Paratullbergia chuana, Gao & Bu, 2020
|
publication ID |
https://doi.org/ 10.11646/zootaxa.4808.1.6 |
|
publication LSID |
lsid:zoobank.org:pub:6AC6B1BD-321B-4A7C-98F5-FE2563DE496F |
|
persistent identifier |
https://treatment.plazi.org/id/F614CE57-5378-FFC9-FF39-DB82FD6CFB3B |
|
treatment provided by |
Plazi |
|
scientific name |
Paratullbergia chuana |
| status |
sp. nov. |
Paratullbergia chuana sp. nov.
Figures 1–33 View FIGURES 1–8 View FIGURES 9–12 View FIGURES 13–18 View FIGURES 19–27 View FIGURES 28–33 , Tables 1, 2
Material examined. Holotype: ♂, (slide No. SC-2017011), Southwest China, Sichuan Province, Ganzi Tibetan Autonomous Region , Daocheng County, Xingmu town , 29°03’ N 100°30’E, elev. 3500 m, soil samples from coniferous forest, 15-VIII-2017, C.W. Huang coll. GoogleMaps Paratypes: 5♀ (slide Nos. SC-2017006–SC-2017009) , 2♂ (slide Nos. SC-2017010, SC-2017012), same data as holotype GoogleMaps ; 2♀ (slide Nos. SC-2017001, SC-2017002), Sichuan Province, Ganzi Region, Kangding City , Yala town , 30°06’ N 101°57’E, elev. 3100 m, soil samples from mixed forest, 11-VIII-2017 GoogleMaps ; 1♀ (slide No. SC-2017005), Sichuan Province, Ganzi Region , Litang County, 29°46’ N 100°26’E, elev. 3600 m, soil samples from coniferous forest, 13-VIII-2017 GoogleMaps ; 2♂ (slide Nos. SC-2017003, SC-2017004), Sich- uan Province , Ganzi Region , Daocheng County, Mula town , 28°44’ N 100°16’E, elev. 3900 m, soil samples from coniferous forest, 14-VIII-2017 GoogleMaps ; 3♀ (slide Nos. SC-2017013–SC-2017015) , 1♂ (slide No. SC-2017016), Sichuan Province, Ganzi Region , Derong County, Zhengdou town , 29°08’ N 99°35’E, elev. 3480 m, soil samples from coniferous forest, 17-VIII-2017 GoogleMaps ; 1♀ (slide No. SC-2017017), Sichuan Province, Ganzi Region , Daofu County, 30°40’ N 101°17’E, elev. 3550 m, soil samples from mixed forest, 23-VIII-2017 GoogleMaps ; 2♀ (slide Nos. SC-2017018, SC-2017019) , 1♂ (slide No. SC-2017021) , 2 juveniles (slide Nos. SC-2017020, SC-2017022), Sichuan Province, Ganzi Region , Danba County, 30°36’ N 101°40’E, elev. 2940 m, soil samples under bushes, 23-VIII-2017, C.W. Huang coll. GoogleMaps Holotype and most paratype specimens are deposited in Shanghai Natural History Museum ( SNHM), except two paratypes (slide Nos. SC-2017013, SC-2017016) which are deposited in Shanghai Entomological Museum ( SEM) .
Diagnosis. Paratullbergia chuana sp. nov. is characterised by the presence of one pair of pseudocelli on each segment of Th. I–Abd. V with a formula of 11/111/11111, the postantennal organ composed by 34–50 vesicles arranged in two rows, seta px present on Abd. IV, setae a2 and a4 on Abd. V as macrosetae, p4 as mesoseta, and only a weakly differentiated sensillum p3 on Abd. V.
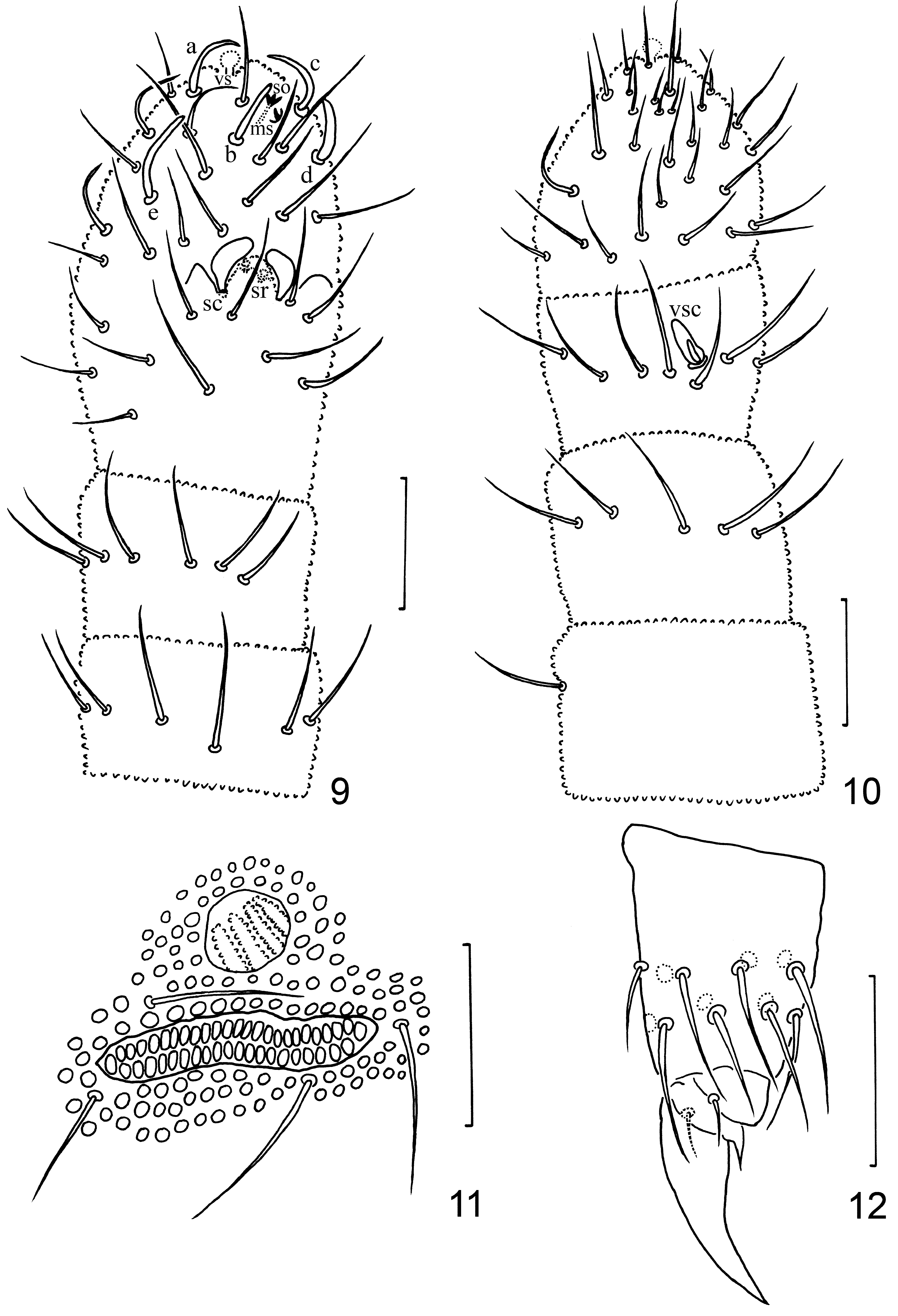
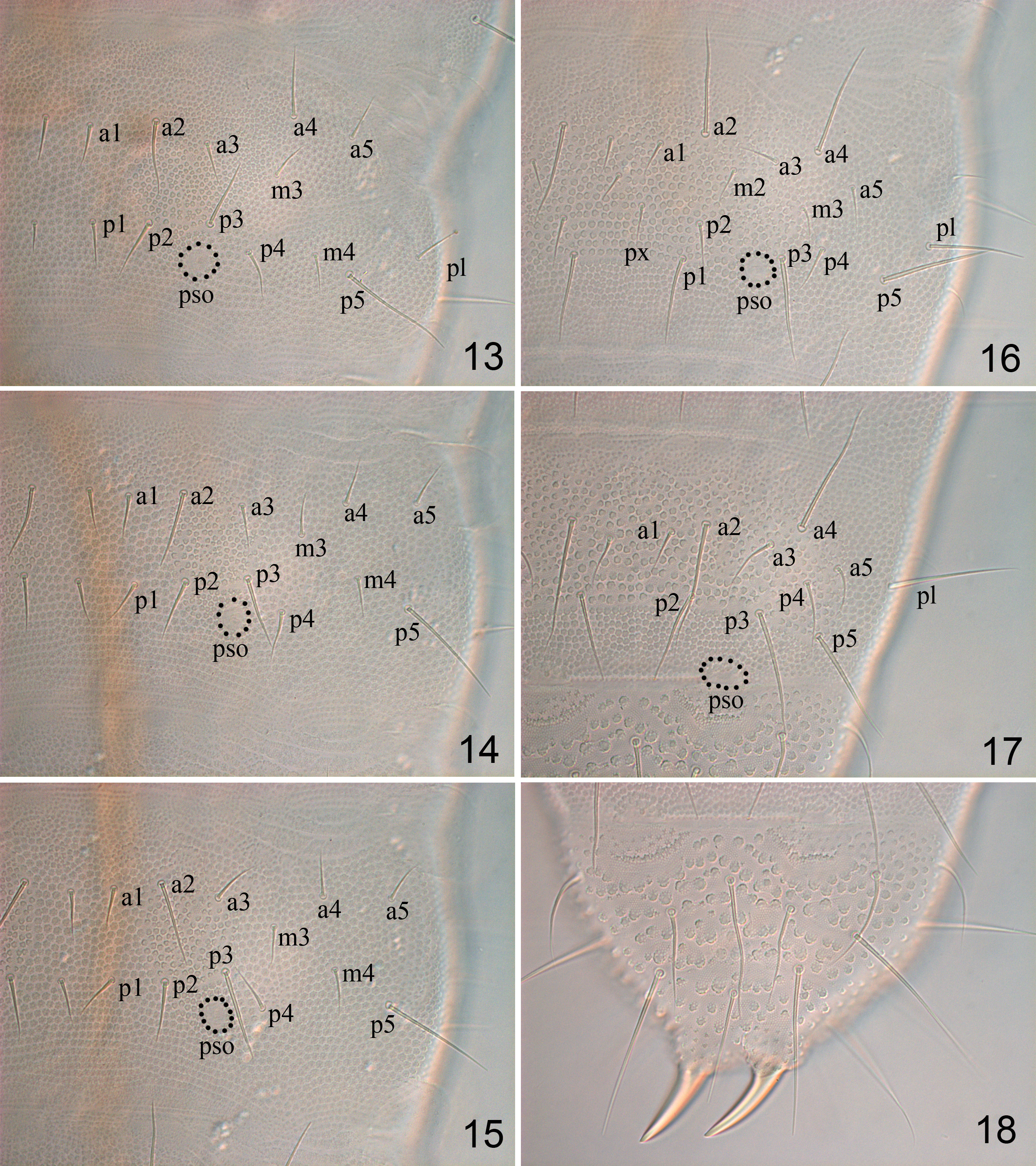
Description. Adult body 0.97 mm long on average (0.82–1.18 mm, n = 21), holotype 0.96 mm. Dorsal setae well differentiated into micro- and macrosetae ( Figs. 1, 7, 8 View FIGURES 1–8 ). Granulations coarse, formed by secondary granules, 3.5–4.0 μm in diameter. Pso formula as 11/111/11111 from head to abdomen, 8–10 μm in diameter. Antennal base with pso of type III, composed by 3–4 ridges from one side only ( Figs. 4 View FIGURES 1–8 , 11 View FIGURES 9–12 ). Posterior head, thorax and Abd. I–V with pso of type II, composed of 10–12 ridges from two sides ( Figs. 7, 8 View FIGURES 1–8 , 13–17 View FIGURES 13–18 ). On Th. I pos set behind seta m2, close to hind margin ( Fig. 7 View FIGURES 1–8 ). Th. II and III each with one pair of pso between setae p3/p4 and closer to p3 ( Fig. 8 View FIGURES 1–8 ); pos on Abd. I–III posterior to seta p3 ( Figs. 13–15 View FIGURES 13–18 ), on Abd. IV close to and at the same level as seta p3 ( Fig. 16 View FIGURES 13–18 ), on Abd. V between the border of Abd. V and VI ( Fig. 17 View FIGURES 13–18 ).
Cephalic seta a0 present (14–16 μm), two additional setae also present anteriorly, c1 absent, oc2 and sd5 as macroseta, 23–40 μm ( Figs. 2, 7 View FIGURES 1–8 ). Postantennal organ narrow, 25–35 μm long and 5–7 μm wide, composed of 34–50 oblong vesicles arranged in two rows, 45–47 in holotype ( Figs. 4–6 View FIGURES 1–8 ). Labrum with 4/5/4 setae. Labium with 5 papilla, 6 apical guard setae, 6 proximal setae, 4 basomedian setae, and 5 basolateral setae ( Fig. 3 View FIGURES 1–8 ).
Antenna (110–130 μm) shorter than head (140–165 μm), 150 μm in holotype. Antennal segment IV with five slightly thickened sensilla a–e, without basal heel; sensilla a, c, e long and curved toward inside, b straight and short, d thick and short, slightly curved ( Fig. 9 View FIGURES 9–12 ). Small microsensillum, subapical organite and one apical vesicle present. Antennal organ III ( Fig. 9 View FIGURES 9–12 ) consists of two small sensory rods concealed behind one large papilla and two thick sensory clubs bent toward each other, with four guard setae; one large ventral sensory club present ( Fig. 10 View FIGURES 9–12 ).
Legs short, without clavate tibiotarsal hairs ( Figs. 12 View FIGURES 9–12 , 21 View FIGURES 19–27 ). Coxa, trochanter, femur and tibiotarsus with 3/7/7; 6/6/5; 10/10/10; 15/15/14 setae on Leg I, II and III respectively. Claw 20–25 μm long ( Fig. 12 View FIGURES 9–12 ), with distinct empo- dial appendage (4–5 μm). Anal spines robust, 26–35 μm long ( Figs. 1 View FIGURES 1–8 , 18 View FIGURES 13–18 ).
Adult chaetotaxy given in Figs. 7, 8 View FIGURES 1–8 , 13–18 View FIGURES 13–18 , 22–27 View FIGURES 19–27 and Table 1. Microsensilla present on Th. II–III, lateral sensilla 35–45 μm long ( Fig. 8 View FIGURES 1–8 ). Thorax with 0, 2, 2 ventral setae. Abd. I–III with 2+2 axial setae each, with setae m3 and m4 present ( Figs. 13–15 View FIGURES 13–18 ). Abd. IV with seta px, m2 and m3 present, p4 as microseta ( Fig. 16 View FIGURES 13–18 ). Abdominal segment V ( Fig. 17 View FIGURES 13–18 ) with sensillum p3 slightly differentiated, 30–45 μm in length; seta a2 (33–55 μm) and a4 as macroseta (30–50 μm), p4 as mesoseta (17–30 μm). Crescentic ridges on abdominal segment VI present ( Fig. 18 View FIGURES 13–18 ).
Ventral tube with 4+4 apical setae and 2+2 basal setae ( Figs. 19, 20 View FIGURES 19–27 ). Number of ventral setae on Abd. II, III and IV variable, with 20–22, 22–23, and 26–28 setae respectively ( Figs. 22–24 View FIGURES 19–27 ). Female genital plate with 5+5 pre- genital setae, 8–11 circumgenital setae, one pair of eugenital setae and 2 pairs of post-genital setae ( Fig. 26 View FIGURES 19–27 ). Male genital plate with 18–30 circumgenital setae, 5+5 pregenital setae and 2 pairs of post-genital setae. ( Fig. 25 View FIGURES 19–27 ). Anal lobes with both setae 12’ and l3’ present ( Fig. 27 View FIGURES 19–27 ).
Distribution. China (Sichuan).
Etymology. The new species is named after “Chuan”, the abbreviated name of “ Sichuan ” in Chinese.
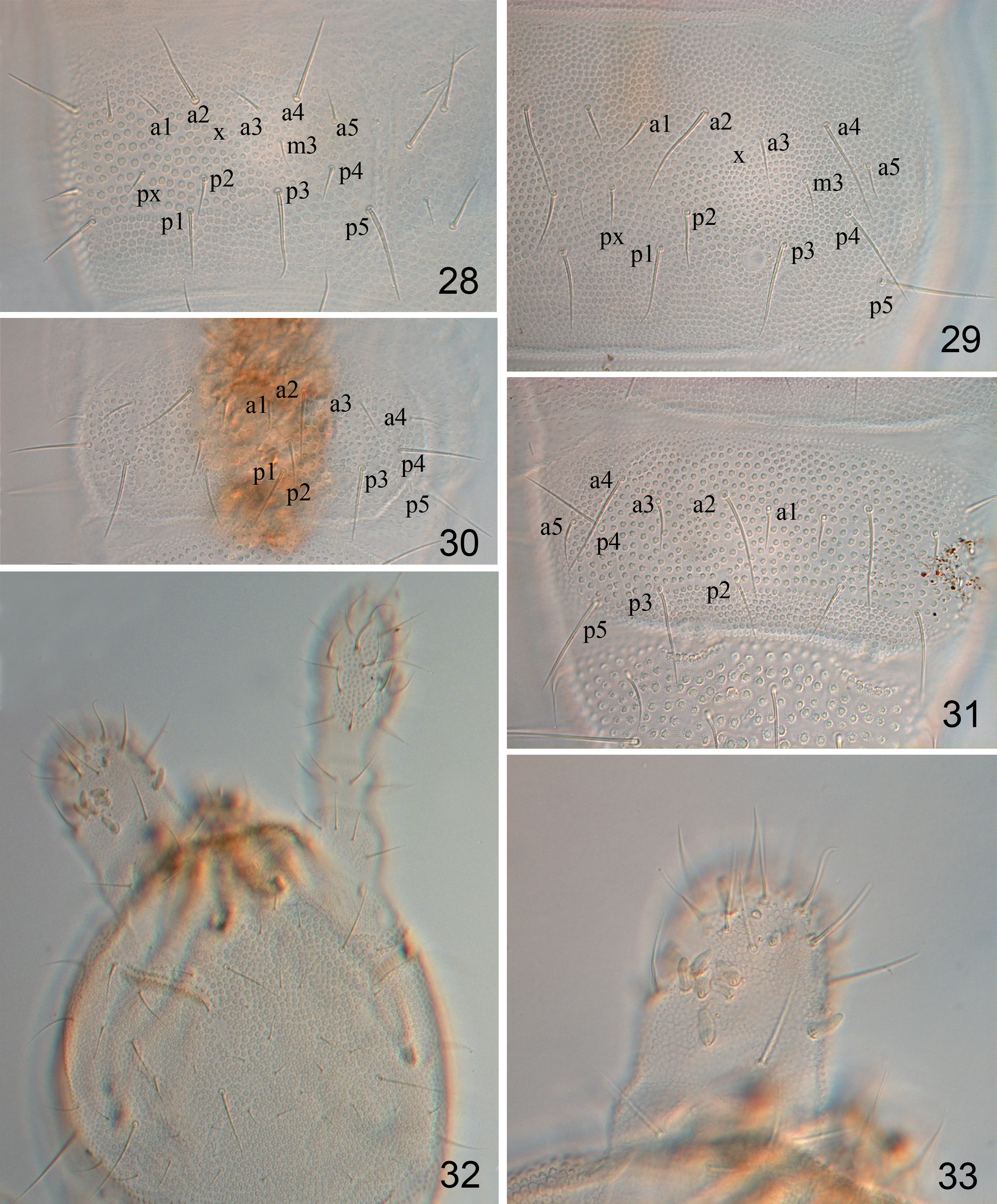
Variation. The chaetotaxy on tergite of Abd. IV is often variable: both m2 and m3 setae present in 11 adults (including holotype) ( Fig. 16 View FIGURES 13–18 ), with only m3 seta present in 9 adults and 1 juvenile ( Figs. 28, 29 View FIGURES 28–33 ), and both m2 and m3 absent in 1 adult and 1 juvenile ( Fig. 30 View FIGURES 28–33 ). Since the m row setae appear during ontogenesis, we described the present new species based on the fully matured individuals. In 6 individuals (slide Nos. SC-2017017–SC-2017022) from two close localities, seta p4 on both Abd. IV and V are macrosetae and much longer than in the specimens from other localities ( Figs. 29–31 View FIGURES 28–33 ), 36–40 and 30–35 μm respectively, which might reflect a variation between two populations.
A malformed antenna was observed in one adult individual (slide No. SC-2017008) ( Figs. 32, 33 View FIGURES 28–33 ), This antenna has only two segments, the second one having about 12 sensory clubs and 15 normal setae irregularly inserted dorsally ( Fig. 33 View FIGURES 28–33 ). This could have been caused by a fault in development or incomplete regeneration after damage.
Remarks. Paratullbergia chuana sp. nov. is the third Chinese species of the genus. It can be easily distinguished from P. changfengensis Bu & Gao, 2015 and P. qilianensis Bu & Gao, 2019 by the pseudocelli pattern: 11/111/ 11111 in P. chuana sp. nov., 11/122/ 11111 in other two species. According to the latest key by Bu and Gao (2015), it is similar to P. caroli Luciáñez, Ruiz & Simón, 1991 from Spain in having similar numbers of vesicles on PAO, but differs in the pseudocelli pattern (11/011/ 10011 in P. caroli ) and in the chaetotaxy of Abd. IV (setae m2 and m3 present in P. chuana sp. nov. vs. without m setae in P. caroli ). It is also related to P. indica Salmon, 1965 from India in having 1+1 pseudocelli on Abd. I–V, but differs in the pseudocelli on Th. I (present in P.chuana sp. nov. vs. absent in P. indica ) and the number of vesicles of PAO (34–50 in P. chuana sp. nov. vs. 22–24 in P. indica ). ( Table 2).
Discussion. The genus Paratullbergia has a similar habitus to Mesaphorura , and contains 11 species occurring in the Holarctic, China, India, South Africa and Australia. Six species: P. concolor Womersley, 1930 , P. macdougalli Bagnall, 1936 , P. callipygos (Börner, 1902) , P. changfengensis Bu & Gao, 2015 , P. qilianensis Bu & Gao, 2019 and P. chuana sp. nov. have a relative large, robust body (maximum 1.2 mm), and coarse cuticular granules; while the remaining five species are small with delicate body (maximum 0.7), and fine cuticular granules. The numbers and shapes of pso on thorax and abdomen, as well as the chaetotaxy on Abd. IV and V are very informative for species definition in this genus.
The species of the Paratullbergia have been found in diverse habitats, from rotten logs in coniferous and broadleaf forest, meadow, to coastal dunes associated with plant roots ( Dunger & Schlitt 2011). In China, P. changfengensis is only found at low density in a Park of Shanghai City, while P. qilianensis and P. chuana sp. nov. occur in natural forest of high altitude (more than 3000 m) with relatively large populations. It seems different species are adapted to different environments. Spores and mycelium fragments of soil fungi can be recognised in their gut contents, which indicates that they are fungivorous.
| SNHM |
Sudan Natural History Museum |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |