Petropedetes johnstoni
|
publication ID |
https://doi.org/10.5281/zenodo.193288 |
|
DOI |
https://doi.org/10.5281/zenodo.6199586 |
|
persistent identifier |
https://treatment.plazi.org/id/B479090F-FFCC-FF9A-D7B4-FBB94342083B |
|
treatment provided by |
Plazi |
|
scientific name |
Petropedetes johnstoni |
| status |
|
Petropedetes johnstoni View in CoL ( Boulenger, 1888 “1887”)
Figs. 1 View FIGURE 1 a, 2a, 3a, 4, 5, 7g
Cornufer johnstoni Boulenger, 1888 “1887”, Proc. Zool. Soc. London 1887: 564
Tympanoceros newtonii View in CoL du Bocage, 1895, J. Sci. Math. Phys. Nat. Lisboa, Ser.2, 3: 270 Petropedetes johnstoni View in CoL – Boulenger, 1900. Proc. Zool. Soc. London, 1900: 439
Material examined. BM 1947.2.7.44 ( holotype, described as Cornufer johnstoni ), subadult male, Cameroon (“ Cameroons ”), Rio del Rey, coll. H.H. Johnston; MSNG 29890 ( 5 males, 2 females), Equatorial Guinea, Bioko, Musola, app. 500-800 m a.s.l., I-III.1902, coll. L. Fea; MSNG 50032 (female and tadpole), app. 500- 700 m a.s.l., other data as MSNG 29890; ZFMK 87709 (male), Cameroon, Campo region, Nkoelon, app. 125 m a.s.l., 30.X.2007, coll. J.A.M. Wurstner & M.F. Barej; ZFMK 87710 (female), 26.X.2007, other data as for ZFMK 87709.
Diagnosis. A medium sized Petropedetes ; slender body shape; tympanum round, distinctly smaller than eye diameter (smallest distinct tympanum in the genus); characters of breeding males: tympanic papilla present (papilla slim, compared to other larger Petropedetes ), papilla close to the upper border of tympanum; forearm hypertrophy weakly developed; carpal spike present; spinosities on upper arm, throat and breast; few minuscule spines on supratympanal gland; femoral glands brightly orange, prominent and very large; webbing rudimentary.
Description. Medium sized Petropedetes with slender body shape; both sexes almost equally large (SUL in males: 28.5–45.6 mm, females: 28.1–47.8 mm); mean head width in males about 41% of SUL, in females about 38%; snout rounded to slightly pointed in lateral view; canthus rostralis sharp; loreal region concave; eye diameter about 1.5 times eye-narial distance; nose closer to snout-tip than to eye; distinct round tympanum smaller than eye in both sexes (tympanum / eye in males: 0.49–0.63; in females: 0.45–0.48; Fig. View FIGURE 1
1a); tympanic papilla of breeding males close to upper border of tympanum, comparatively slim; supratympanic fold distinct; fingers slender with T-shaped fingertips; relative length of fingers: III> IV> II> I; manual subarticular tubercles single; palmar tubercle and thenar tubercle present; manual webbing absent; forearm hypertrophy weakly developed; carpal spikes present in males; spinosities on upper arm, throat and breast; few minuscule spines on supratympanal gland; back skin granular, beset with small warts; fewer larger warts arranged in more or less longitudinal rows; flanks with few large warts; ventral skin smooth; mean femur length in males 53% of SUL, in females about 51%, mean tibia length in both sexes 61% of SUL; mean foot length in males 79% of SUL, in females 78%; pedal subarticular tubercles single; hind limbs slender; femoral glands very large in males, larger and more prominent in males than in females (femoral gland / femur length in males: 0.28–0.46, in females: 0.25–0.27; Fig. 2 View FIGURE 2 a); relative length of toes: IV> III> V> II> I; webbing rudimentary: 1 (1) 2 (1-1) 3 (2-2) 4 (3-3) 5 (2).
According to Amiet (1973) the tympanic papilla is shorter than the tympanum diameter and thickened at its tip. This character is hard to verify in preserved specimens, as the papilla is then often shrunken (M.F. Barej pers. obs.). Amiet (1983) remarked that skin on throat and basis of the arms is smooth, showing no trace of spinosities. This observation is not consistent with the MSNG material. The most detailed description referring to “ P. johnstoni ” has been published by Parker (1936). However, this was based on specimens which have later been recognized as a new species, P. parkeri ( Amiet 1983) .
Coloration. Dorsum brownish with small dark spots, marking larger warts; colour on flanks as on back, often with larger spots than on back ( Fig. 1 View FIGURE 1 a); iris speckled with gold and silver; throat marbled blackish; belly whitish with minuscule dark speckles (recognizable only under the microscope); upper and lower legs dorsally with large almost bar like dark spots; femoral glands brightly orange in males. Coloration in preservation: as in life, but colours pale. Boulenger (1888) mentioned a dark inter-orbital bar. In the material examined this bar was often incomplete.
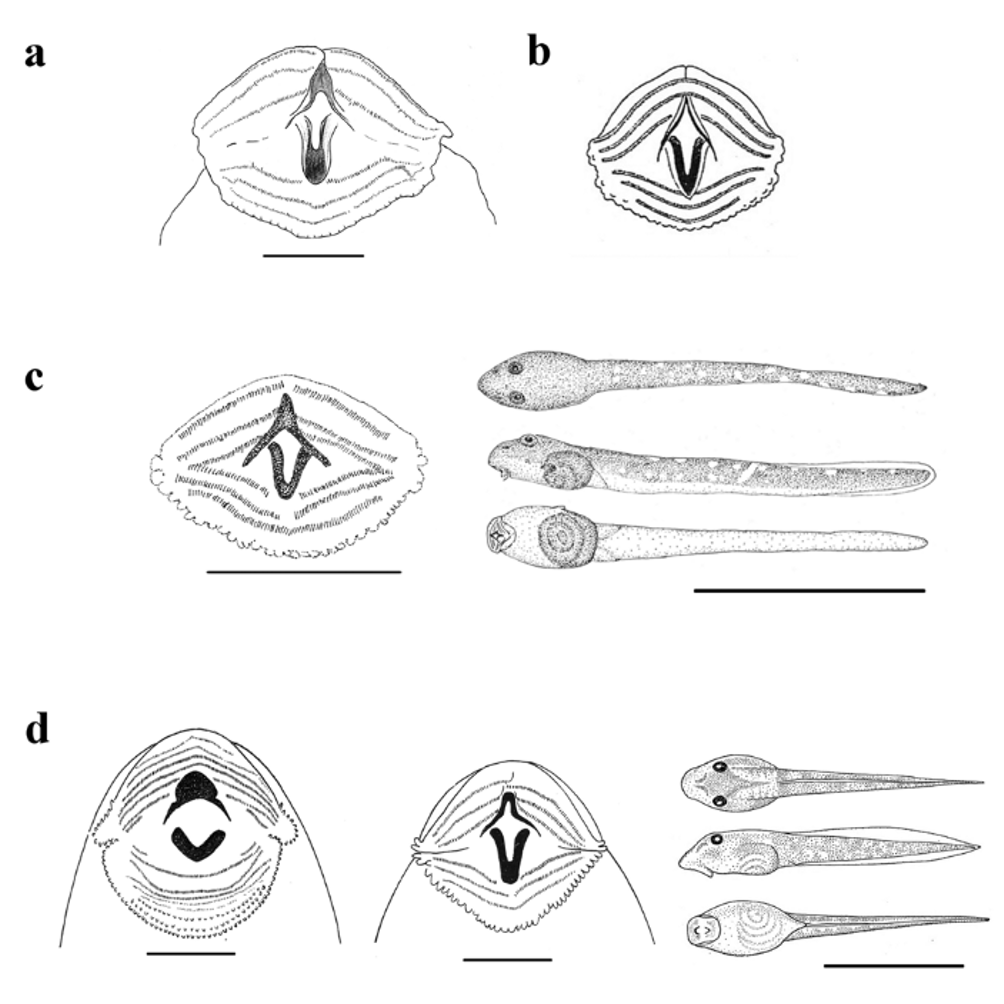
Tadpole description. The MSNG collection comprises one tadpole ( MSNG 50032) collected by L. Fea in March 1902 at Musola ( 500–700 m) on Bioko. The tadpole (Gosner stage 41) has already fully developed hind limbs with rudimentary webbing. The vent tube is regressed. It had a body length of 11.3 mm (total length 26.3 mm) and long hind legs ( 18.6 mm), its femoral glands were already 1.8 mm long. As from Bioko, no other Petropedetes species with rudimentary webbing is known ( P. cameronensis possesses half-webbed feet) we believe that it is safe to assume that the tadpole is a P. johnstoni . Its body is broad and slightly flattened; flanks not parallel but slightly convex; nostrils closer to eye than to snout tip; tail axis long and very muscular; no tail fins visible; end of tail shrunken, in life probably pointed; keratodont formula: 3+3//1+1:2; the upper three rows interrupted by the upper jaw sheath ( Fig. 3 View FIGURE 3 a). The body shape and tooth formula corresponds to the most advanced P. palmipes tadpoles ( Lamotte et al. 1959) and data on P. cameronensis ( Schiøtz 1966) .
Natural history. P. johnstoni inhabits lowland forest and in contrast to other species occurs in areas without flowing water ( Amiet 1983; own data). According to Gartshore (1986) this species inhabits littoral forests. In contrast, Fea (in du Bocage 1903) finds the species at a river with rapids, overgrown by vegetation. The call is very characteristic and can be heard from far (du Bocage 1903). Amiet (1989, 1991) finds an egg clutch on a large leaf in the forest, some centimetres above the ground. A male was sitting close to the clutch during daytime. Because of the absence of an open water source he assumed that the tadpoles of P. johnstoni are terrestrial ( Amiet 1989, 1991). Lawson (1993) reports Petropedetes clutches in the centre of leaf whorls, deposited at least 75 m away from the next water source. Tadpoles started to hatch after about two weeks. Lawson (1993) assigns these clutches to what he termed “ P. newtonii ” (described as a new species below). However, according to Amiet’s (1989, 1991) and our own observations this behaviour much better fits to the sympatric P. johnstoni .
Distribution. An exact type locality of P. johnstoni has not been given by Boulenger (1888). However, due to the title of the publication it has always been regarded as “Rio del Rey, Cameroon ”. Amiet (1983) points out that this region corresponds to a creek of the river Ndian in western Cameroon, close to the Nigerian border. P. johnstoni is a typical lowland species and is known from Victoria (= Limbe) and Bibundi ( Werner 1898; Andersson 1907; Amiet 1983) in western Cameroon and further localities in the lowland of the Biafra Bay down to southern Cameroon ( Amiet 1983). P. johnstoni has also been reported from Bioko (du Bocage 1895a, 1895b, 1903; Boulenger 1906; Mertens 1965; Weinberg 2008; Fig. 4 View FIGURE 4 ). J.P. Vande weghe photographed a Petropedetes with a tympanum smaller than the eye and a tympanic papilla in Langoué Bai, eastern Gabon ( Fig. 7 View FIGURE 7 g). This would be the first record of the species in Gabon. As no voucher is available this record still needs to be confirmed and the taxonomic status of the population requires further study.
Genetics. A genetic comparison of a fragment of the mitochondrial 16S rRNA gene (611 bp, including gaps) using a calculation of the uncorrected p-distances gave the following results for Petropedetes johnstoni : interspecific comparison between P. johnstoni and all other Central African Petropedetes taxa ranged between 3.85%-9.03%, while the intraspecific variation within P. johnstoni was much lower at 0.17% (N= 2) (Table 4).
Systematic remarks. Boulenger (1888) states that the holotype of Cornufer ( Petropedetes) johnstoni is a female specimen. However, according to Parker (1936) the determination of the sex of the holotype’s sex is wrong and it indeed is a subadult male of a size, where it is not distinguishable from subadult females. Parker’s correction has been later adopted by Amiet (1983). The holotype, showing the size, the large femoral glands and the lateral aspect of the head is shown in Fig. 5 View FIGURE 5 .
By the end of the 19th century Tympanoceros newtonii has been described by du Bocage (1895a) from the Island Fernando Po (= Bioko). The type of Tympanoceros newtonii deposited in the Musée Bocage in Lisbon, is lost ( Perret 1976). In contrast to the type of Cornufer johnstoni , the holotype of Tympanoceros newtonii must have been a male in breeding condition. The description of a second specimen form Bioko has been supplemented by an illustration of a male with well developed papilla (du Bocage 1895b). Both descriptions agree, among other characters, in rudimentary webbing, occurrence of a conical papilla on the tongue, and black bars on the limbs; but differ in the tympanum size. While the tympanum of C. johnstoni is given as half the diameter of the eye, it has been stated as equal to three-quarters of the eye for T. newtonii . However, this difference might be due to different methods of measuring the eye diameter. Another difference between the two descriptions concerns the tibiotarsal articulation. It is said to just reach the snout in the description of C. johnstoni ( Boulenger 1888) , while it is given to exceed the snout by four to five millimetres in T. newtonii (du Bocage 1895a). Although this difference appears crucial, the examination of this character state in seven specimens of P. “ newtonii ” from Bioko revealed very variable results, ranging from articulation reaching the snout-tip to exceeding the snout-tip by some millimetres. Werner (1898) reports P. johnstoni from Victoria, Cameroon. These frogs possess a small tympanum and their tibiotarsal articulations extend beyond the snout.
In 1900 Boulenger synonymised Tympanoceros and Cornufer with Petropedetes and provided the first key to the three species, known at that time. This key was based on the development of webbing and tympanum size. According to Boulenger (1900) the tympanum reaches half the size of the eye in P. johnstoni and more than half the eye diameter in P. newtonii . The differentiation of the species in Boulenger’s (1900) key was based on specimens collected by Mr. Bates on the Benito River (= River Mbini or Woleu River, today Equatorial Guinea) and not from Fernando Po. In this paper Boulenger (1900) describes the tympanum as being even larger than the eye. Subsequently this statement has been adopted by later authors. Amiet (1983) gives a relation of tympanum to eye size of 100.00–120.00% typical for “ P. newtonii ” and 58.18–75.00% in P. johnstoni . A second diagnostic character presented by Boulenger (1900) is the development of a tympanic papilla in males of “ P. n e w t o n i i ”; a character obviously unknown at that time in P. johnstoni ; e.g. Werner (1898) likewise describes males of that species without tympanic papillae. The occurrence of males’ secondary sexual characters like tympanic papillae and the bony carpal spikes in P. johnstoni was first reported by Andersson (1907). Unfortunately Andersson’s (1907) specimens from Bibundi on the foot of Mt. Cameroon, were destroyed during World War II (F. Geller-Grimm, Museum of Wiesbaden, pers. comm.).
Another character believed to distinguish “ P. newtonii ” and P. johnstoni was the position of the tympanic papilla ( Amiet 1983). According to Amiet the papilla is positioned close to the upper tympanic border in P. parkeri and P. johnstoni , but is positioned more centrally in “ P. newtonii ” ( Amiet 1983) . However, the papilla of the holotype from Bioko has been described as being close to the upper tympanic border and likewise illustrated for a second specimen of T. newtonii (du Bocage 1895a, 1895b). du Bocage’s (1895b) drawing is thus more similar to P. johnstoni than to “ P. newtonii ” as defined by Amiet (1983). It is hence plausible to assume that the morphological similarities between C. johnstoni and T. newtonii have been simply overseen due to the fact that Boulenger’s (1888) holotype of C. johnstoni does not possess the characteristic seasonal traits of breeding males and consequently never has been associated with du Bocage’ (1895a) holotype of T. newtonii . Furthermore Boulenger’s (1900) wrong assignment of specimens from the Benito River ( Equatorial Guinea) to P. newtonii from Bioko led to the fact that a wrong diagnostic character for P. newtonii (tympanum larger than the eye) has been adopted in subsequent literature. As P. johnstoni from Bioko and the mainland are morphologically indistinguishable, we herein place P. newtonii into the synonymy with the older name P. johnstoni: Tympanoceros newtonii du Bocage, 1895 = Petropedetes johnstoni ( Boulenger, 1888 “1887”) new synonym. Hence, P. “ newtonii ” (sensu Amiet 1983) on the mainland of western Central Africa requires a new scientific name (see below).
Bioko is located only 32 km from mainland Africa and is separated by shallow water (< 60 m). This island has probably been connected to the mainland during the last ice age ( Jones 1994; Rohling et al. 1998). The fauna of Bioko is of continental origin, comprising various faunal elements, like chameleons, lacertids and anurans, being present on the mainland and Bioko, but not occurring on other islands of the Cameroon volcanic line ( Jones 1994; Jesus et al. 2005; Weinberg 2008). In contrast, the islands São Tomé and Príncipe in the Gulf of Guinea are seperated by a depth of more than 1800 m and are inhabited by their own endemic fauna with a deviating origin ( Loumont 1992; Drewes & Stoelting 2004; Measey et al. 2007). Hence, the occurrence of the same Petropedetes species on Bioko and the mainland seems possible. However, we cannot exclude the possibility that molecular investigations comparing P. johnstoni from Bioko with those from the mainland may reveal differences on the species level. For example Bioko is inhabited by its own subspecies of the Cameroon toad ( Amietophrynus camerunensis poensis ), with longer extremities, toes and fingers ( Perret 1966). On the other hand specimens of Hyperolius ocellatus from Bioko and adjacent mainland are currently regarded as belonging to one subspecies, different to those from south of the Sanaga River ( Perret 1975). If future investigations would reveal that specimens from Bioko do belong to a separate evolutionary lineage, the name P. newtonii would be available for the island populations.
As herein defined, P. johnstoni is easily differentiated from other Central African congeners by its rudimentary webbing. P. palmipes and P. perreti are both fully webbed, P. c a m e ro n e n s i s and P. juliawurstnerae sp. nov. are half-webbed. The presence of a distinct tympanum and a tympanal papilla distinguishes P. johnstoni from P. cameronensis and P. palmipes , which lack these characters. The small but distinct tympanum and the large femoral glands differentiate P. johnstoni from P. euskircheni sp. nov., P.
parkeri and P. v u l p i a e sp. nov., species with reduced webbing. Furthermore P. johnstoni seems to be strictly restricted to lowland localities in coastal areas, whereas other species live in more mountainous areas and further away from the coast.
Etymology. This species has been named by Boulenger (1888) to honour Mr. Henry Hamilton Johnston, who collected the type specimen.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Petropedetes johnstoni
| Barej, Michael F., Rödel, Mark-Oliver, Gonwouo, Legrand Nono, Pauwels, Olivier S. G., Böhme, Wolfgang & Schmitz, Andreas 2010 |
Cornufer johnstoni
| Boulenger 1888 |