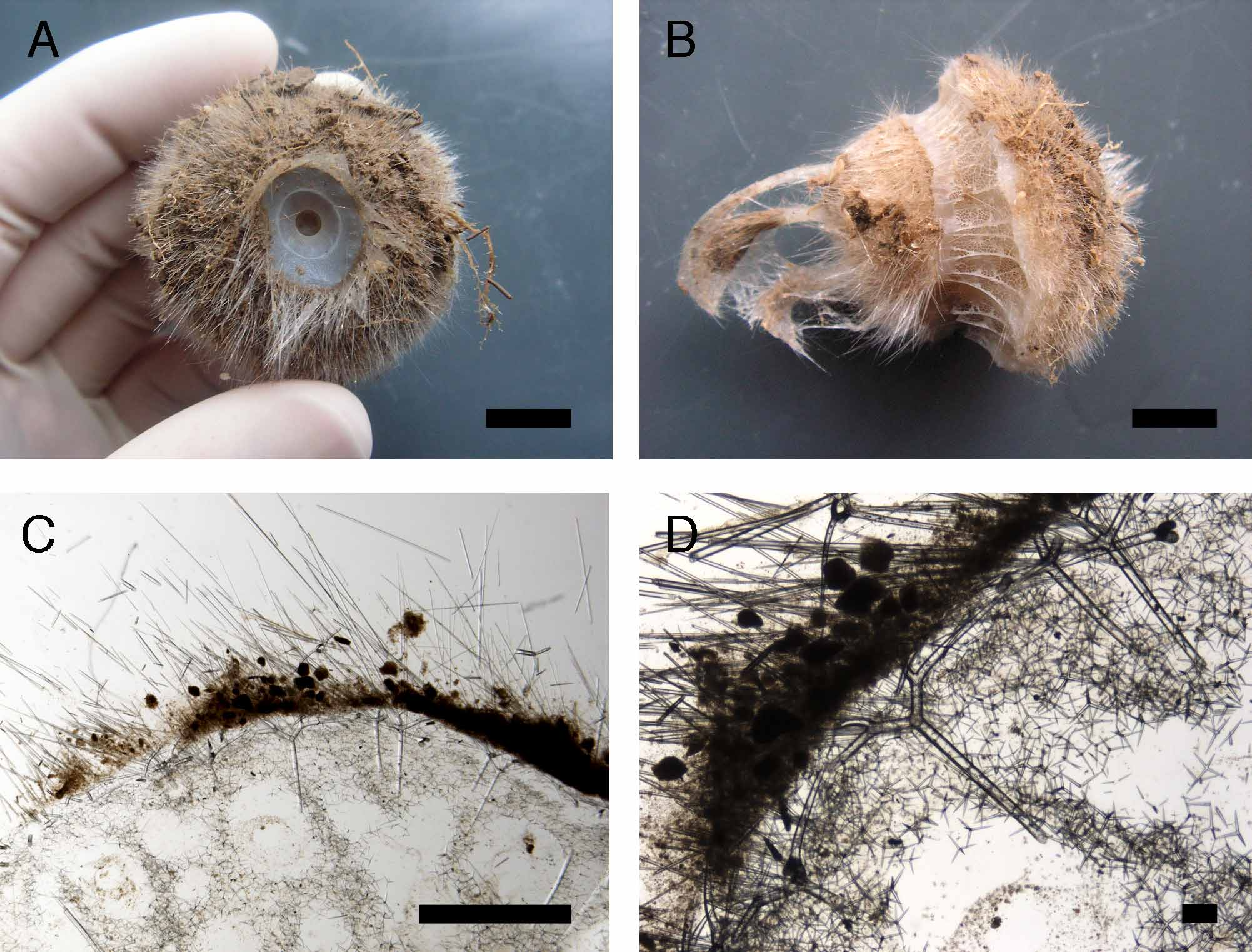
T. schmidti is distributed from the Azores to Gibraltar ( Fig. 22 View FIGURE 22 ) and originally characterized by i) a highdensity of larger plesiasters, ii) a poorly developed choanosome tissue, iii) larger choanocyte chambers and iv) a large oscule sphincter ( Sollas, 1888). Our specimen of T. schmidti ( Fig. 23 View FIGURE 23 ) corresponds in every point to this description. Stephens (1915) compared her Irish T. muricata with T. schmidti and often could not tell which one had the most plesiasters. Indeed, Norwegian T. muricata could show abundant plesiasters but these were localized in certain areas (e.g. under the ectosome), whereas T. schmidti appeared uniformly filled with plesiasters ( Fig. 23 View FIGURE 23 C–D).
The choanocyte chambers seemed a bit bigger (50-60 µm) for T. schmidti , but not as much as the ones measured by Sollas (1888): up to 90 µm. Mediterranean specimens of Thenea have raised doubts concerning the validity of T. schmidti . Sollas (1888) described T. intermedia with few plesiasters associated with poorly developed choanosome tissue and large choanocyte chambers (with measurements intermediate between T. schmidti and T. muricata from Norway). On the contrary, Topsent (1904) described a Mediterranean specimen with numerous plesiasters (as large as in T. schmidti ) associated with dense choanosomal tissue. Therefore, these characters have been considered to be variable and T. schmidti was synonymized with T. muricata ( Uriz 1981; Steenstrup & Tendal 1982). The Mediterranean specimen we examined is small (6 mm high, without the roots). It has numerous plesiasters, but not as large as in T. schmidti ( Table 5 View TABLE 5 ). Genetic data ( Fig. 25 View FIGURE 25 ) show that our Mediterranean specimen has the same COI and 28S sequence as T. muricata (ZMBN 85231) so Mediterranean specimens are likely to be T. muricata with numerous plesiasters. Meanwhile, even though T. schmidti (ZMAPOR 18036) has the exact same COI Folmer fragment as T. muricata , its faster evolving 28S (C1–D2) sequence is significantly different ( Fig. 25 View FIGURE 25 ). This strongly suggests that T. schmidti is a valid NEA species and that we are not focusing on the right characters to differentiate it. The amount of plesiasters and the size of the choanocyte chambers do not seem to be reliable characters; the size of plesiasters does not seem to be a good character as they can be fairly large in T. muricata as well ( Table 5 View TABLE 5 ). The very large plesiasters (actins of 420–540 µm length) found by Babiç (1915) in Thenea from the Adriatic Sea ( Table 5 View TABLE 5 ) are puzzling at the moment. The white broad sphincter around the oscule of T. schmidti might be a more reliable character, but we need more specimens to confirm this.
An even more southern species is Thenea bojeadori von Lendenfeld, 1907 (and its possible synonyms T. microclada von Lendenfeld, 1907 and T. megastrella von Lendenfeld, 1907) which can be found all along the northern coast of Africa ( Lévi 1959; Cruz 2002) ( Fig. 22 View FIGURE 22 ). The status of T. bojeadori would also need to be reevaluated in order to clearly separate it from T. schmidti and T. muricata ( Table 5 View TABLE 5 ).
Material Depth Plesiaster Spiraster/metaster
(m) (actin length) (length) ..... continued on the next page Material Depth Plesiaster Spiraster/metaster (m) (actin length) (length) MNHN-DCL3436 * 800 31– 126.9 –247 18– 25.2 –39 Banyuls France (abundant) (10) Adriatic Sea - 420–540 21 ( Babiç 1915) (abundant)
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Thenea muricata ( Bowerbank, 1858 )
| Cárdenas, Paco & Rapp, Hans Tore 2012 |
Ancorina (Thenea) muricata: Babiç 1914
| Babic 1914 |
Thenea intermedia
| Sollas 1888 |
schmidti
| Sollas 1886 |
Thenea schmidti
| Sollas 1886 |
Thenea schmidti
| Sollas 1886 |
Clavellomorpha minima
| Hansen 1885 |
Thenea wallichii:
| Sollas 1882 |
Tisiphonia agariciformis:
| Thomson 1873 |
Wyvillethomsonia wallichii
| Wright 1870 |
Dorvillia agariciformis
| Kent 1870 |
Thenea muricata:
| Gray 1867 |
Tethea muricata
| Bowerbank 1858 |