Laemosaccus andersoni Hespenheide, 2019
|
publication ID |
https://doi.org/10.1649/0010-065X-73.4.905 |
|
DOI |
https://doi.org/10.5281/zenodo.5213732 |
|
persistent identifier |
https://treatment.plazi.org/id/DB5AFC3E-C72C-5724-C0B1-E0B7FDD7F93B |
|
treatment provided by |
Carolina |
|
scientific name |
Laemosaccus andersoni Hespenheide |
| status |
sp. nov. |
Laemosaccus andersoni Hespenheide , new species
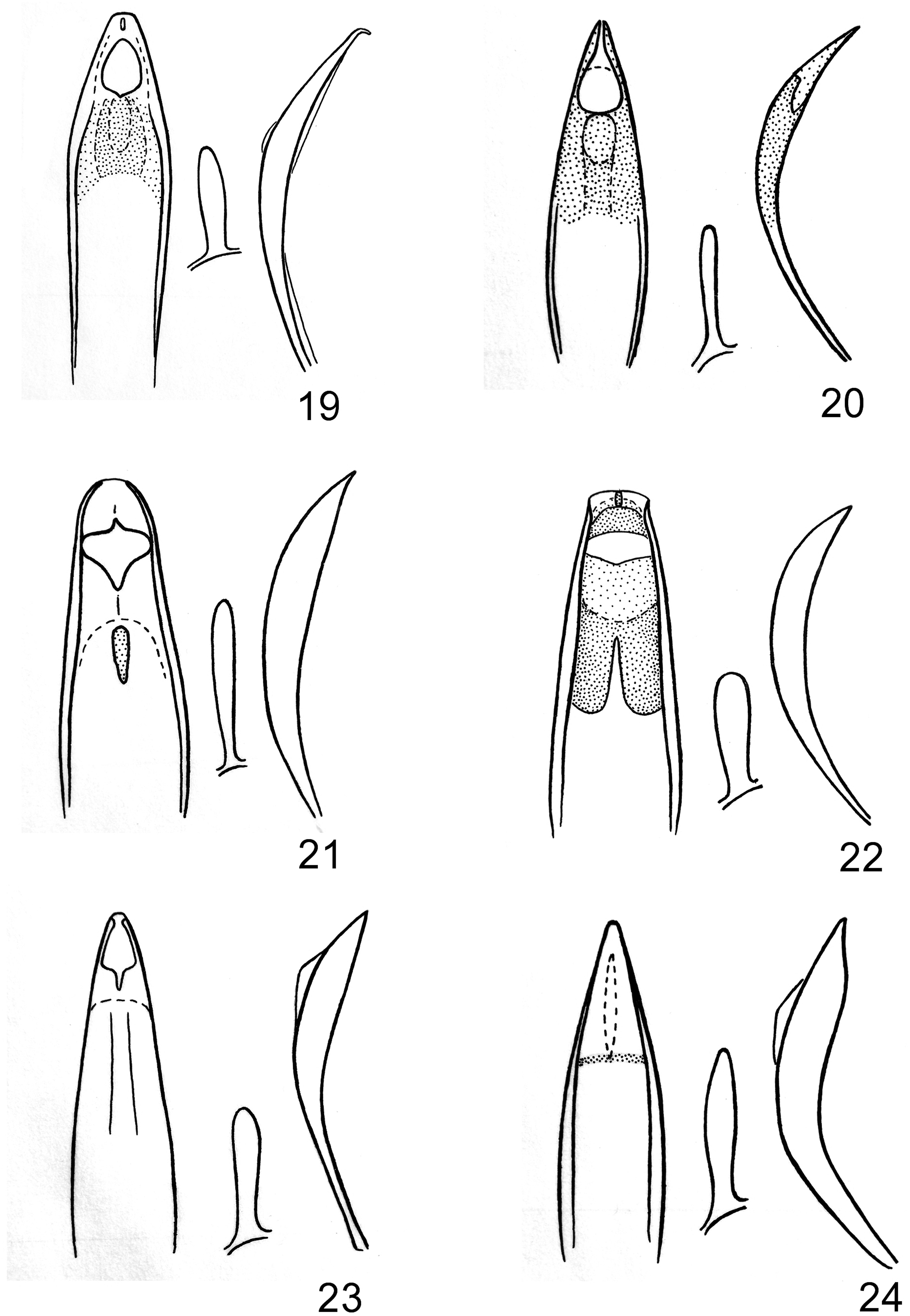
Zoobank.org/ urn:lsid:zoobank.org:act: ( Figs. 7, 8 View Figs , 19 View Figs )
Description. Holotype Male. Length 3.6 mm, width 1.75 mm ( Figs. 7, 8 View Figs ). Robust, subcylindrical in cross section, slightly obovate, abruptly rounded and nearly transverse behind, attenuate and narrowly rounded in front, black except each elytron with large, oval, red-orange spot on anterior 2/3 and just interior of lateral margins nearly to suture; pronotum and elytra glabrous, thorax and abdomen ventrally with punctures each with a silvery seta, nearly covering the ventral surface except for small area along posterior portion of metasternum along midline, setae semierect on abdominal ventrite 5, head with sparse, inconspicuous setae on sides of rostrum between eyes and antennal insertions and behind eyes, setae more slender and semi-erect on femora, long, hair-like, and semierect on tergite 8; tergite 7 with small, scale-like setae on basal margin, otherwise glabrous. Head hemispherical, 0.8 mm wide, rostrum rounded-terete, moderately coarsely punctate with distinct medial carina, 0.6 mm long, antennae inserted at middle. Pronotum globose from above, nearly flat in lateral view, constricted before anterior margin, 1.25 mm long, 1.4 mm wide, broadest at basal third, lateral margins conspicuously arcuate, finely, evenly punctate, punctures rounded and separate on basal half, somewhat elongate and nearly confluent near midline on apical half, with distinct, fine medial carina on basal 3/4. Elytra distinctly wider than pronotum at base, 2.3 mm long, 1.75 mm maximum width, elytral striae narrower than intervals, striae distinctly, finely punctate, intervals raised and rounded, intervals 3 and 5 very weakly toothed. First abdominal ventrite very slightly concave along midline, with setae less conspicuous. Pygidium conspicuously convex (“domed”) at middle, postpygidium with broad, raised, polished prominence along basal half of midline. Profemora with very small, forward-directed, acute ventral tooth beyond middle. Genitalia as in Fig. 19 View Figs ; aedeagus 1.10 mm long.
Allotype Female. As male but 3.45 mm long, 1.45 mm wide, rostrum subcylindrical, more finely punctate; abdominal ventrites broadly glabrous along postero-medial margins, ventrite 5 subequal to ventrite 4 at middle, expanded at sides and equal to ventrites 3 + 4; tergite 7 very strongly convex, granular, glabrous except with erect, hair-like setae at apical fifth.
Specimens Examined. Holotype: Arizona: Cochise Co., Chiricahua Mts. , 2.5 mi. SW Portal, 5000’, 10.08.1983, R. S. Anderson ( CMNC) . Allotype: Same data as holotype ( CMNC) . Paratypes: USA: Arizona: Santa Rita Mts. , 5 to 8000 ft, July, F. H. Snow (9, SEMC) , 12.07.1950, P. P. Cook (1, SEMC) . Cochise Co., 2.5-5 km W Portal , 1646 m, 23.07.1989, R. S. Anderson, 89-12 (2, CMNC) ; same data as holotype (3, CMNC) ; Chiricahua M., 15.06.1939, 24.07.1955, D. J. & J. N. Knull (2, OSU) ; Chiricahua Mts., Horseshoe Can. , 6000’, 4.07.1977, S. McCleve (1, TAMU) ; Chiricahua Mts., Rucker Cyn. Dam , 25.07.2005, C. W. O’ Brien (1, ASUHIC) ; Cave Ck. Canyon , 8.08.1974, A. J. Gilbert, H. E. Gilbert (1, ASUHIC) ; Cava [sic] Cr , 3.09.1943 (1, EMEC) ; Chiricahua Mountains, South Fork Cave Creek , 31°52.47’N 109°10.92’W, 5440’, 23.07.2003, S. M. Clark (1, ASUHIC) GoogleMaps ; Douglas , 7.08.1933, 1.08.1936, W. W. Jones (5, EMEC, USNM) ; Bisbee, Quarry Cyn. , 5600’, 7-8- 1978 (1, SWRS) ; Huachuca Mts., Copper Canyon , 1737-1829m, 26.07.1989, R. S. Anderson, 89-23 (1, CMNC) ; Coconino Co., Oak Cr. Can., 21.07.50, D. J. & J. N. Knull (1, OSU) ; Oak Creek Cyn., Manzanita Camp , 30.07.1950, B. E. White (7, CASC) ; Gila Co., Sierra Ancha Mts., Workman Creek , 5.08.1977, S. McCleve (1, TAMU) ; Pima Co., Mt. Lemmon Hwy. , mi. 1-17, 1.08.2000, J. Huether (1, CMNC) ; [ Pinal Co.] Oracle , 7-7, Hubbard & Schwarz (1, USNM) ; Santa Cruz Co., Pena Blanca , 14.07.2000, F. W. Skillman Jr. (2, ASUHIC) . Mexico: Chihuahua: Basaseachi Falls , 1950 m, 28°11’N 108°13’W, 5440’, 5.07.2006, D. Furth (3, CHAH) GoogleMaps ; Coahuila: nr Jame , 33 mi. SE Saltillo, 7500’, 25.07.63, A. T. Howden (2, CMNC) ; Durango: 3 km W El Palmito , 2- 3.10.1976, E. Giesbert (1, EMEC) ; 25 km SW La Ciudad , 7300’, 22.06.1991, J. Rifkind, Pine / Oak (1, WFBM) ; 36 mi. W Cd. Durango , 9.06.1967, [A. R. Moldenke], on Ceanothus buxifolia (3. BMNH, CHAH, USNM) . Estado de México: Santa Catarina del Monte ± 12 km (air) ESE Texcoco , 2800 m, 20.06.2000, R. L. Westcott, beating Quercus sp. (1, TAMU) ; Nuevo León: Chipinque Mesa, Zoo , 3700’, 21.06.1983, C. W. & L. O’ Brien & G. B. Marshall (1, ASUHIC) .
Host. Adults of L. andersoni have been collected on Quercus sp. and Ceanothus “ buxifolia ” [= Ceanothus buxifolius Willd. ex Schult. f., Rhamnaceae ] in Mexico.
Etymology. This species is named in honor of Robert S. Anderson, the collector of the holotype and major collector and student of the Curculionidae ( e.g., Anderson 1989, 2002).
Discussion: This is one of the most distinctive of the L. nephele group of species in both the domed pygidium of both sexes and the form of the male genitalia. Some fresher specimens have a few postscutellar setae on the first elytral interval. Males vary in size from 2.85 to 4.10 mm (mean = 3.46 mm, n = 17); females vary from 3.10 to 4.40 mm (mean = 3.92 mm, n = 20).
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |