Altihoratosphaga, Hemp & Voje & Heller & Warchałowska-Śliwa & Hemp, 2010
|
publication ID |
https://doi.org/ 10.1111/j.1096-3642.2009.00542.x |
|
persistent identifier |
https://treatment.plazi.org/id/FF070E4D-CB43-FFA9-FC16-3B9102A36A14 |
|
treatment provided by |
Carolina |
|
scientific name |
Altihoratosphaga |
| status |
gen. nov. |
ALTIHORATOSPHAGA GEN. NOV.
Type species: Peronura montivaga Sjöstedt, 1909 .
Description: Male: fastigium verticis with median ridge in front antennal sockets, sulcate; small conus between antennal sockets; fastigium narrower than first antennal segment; antennal sockets bowl-like; eyes small, round, and prominent. Pronotum: broad, posterior margin upcurved, about a third of posterior pronotum triangle-shaped; surface of pronotum rugose to verrucose. Lateral lobes longer than deep; fore coxae unarmed, at outer margin laterad trace of blunt process; fore and mid femora with welldeveloped stout spines, to few and almost reduced spination; hind femora with numerous small stout spines. Tympana of fore tibiae conchate, not inflated. Forewings reduced, broad with broadly rounded tips. Venation reduced, but with basal modifications of areas MA and Cu 1a; hindwings rudimentary; emarginated tenth abdominal tergite. Subgenital plate large, protruding beyond abdomen; without styles; cerci long and slender, decussate.
Female: similar to males but mostly larger and somewhat stouter; rugosities of pronotal disc not as elevated as in males; ovipositor long and slender, moderately upcurved. Sexual dimorphism not very pronounced, with males and females quite similar in their general appearance.
Diagnosis
Altihoratosphaga species may be identified by a combination of characters: round tegmina with reduced venation and scattered black spots (except for A. nomima ), vestigial alae, shape of pronotum that is verrucose in most species, and the emarginate tenth abdominal tergite (only found similarly in the fullywinged Horatosphaga concava Ragge, 1980 ). Females may be recognized by their slender, long, and slightly upcurved ovipositor. Furthermore, Altihoratosphaga species are dark-green plump insects with little sexual dimorphism. They dwell in the lush herbaceous vegetation of forest edges and clearings in submontane–montane areas. The calling song is performed in the evening and during the night hours.
The shape of the pronotum of typical Horatosphaga and also Lamecosoma species (e.g. H. heteromorpha ) is uniform over its entire length, and is not divided into a broad anterior part and a triangle-shaped elevated posterior part, as in Altihoratosphaga . Both sexes in species of Altihoratosphaga are rather plump and dark green in colour, with rounded broad wings lacking web-like venation, whereas typical Horatosphaga are more slender, especially the males, and are mostly light green in colour, with more elongated wings, and with web-like venation in the male forewings.
Many species of Acrometopini are adapted to grassland habitats, often in savanna areas, whereas Altihoratosphaga species are bound to clearings and the forest edges of submontane–montane localities. All known species of Altihoratosphaga are endemic to particular areas, whereas many of the ‘typical’ Horatosphaga species are rather widespread forms [e.g. H. heteromorpha , Horatosphaga media Ragge, 1960 , Horatosphaga ruspolii (Schulthess, 1898) , Horatosphaga serrifera Schaum, 1853 , and Horatosphaga stylifera (Karny, 1910) ].
However, a number of Horatosphaga species are also adapted – like Altihoratosphaga species – to montane forest habitats. These are Horatosphaga parensis Hemp, 2006 , from the South and North Pare Mts of Tanzania, and Horatosphaga sabuk Hemp, 2006 , occurring in the eastern part of the Kenyan highlands. As in Altihoratosphaga , the males are flightless, with the alae being reduced. Compared with Altihoratosphaga , the shape of the tegmina is different, as is the general venation. The modification (web-like venation) of the areas MA and Cu 1a is pronounced in H. parensis and H. sabuk , whereas in Altihoratosphaga species this modification is hardly present. Furthermore, the general body shape of Altihoratosphaga is plumper in both sexes, and the colour of living insects is dark green, whereas in H. parensis and H. sabuk males and females are rather slender insects, especially the males, and the overall colour is a lighter green. Also, molecularly both the groups Altihoratosphaga and H. parensis plus H. sabuk cluster far apart, with H. parensis and H. sabuk forming a separate cluster within the Acrometopini investigated ( Voje, 2007; Voje et al., 2009).
Chromosomal characters
Analyses of the standard karyotype of A. montivaga , A. nou , H. parensis , H. heteromorpha , and M. kilimandjarica revealed diploid chromosome numbers of 2 n = 31 for males, consistent with the X0/XX sex chromosome system. In these five species, belonging to three genera, all chromosomes were acrocentric, consisting of three long and twelve medium to short pairs gradually decreasing in size; the X chromosome was the largest element ( Fig. 1A–C View Figure 1 ). The chromosomes of all African species of the tribe Acrometopini and the genus Monticolaria are characterized by paracentromeic C-bands that are similar in size. In species of Altihoratosphaga and Horatosphaga , interstitial C-bands are located in the pair L 3 in some of the individuals, and additionally in the pair M 6/7 of A. montivaga . In all cases these bands show polymor- phism in the quantity of heterochromatin (more or less) ( Fig. 1A, B View Figure 1 ). Only in M. kilimandjarica are thin terminal C-bands present in the three largest autosome pairs, and in the X chromosome ( Fig. 1C View Figure 1 ). B chromosomes, which are supernumerary to the standard complement, were found in two out of the four specimens of A. montivaga analysed. They were of the acrocentric type, similar to small pairs of autosomes. They show a large C-band (heterochromatic) near the centromere, and a smaller euchromatic part in the distal region ( Fig. 1A View Figure 1 ). B chromosomes were also observed in one of the two males of M. kilimandjarica . However, in contrast to those of M. kilimandjarica , the B chromosomes in A. montivaga are similar to medium-sized autosomes, and are probably all heterochromatic. In metaphase I this element was always distributed in the periphery plate ( Fig. 1C View Figure 1 ). Both types of B chromosomes were unstable in both the mitotic and the meiotic division in males.
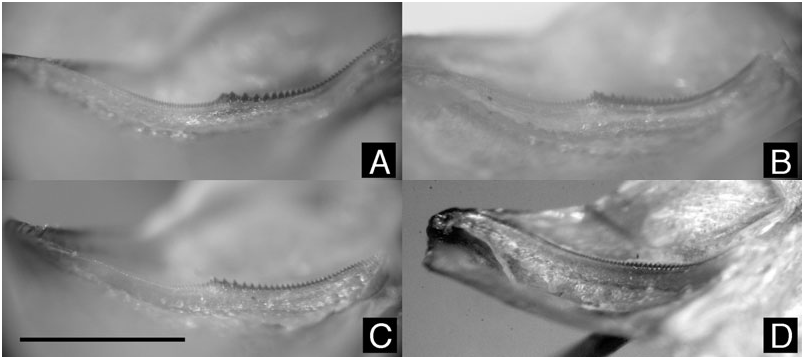
Stridulatory file
In all of the four species studied in detail ( A. montivaga , A. nou , A. hanangensis sp. nov., and A. nomima ), the stridulatory file on the lower side of the left tegmen showed a similar structure ( Fig. 2 View Figure 2 ). In its proximal part it bears relatively large teeth, whereas they decrease slowly in size to the end of the file at the edge of the tegmen. In A. nomima both parts are connected by a more or less continuous transition in tooth size ( Fig. 2D View Figure 2 ). In the other three species the teeth gradually become larger from the articulation towards the centre of the file. Distal of the middle the teeth become abruptly smaller, and then decrease slowly in size ( Fig. 2A–C View Figure 2 ).
A file structure with two distinct parts in three of the four known species of Altihoratosphaga is quite different from that observed in other species of the genus Horatosphaga ( H. heteromorpha , Horatosphaga leggei (Kirby, 1909) , and H. parensis ; K.-G. Heller, C. Hemp, unpubl. data). Differences in the number of teeth and their size, as can be seen in Figure 2 View Figure 2 , may indicate species-specific parameters, but cannot be evaluated at present because of insufficient data.
Songs
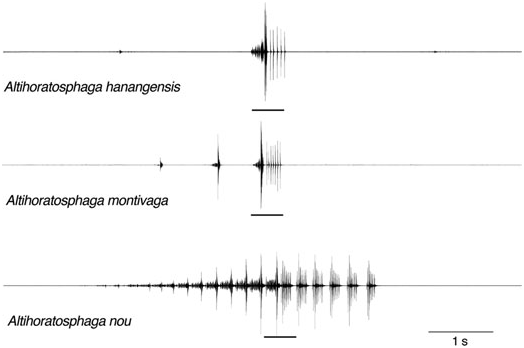
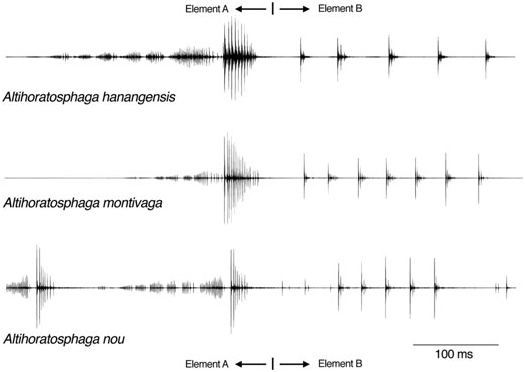
All three recorded species produced songs containing the same two types of elements, A and B ( Figs 3 View Figure 3 , 4 View Figure 4 ). The simplest verse structure is found in A. hanangensis sp. nov., where almost invariably one A element and one B element were combined. These AB verses were repeated at intervals of 12.5 ± 3 s (N = 10; 18 °C, estimate). Only once was an isolated element A recorded. Element A consisted of seven groups of impulses, with the last group being by far the loudest, and element B consisted of five or six single impulses or impulse groups, with the first of each group being by far the loudest ( Fig. 3 View Figure 3 ).
In the acoustic repertoire of A. montivaga , the combination AB is also the most distinctive verse type. However, in our recordings it was never found to be isolated, but was always preceded by several (mostly two) A elements, with increasing amplitude ( Fig. 3 View Figure 3 ). This complete final verse (A + A + AB) is usually preceded by a series of often three, rarely up to 15, softer verses, consisting of A elements only or additionally with an abbreviated B element (one or two impulses only), separated from each other and from the final verse by intervals of around 10–20 s. Rarely, other final structures such as A + AB or A + A + AB + AB were observed (N = 81 final verses in 90-min recording time). The structure of both elements was the same as in A. hanangensis sp. nov., with between six and eight single impulses or impulse groups in element B. Differences in the duration of both parts may be at least partly related to temperature differences.
The song of A. nou was also composed of the two elements A and B. However, both elements were repeated several times to build one verse. On average, a verse of A. nou started with 13 ± 1.4 A elements, ing a similar sound production mechanism, possibly element A of Altihoratosphaga is produced by a fixed pattern of several opening and closing movements using the distal part of the file only, whereas the B elements may result from scraper–file contacts in the proximal part. [In this way complete syllables would never be produced, or would only be produced once per verse; instead, what may be called dista-syllables (different types) and proxi-syllables (one type) are sounded.]
Distribution
All species of Altihoratosphaga are restricted to submontane and montane habitats of Tanzania.
followed by 5 ± 0.9 B elements (range 9–16 A, 3–7 B; N = 58). The B elements mostly consisted of several impulse groups, but in some records they also consisted of single impulses ( Fig. 3 View Figure 3 ). The A elements were repeated at 3.6 Hz (a period of 275 ms), whereas within the B elements the impulse group period was 27 ms, on average.
The frequency spectrum of the song was quite similar in all species examined ( Fig. 5 View Figure 5 ). Its maximum was in the audible range, between about 10 and 15 kHz. All three species typically performed their calling songs after sunset.
Probable mechanism of sound production
As can be recognized from the description (see also Figs 3 View Figure 3 , 4 View Figure 4 ), the song has a fairly complicated structure that cannot easily be related to any pattern of stridulatory movement. However, the stridulatory file and song have some similarity to that of Acrometopa species ( Heller, 1988) . In these species the first part of a verse is produced by stridulatory movements using the distal part of the file only, whereas the end of the verse is produced by contact of the scraper with the large, proximal teeth ( Heller, 1988: fig. 21–2). Assum- Phylogenetic reconstruction
The three different phylogenetic analyses (BI, NJ, and MP) all resulted in the same identical tree topology, shown in Figure 6 View Figure 6 . All three investigated Altihoratosphaga species cluster together, and the genus shows high clade support in all analyses. The genetic distances confirm the distinctiveness of Altihoratosphaga both within the Acrometopini and the subfamily Phaneropterinae ( Table 2). The largest genetic distance (calculated using the GTR + I + G model) within Altihoratosphaga is 0.025, whereas the smallest genetic distance between the genus and all other in-group taxa are 0.289 ( H. tenera ).
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.