Tonnacypris estonica ( Järvekülg, 1960 )
|
publication ID |
https://doi.org/10.11646/zootaxa.4942.4.2 |
|
publication LSID |
lsid:zoobank.org:pub:EB79F747-805C-46AB-BBC1-191191B860A2 |
|
DOI |
https://doi.org/10.5281/zenodo.4612060 |
|
persistent identifier |
https://treatment.plazi.org/id/ED6E87E9-FFFA-FFB5-FF66-FAFAFCC968D1 |
|
treatment provided by |
Plazi |
|
scientific name |
Tonnacypris estonica ( Järvekülg, 1960 ) |
| status |
|
Tonnacypris estonica ( Järvekülg, 1960) View in CoL
( Figs 2–6 View FIGURE 2 View FIGURE 3 View FIGURE 4 View FIGURE 5 View FIGURE 6 )
For an abbreviated synonym list see Van der Meeren et al. (2009).
Material examined. Five dissected females ( WOC 13, WOC53, WOC55, WOC58, WOC59) and two dissected males ( WOC 54, WOC56) ( Table 1 View TABLE 1 ).
Dimensions. Male (LV, n = 2) length 1.03–1.22 mm, H/L ratio 0.48–0.50. Female (LV) length 1.16–1.28 mm (n = 5), H/L ratio 0.47–0.49 (n = 4).
Description. Carapace ( Fig. 2 View FIGURE 2 ) sub-reniform in lateral view. Dorsal margin arched, with greatest height situated at about 38% to 42% of length from anterior margin (n = 8 valves). Anterior margin evenly and broadly rounded. Posterior margin narrowly rounded and more or less pointing downwards. Ventral margin slightly concave at middle part with wide and blunt inward projection. In dorsal view, greatest width situated at mid-length, equalling to about 1/3 of length. Both anterior and posterior margins pointed. Valves in interior view, calcified inner lamella wide, with selvage running very close to margin of outer lamella. With up to eight fine striae on anterior calcified inner lamella ( Fig. 2I–K View FIGURE 2 ), less developed striae on posterior calcified inner lamella. Small antero-ventral peg of female LV (as described in Mongolian specimens; Fig. 1D, F, G View FIGURE 1 of Van der Meeren et al. 2009) observed on both male (e.g., WOC56, Fig. 2D View FIGURE 2 ) and female (e.g., WOC53, Fig. 2J View FIGURE 2 ) Tibetan specimens, but absent in some specimens (e.g., WOC13, Fig. 2I View FIGURE 2 , WOC54, unfigured).
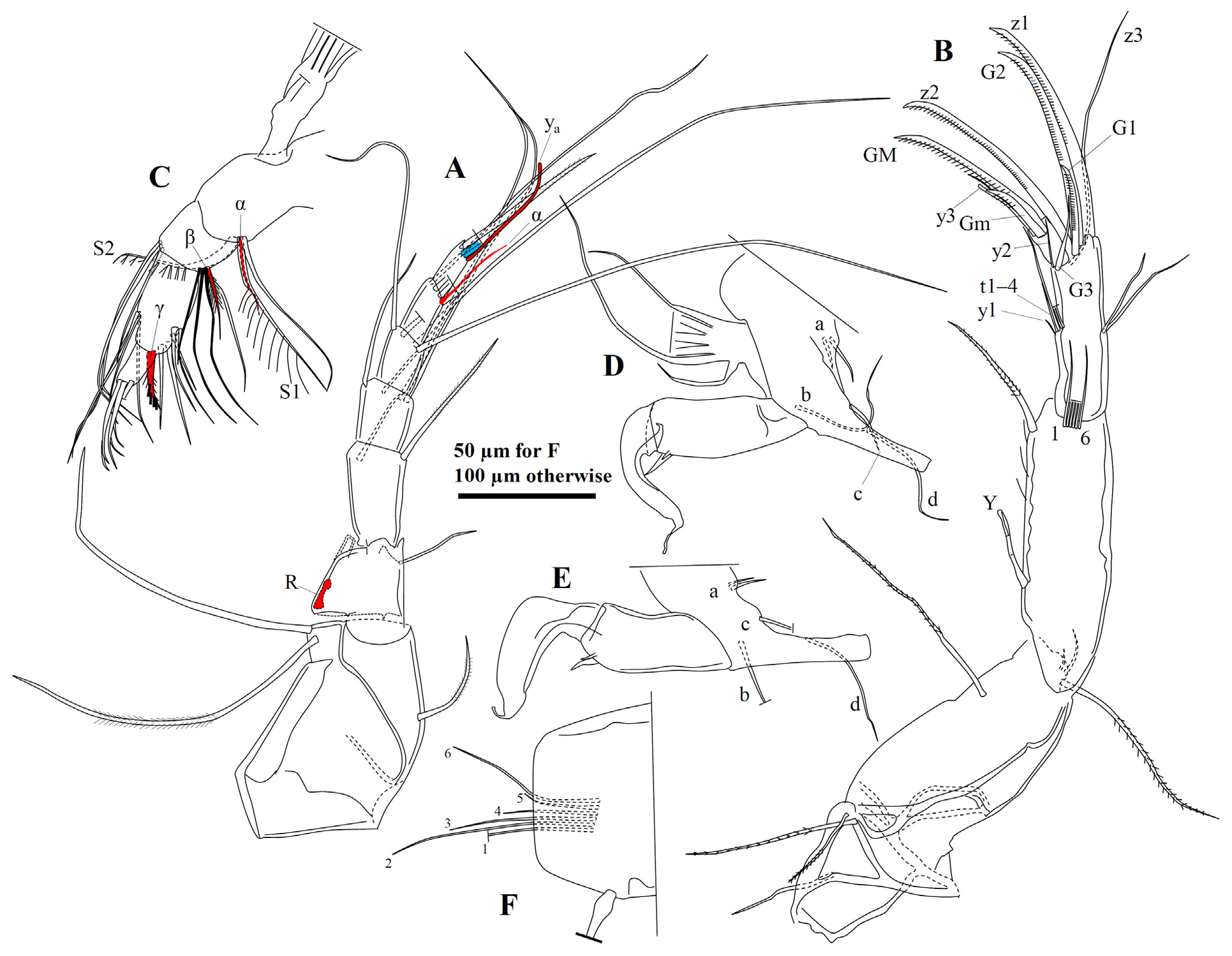
Soft parts of male. A1 ( Fig. 3A View FIGURE 3 ) morphology similar to that of female (see Van der Meeren et al. 2009), with eight segments, first two of which fused into large base, supporting one short dorsal seta and two long, thick ventral setae. Third segment trapezoidal, with one fine dorso-apical seta extending to end of fourth segment and elongate RO, situated on ventro-apical part of segment. Fourth segment elongate, with one dorso-apical seta extending to mid-length of seventh segment, and one ventro-apical seta extending to end of fifth segment. Fifth segment subquadrate, with two long dorso-apical setae extending well beyond end of terminal segment and two ventro-apical setae. Sixth segment sub-rectangular, with two very long dorso-apical setae and two shorter ventro-apical setae. Seventh segment elongate, with four long setae on inner side, and one fine seta (α) on outer side extending beyond end of terminal segment with ca. 2/5 of its length. Eighth segment slender, apically with three setae, and comparatively long and fine aesthetasc y a ca. twice as long as terminal segment. Ventral seta thick and slightly setulose.
A2 ( Fig. 3B, F View FIGURE 3 ) first protopodal segment with one slender ventro-posterior seta and two slender, setulose, unequal ventro-apical setae. Second protopodal segment robust, with one plumose, ventro-subapical seta extending almost to end of first endopodal segment. Longest seta of exopodite slightly setulous along distal half, and extending about three-quarters of length of first endopodal segment. Other two exopodal setae much shorter. Aesthetasc Y situated well behind mid-length of first endopodal segment, reaching slightly beyond mid-length of this segment. Six setae on intero-apical part of first endopodal segment unequally long, with second one reaching mid-length of penultimate segment, second to fifth ones progressively shorter. Ventro-apical seta of first endopodal segment robust and setulose, reaching base of terminal segment or slightly beyond. Second endopodal segment not sub-divided, aesthetasc y1 minute, situated immediately behind t setae. t setae unequal in length, longest one reaching distal end of terminal segment. z1 and z2 transformed into robust, serrated claws, with z1 slightly longer. z3 not transformed, reaching slightly beyond z1. G1 reduced, ca. 1/3 of length of z1, serrated. G2 slightly shorter than z1, and serrated. G3 very slender, seta-like, shorter than G1. Aesthetasc y2 fine, extending to about end of terminal segment. GM about 2.5 times length of Gm. GM and Gm serrated. Aesthetasc part of y3 slightly longer than Gm .
Md ( Fig. 3C View FIGURE 3 ) palp four-segmented, first segment’s vibratory plate with 5+1 wide rays. Setae S1 and S2 robust, plumose, accompanied by smooth seta of sub-equal length. Setae α fine and long, reaching to almost mid-length of S1. Second segment with three dorso-apical setae extending almost to tips of terminal claws. Antero-apical setal group on second segment with three setae extending to about end of terminal segment, accompanying seta slightly shorter. Seta β setulose, reaching mid-length of grouped setae. Third segment with 10 setae, including four long dorso-apical setae, seta γ (robust, plumose or serrated, situated on interior surface) and five unequal ventro-apical setae. Terminal segment with three robust, smooth claws and about four smaller setae.
Mx (not shown) nearly identical to female described by Van der Meeren et al. (2009). First segment of palp with eight setae. Second segment of palp with three larger and three smaller claws/setae. Tooth bristles on 3rd (distal) endite lobe smooth. Two setae on first (proximal) endite lobe with long setules (resembling those on S1 and S2 of Md palp).
L5 palps ( Fig. 3D, E View FIGURE 3 ) asymmetrical. Each trunk (1st endopodal segment) with two unequally sized sensory organs on ventro-apical part. 2nd endopodal segment finger-shaped and curved, with small terminal sensory organ. Right ‘finger’ wider than left one. Setae a and c much shorter than b and d. Vibratory plate with 6–7 rays, 1–2 of which shorter than others.
L6 ( Fig. 4A View FIGURE 4 ) with five segments. Setae d1 and d2 short. Seta e and f comparatively long. Seta g accompanied by much smaller seta. h1 similar length to g, conspicuously setulose. Claw h2 serrated along distal half. Seta h3 slightly shorter than h1. Setae d1, d2, e, f, g, and h3 faintly setulous.
L7 ( Fig. 4B, E View FIGURE 4 ) with three articulated segments, less sclerotized compared with other limbs. Seta d1 reaching well beyond distal end of first segment. Seta d2 sub-equal in length to d1, reaching slightly beyond mid-length of second segment. Seta dp extending to about distal end of second segment. Seta e setulose, extending to about end of terminal segment. Seta f setulose, situated at mid-length of third segment, extending to about end of segment. Seta h2 stiff. Seta h3 comparatively long and bearing small curve at middle part. Second to fourth segments with areas of clustered pseudochaetae.
Ur ramus ( Fig. 4C View FIGURE 4 ) comparatively long, bearing fine, short pseudochaetae along ventral edge. Sp situated close to distal end of ramus, robust, claw-like, serrated. Gp shorter than Ga, both serrated. Sa about 1/3 length of Ga. Ur attachment simple, unequally bifurcated.
Hp ( Figs 4D View FIGURE 4 and 5 View FIGURE 5 ) lateral shield with wide, sub-triangular medial lobe (ml) and small dorsal lobe (dl). Medial shield (ms) triangular, distally rounded and smooth. Loop ‘e’ distinctly recurving.
ZO ( Fig. 6 View FIGURE 6 ) with 20–21 internal rosettes of spines.
Remarks. The present specimens match the type specimens from Estonia ( Järvekülg 1960), and specimens from Mongolia ( Van der Meeren et al. 2009), in both valve morphology and chaetotaxy structures, especially in some details that are apparently very specific. For the soft parts, the key morphologies include lengths of the A2 swimming setae and the L5 a–d setae, setae and claw lengths of the L6, the presence and arrangements of pseudochaetae on the L6 and Ur, as well as the claw-like appearance of seta Sp on the Ur. Other aspects of the chaetotaxy are also generally consistent. Although some of the present specimens lack the small peg on the antero-ventral part of the LV, we consider this as intra-specific variation.
With the presence of males, however, it is shown that several aspects of the morphology of this species differ from other members of this genus. Firstly, the medial lobe (outer lobe) of the Hp is very wide, triangular, and unlike the slender and tapering distal medial lobe in other Tonnacypris species described in Van der Meeren et al. (2009). Secondly, the apical part of the medial shield is smooth, not with a roughened surface as in other Tonnacypris species. Thirdly, the number of ZO internal rosettes (20–21) is significantly smaller ( 39 in Tonnacypris edlundi Van der Meeren et al., 2009 , about 43 in Tonnacypris mazepovae Van der Meeren et al., 2009 , and about 46 in Tonnacypris lutaria ( Koch, 1838)) .
The above differences imply that T. estonica may be phylogenetic distant from some of the other species in the genus for which males are known. The type species of the genus, Tonnacypris loessica Diebel & Pietrzeniuk, 1975 , is only known from fossil specimens, so it is currently not possible to say which species are the most phylogenetic similar to it based on male characters. However, the large differences in the morphologies of the hemipenes and Zenker organs suggest there are possibly two lineages within the genus.
Eucypris gyirongensis Yang, 1982 (in Huang et al. 1982), later reported by Akita et al. (2016) (as Tonnacypris gyirongensis ) from Tibet, is similar to T. estonica in valve morphology, except that the reticulated pattern on the medio-dorsal part of left valve has not been observed in T. estonica ( Van der Meeren et al. 2009; this study). Mischke et al. (2010b) considered that E. gyirongensis is a junior synonym of Eucypris afghanistanensis Hartmann, 1964 , but detailed study of the soft-part anatomy of T. gyirongensis is required to verify its taxonomic identity.
Tonnacypris estonica has a disjunct known distribution, first described from Estonia (Järvekülg 1960), and later from the Mongolian Valley of the Great Lakes and in the northeastern part of the Mongolian Altai mountains ( Van der Meeren et al. 2009). It has been recorded in high densities in springs, but is also found in streams and lakes, predominantly occurring in oligotrophic habitats ( Van der Meeren et al. 2009). On the Tibetan Plateau it was collected from small ponds and a slowly flowing river at altitudes of 3325–4548 m ( Table 1 View TABLE 1 ). The discovery of males in this study suggests that T. estonica is a geographic parthenogen, similar to T. lutaria (and possibly Tonnacypris glacialis ( Sars, 1890)) ( Griffiths et al. 1998; Van der Meeren et al. 2009).
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
SuperFamily |
Cypridoidea |
|
Family |
|
|
SubFamily |
Eucypridinae |
|
Genus |