Paratischeria grossa Diškus & Stonis, 2021
|
publication ID |
https://doi.org/10.11646/zootaxa.5040.2.5 |
|
publication LSID |
lsid:zoobank.org:pub:9CEFEC89-1A91-47FA-BE07-DD8B9E6EE3C2 |
|
DOI |
https://doi.org/10.5281/zenodo.5531317 |
|
persistent identifier |
https://treatment.plazi.org/id/79DA8D55-5963-4EC4-9579-766A7C74EB5C |
|
taxon LSID |
lsid:zoobank.org:act:79DA8D55-5963-4EC4-9579-766A7C74EB5C |
|
treatment provided by |
Plazi |
|
scientific name |
Paratischeria grossa Diškus & Stonis |
| status |
sp. nov. |
Paratischeria grossa Diškus & Stonis View in CoL , sp. nov.
urn:lsid:zoobank.org:act:
( Figs. 44–73 View FIGURES 44–52 View FIGURES 53–57 View FIGURES 58–61 View FIGURES 62–69 View FIGURES 70–73 )
Type material. Holotype: ♂, LAOS: Luang Prabang Province, 30 km SW Luang Prabang, 19°44’57”N, 101°59’35”E, elevation ca. 460 m, mining larva on Dendrocnide sp. (Urticaceae), 16.ii.2020, ex pupa ii–iii.2020, field card no. 5335, leg. A. Diškus & M. Jocius, genitalia slide no. AD1066 ♂ ( ZIN) GoogleMaps . Paratypes: 2 ♀, same label data as holotype, genitalia slide no. AD1059 GoogleMaps ♀ ( ZIN) ; 1 ♂, same label data as holotype, genitalia slide no. AD1037 GoogleMaps ♂ (from adult in pupal exuviae, no pinned moth preserved) ( GNU) ; 2 ♂, same label data as holotype, genitalia slide nos. AD1065 GoogleMaps ♂, AD1067 ♂ (from adult in pupal exuviae, no pinned moth preserved) ( NRC) .
Diagnosis. Paratischeria grossa sp. nov. belongs to the P. ferruginea group (for a description of this group see Stonis et al. 2017a). Externally, this new species differs from South American and African members of the group in the ochre colour of the forewing speckled with black-brown scales; from the most similar South East Asian P. boehmerica sp. nov. (described above) it differs in the larger size and darker, intense yellow-ochre or ochre colour of the forewing. In the male genitalia, P. grossa differs from P. boehmerica in the unique, caudally truncated dorsal sclerite ( Figs. 58 View FIGURES 58–61 , 68 View FIGURES 62–69 ), presence of a rounded, lobe-like anterior processes of the tegumen ( Fig. 63 View FIGURES 62–69 ), and the pointed, process-like lobe of the valva ( Figs. 67, 69 View FIGURES 62–69 ). In the female genitalia, the significantly larger size (about 1910 µm) and the longer corpus bursae with large coils distinguish P. grossa from P. boehmerica .
Male ( Figs. 54, 55 View FIGURES 53–57 ). Forewing length about 4.0 mm; wingspan about 8.8 mm (n = 1) (also see Remarks).
Head. Frons, palpus, and pecten pale yellowish ochre to dark ochre; frontal tuft and collar glossy brown-ochre to pale ochre, with some purple iridescence; antenna distinctly longer than one half the length of forewing; flagellum ochre to dark brown-ochre; sensilla long, distinctive, whitish cream.
Thorax. Tegula and thorax dark ochre, pale ochre distally. Forewing intense yellow-ochre with strong blue iridescence, sometimes dark brown at base. Brown-black or black scales very sparse, except for a small, irregular, subapical patch along costa and a small, irregular tornal spot; fringe bright yellow-ochre, except for a black-grey area on costal margin of the forewing; fringe line distinctive, comprised of brown-black scales; forewing underside dark brown-grey to grey-black, with a black basal area along costa and slender area along fold; no androconia. Hindwing grey to dark grey on upper side and underside, without androconia; fringe pale brownish grey or ochreous grey. Legs glossy, pale ochreous yellow on underside, dark grey-black on upper side (also see Remarks).
Abdomen. Metallic glossy, dark brown, with some purple iridescence on upper side; blackish brown with some ochre scales on underside; anal tufts short, indistinctive, yellowish ochre; genital segments golden ochre. Genitalia ( Figs. 58–69 View FIGURES 58–61 View FIGURES 62–69 ) with capsule 670–685 µm long, 380–420 µm wide. Uncus with two long and slender lobes. Valva ( Figs. 58, 61 View FIGURES 58–61 , 69 View FIGURES 62–69 ) 465–480 µm long (excluding basal process), basally with a pointed, horn-like process ( Figs. 61 View FIGURES 58–61 , 67 View FIGURES 62–69 ). Anellus ( Figs. 60 View FIGURES 58–61 , 69 View FIGURES 62–69 ) only strongly thickened laterally and caudally. Dorsal sclerite strongly developed, truncated caudally (58, 62, 68). Vinculum large, widely rounded ( Figs. 61 View FIGURES 58–61 , 69 View FIGURES 62–69 ). Phallus 340–360 µm long, apically deeply bifurcated, without spines, basally very wide ( Figs. 58 View FIGURES 58–61 , 66 View FIGURES 62–69 ).
Female ( Figs. 53, 56, 57 View FIGURES 53–57 ). Forewing length 4.1–4.2 mm; wingspan 9.1–9.3 mm (n = 2). Similar to male.
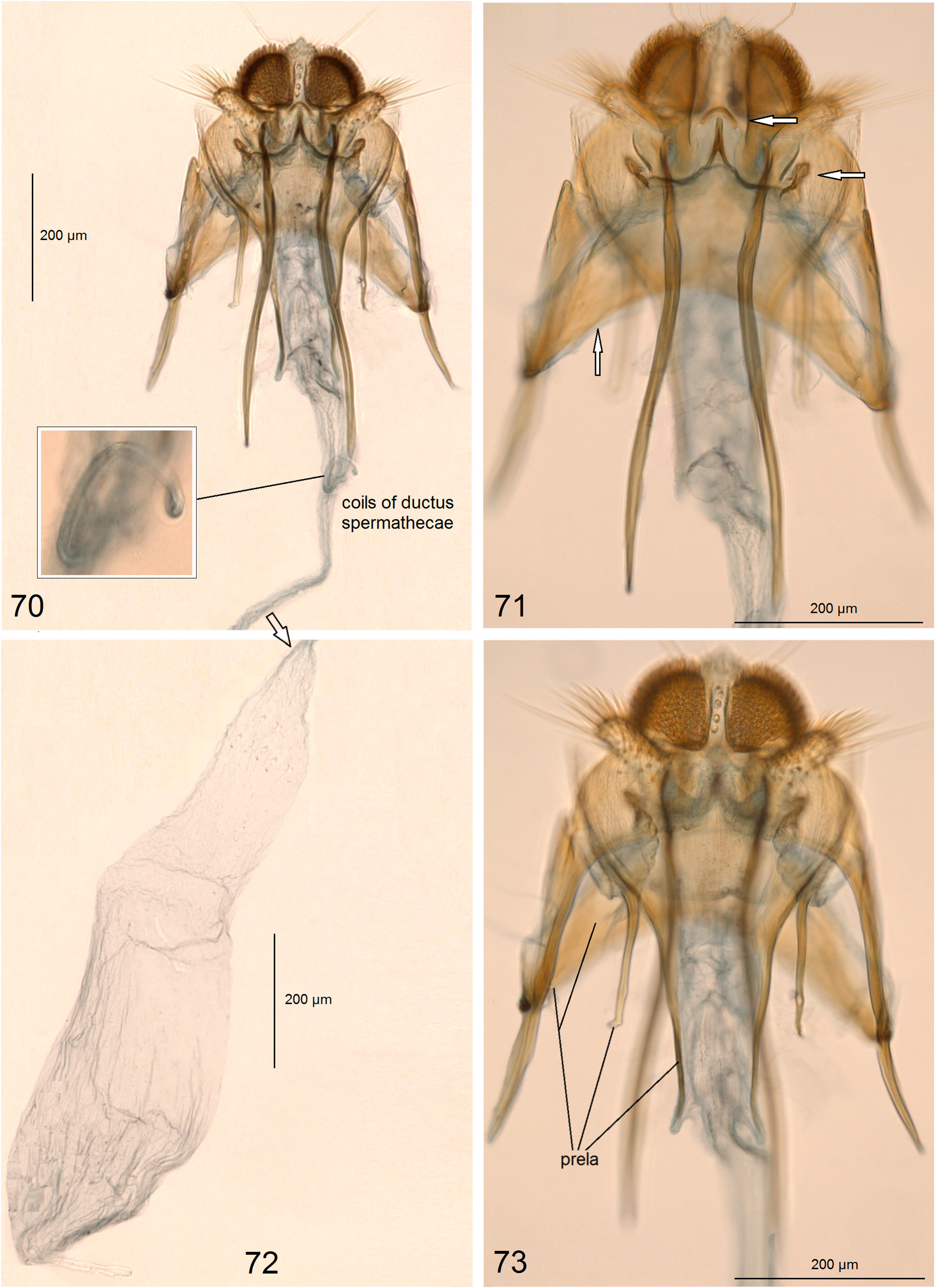
Genitalia ( Figs. 70–73 View FIGURES 70–73 ) 1910 µm long. Ovipositor lobes large, rounded, densely clothed with short, modified setae (‘peg setae’); area between ovipositor lobes wide, with some short setae, tiny papillae, and a larger papilla caudally. Second pair of lobes, lateral and anterior to the ovipositor lobes, 3–4 times smaller than ovipositor lobes, but bearing very long, slender lamellar setae. Anterior apophyses distinctly shorter than posterior apophyses; prela ( Fig. 73 View FIGURES 70–73 ) comprised of three pairs of projections; one of these pairs form a large, angular, plate-like thickening ( Fig. 71 View FIGURES 70–73 ). Corpus bursae long and slender, without pectinations or signum. Accessory sac absent. Ductus spermathecae slender, very short, with two large coils ( Fig. 70 View FIGURES 70–73 ).
Bionomics ( Figs. 44–52 View FIGURES 44–52 ). The host plant is Dendrocnide sp. , possibly D. sinuata (Blume) Chew ( Figs. 45, 46 View FIGURES 44–52 ) which, whilst not documented from Laos or Cambodia (Plants of the World Online), is known to occur in all the surrounding countries ( China, Myanmar, Vietnam, Malaysia) and so is expected to occur there also. The larvae mine leaves in February and produce irregular blotch-like leaf mines, with little or no frass ( Figs. 47–52 View FIGURES 44–52 ). Pupation occurs in the leaf mine; a silken nidus is present but inconspicuous. Adults occur in late February and March. Otherwise, the biology is unknown.
Distribution. The species is known from a single site in Luang Prabang Province, Laos, at an elevation of about 460 m.
Etymology. The species name is derived from the Latin grossus (thick, fat, large), referencing the large and robust male and female genitalia.
Remarks. One male paratype seems to be aberrant, unusually small, with forewing 2.7 mm long and wingspan 5.8 mm; in comparison with the holotype or female paratypes, the forewing of this specimen is slightly paler and with an additional, oblique, postmedian patch of black scales; otherwise, as in the holotype. The genitalia are identical with the holotype but slightly shorter (with capsule 520 µm long). Unlike other specimens, this specimen was reared indoors; it was deposited at NRC ( Fig. 55 View FIGURES 53–57 ).
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |