Cladonota (Falculifera) rex
|
publication ID |
https://doi.org/ 10.11646/zootaxa.4750.4.11 |
|
publication LSID |
lsid:zoobank.org:pub:FAFAF3C9-F9CB-4EC3-9E37-82837E85A964 |
|
DOI |
https://doi.org/10.5281/zenodo.3717950 |
|
persistent identifier |
https://treatment.plazi.org/id/E23CB206-FF85-FFEB-9A98-FA47FC07FE19 |
|
treatment provided by |
Plazi |
|
scientific name |
Cladonota (Falculifera) rex |
| status |
|
Cladonota (Falculifera) rex England sp. nov.
urn:lsid:zoobank.org:act:77DB2A2A-72F7-477D-B313-2C144ADB6511
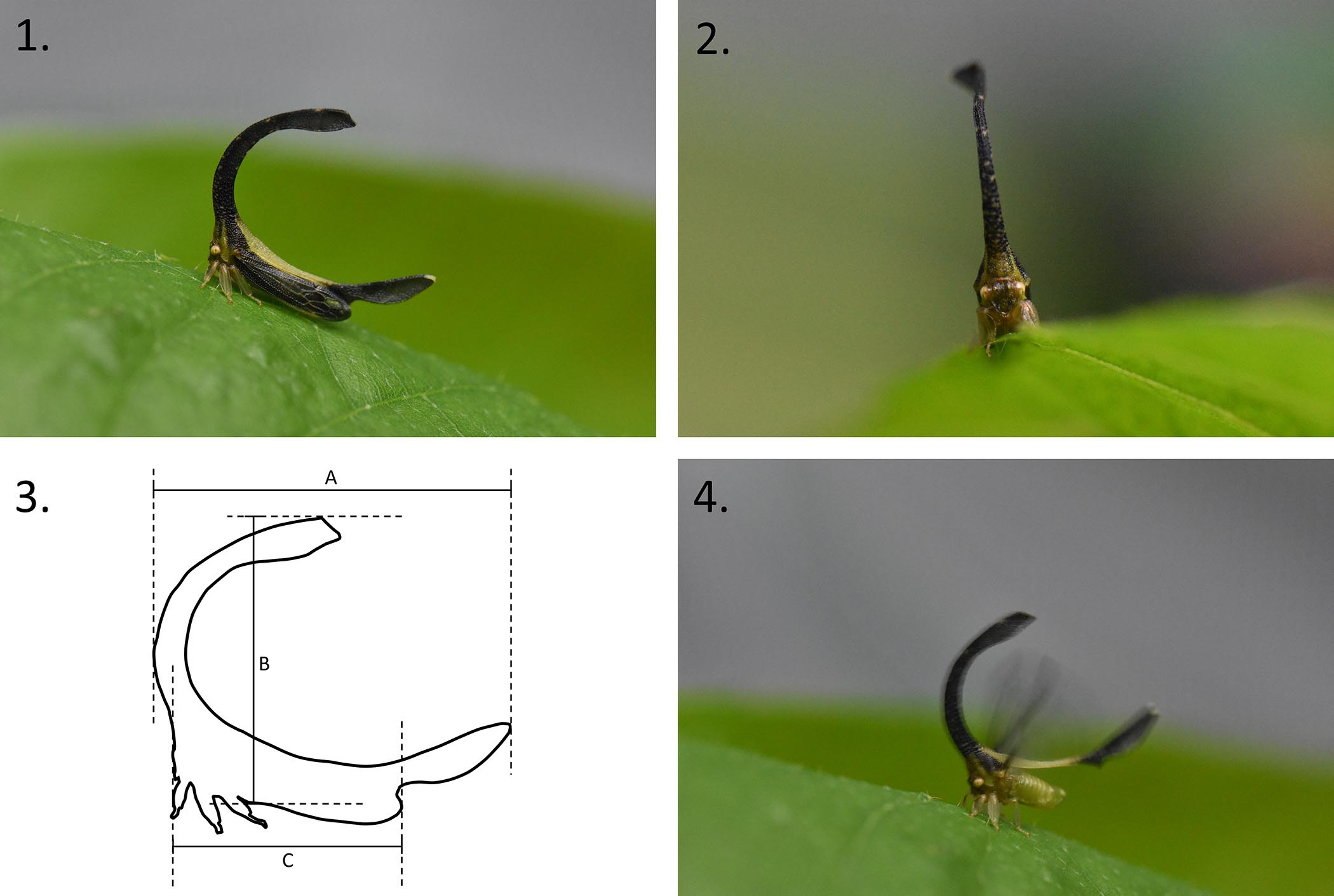
Diagnosis. Cladonota rex can most easily be distinguished from other members of the genus Cladonota by the unique and distinctive yellow-green ‘saddle’ located between the posterior and anterior pronotal processes, in combination with the open C-shaped pronotum (lack of both an intermediate process and a projection on the posterior edge of the anterior process) exhibiting concave regions on both the posterior and anterior process.
Description. Male: unknown. Holotype female ( Figures 1–4 View FIGURES 1–4 ):
Head: trilobed (supra-antennal lobes and clypeus). Clypeus rounded and pilose at the tip. Ocelli located closer to their nearest compound eye than to each other and lie above the centro-ocular line. Metopidium angled slightly forward from the face, blending into anterior process.
Thorax: Pronotum greatly expanded, with arching anterior and posterior processes that together form a C-shape. No intermediate process or projection on posterior edge of anterior process. Dorsal half of anterior process slightly sinuous in dorso-anterior view. Apex of anterior process split bilaterally into a Y-shape. Posterior process extends significantly further than the apex of the forewings at rest. Entirety of posterior process, and approximately the distal third of anterior process, have slightly concave sides. Vast majority of pronotum heavily punctate, with punctations becoming wider and shallower distally. Each punctation contains a single seta. Erect setae found along the ridges of both pronotal processes. Legs all with foliaceous tibia.
Forewings: Proximal regions of the forewings punctate and coriaceous. Punctations also each contain a single seta. Apical limbus broad and wrinkled. Veins predominantly hyaline but dark in places.
Colour: Ocelli pale yellow with darker centres. Compound eyes yellow with darker striations. Pronotum predominantly dark brown to black, with a large ‘saddle’ region of yellow-green between the anterior and posterior processes, and small intermittent specks of green on the anterior process. Yellow-green streaks, with acute apices, immediately adjacent to the distal edge of compound eyes. Posterior process with small white patch at tip. Metopidium largely yellow-green with small dark patches. Legs, abdomen, and underside of thorax all yellow-green in colour. Forewings dark brown, except for translucent segments in entirety of the 2 nd apical cell and a smaller region on 3 rd apical cell, as well as two on apical limbus, at posterior and dorsal most points.
Measurements ( Fig. 3 View FIGURES 1–4 ): face to posterior tip of forewings— 6 mm, dorsal edge of anterior process to ventral edge of abdomen beneath humeral angles— 9 mm, anterior most edge of anterior process to posterior most edge of posterior process— 11 mm.
Natural history and behaviour: Knowledge of the natural history and behaviour of C. rex is clearly limited because only a single specimen has been found thus far. However, some preliminary observations were made. The holotype was found stationary on the underside of a leaf near the top of a Piper sp. plant approximately three metres tall. The plant itself was situated on the bank of a small river, about one metre from the water’s edge. It was not clear as to whether the C. rex individual was feeding on Piper sp., and therefore it cannot be confirmed as a true host plant; however, due to the amount of time the individual remained stationary, some association likely exists. There were no other C. rex individuals found nearby, indicating that, like other Cladonota species ( Lin 2006; Godoy et al. 2006), C. rex is solitary in adulthood. Planthopper nymphs (infraorder Fulgoromorpha) were located on the same leaf as the C. rex individual but no obvious species interaction was evident. A single ant ( Tapinoma sp.) was observed approaching the C. rex individual and probing and tapping the treehopper’s face, near the antennae. This seemingly prompted the treehopper to lift the posterior process of the pronotum away from the abdomen and buzz its wings ( Fig. 4 View FIGURES 1–4 ). The ant immediately fled. Together these observa- tions indicate that C. rex , and likely other Cladonota species, use wing-buzzes as a deterrent to attention from ants and possibly other unwanted mutualists or predators.
Distribution: Known only from the type locality in Costa Rica.
Material examined: Holotype female from COSTA RICA ( MZUCR). With labels: “ COSTA RICA. Heredia Province, La Selva Bio. Station / 10.4337°N, 84.0080°W, 60m. On Piper sp. / 27.Jun.2019. S. J. England” and red holotype label “ HOLOTYPE / Cladonota rex / England”. GoogleMaps
Etymology: the species name rex is given in honour of the immense contributions of Reginald “Rex” B. Cocroft to humankind’s understanding of treehopper behaviour and ecology. It is utilised as a noun in apposition. Discussion: The setae associated with pronotal pits appear to be sensory in function, as has been previously suggested in other treehopper genera ( Wood & Morris 1974; Wood 1975; Dietrich 1989; Stegmann 1998).
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |