Squamapion gardinii, Giusto, 2021
|
publication ID |
https://doi.org/10.11646/zootaxa.4941.2.4 |
|
publication LSID |
lsid:zoobank.org:pub:EF8CA585-BDC0-4D19-A459-84093C05D493 |
|
persistent identifier |
https://treatment.plazi.org/id/974B23EA-E128-4B10-A8C7-E3AA5E9855F1 |
|
taxon LSID |
lsid:zoobank.org:act:974B23EA-E128-4B10-A8C7-E3AA5E9855F1 |
|
treatment provided by |
Plazi |
|
scientific name |
Squamapion gardinii |
| status |
sp. nov. |
Squamapion gardinii n. sp.
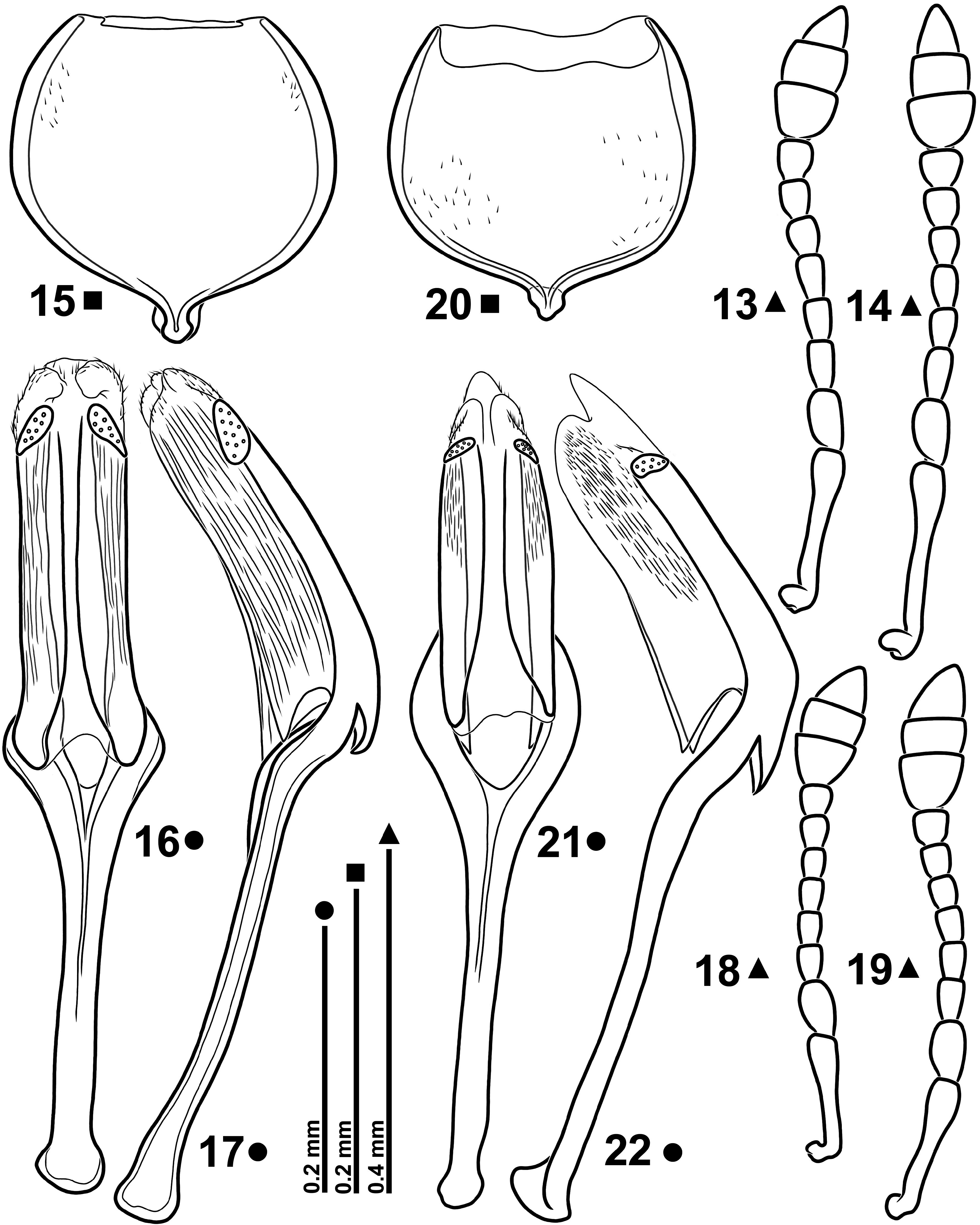
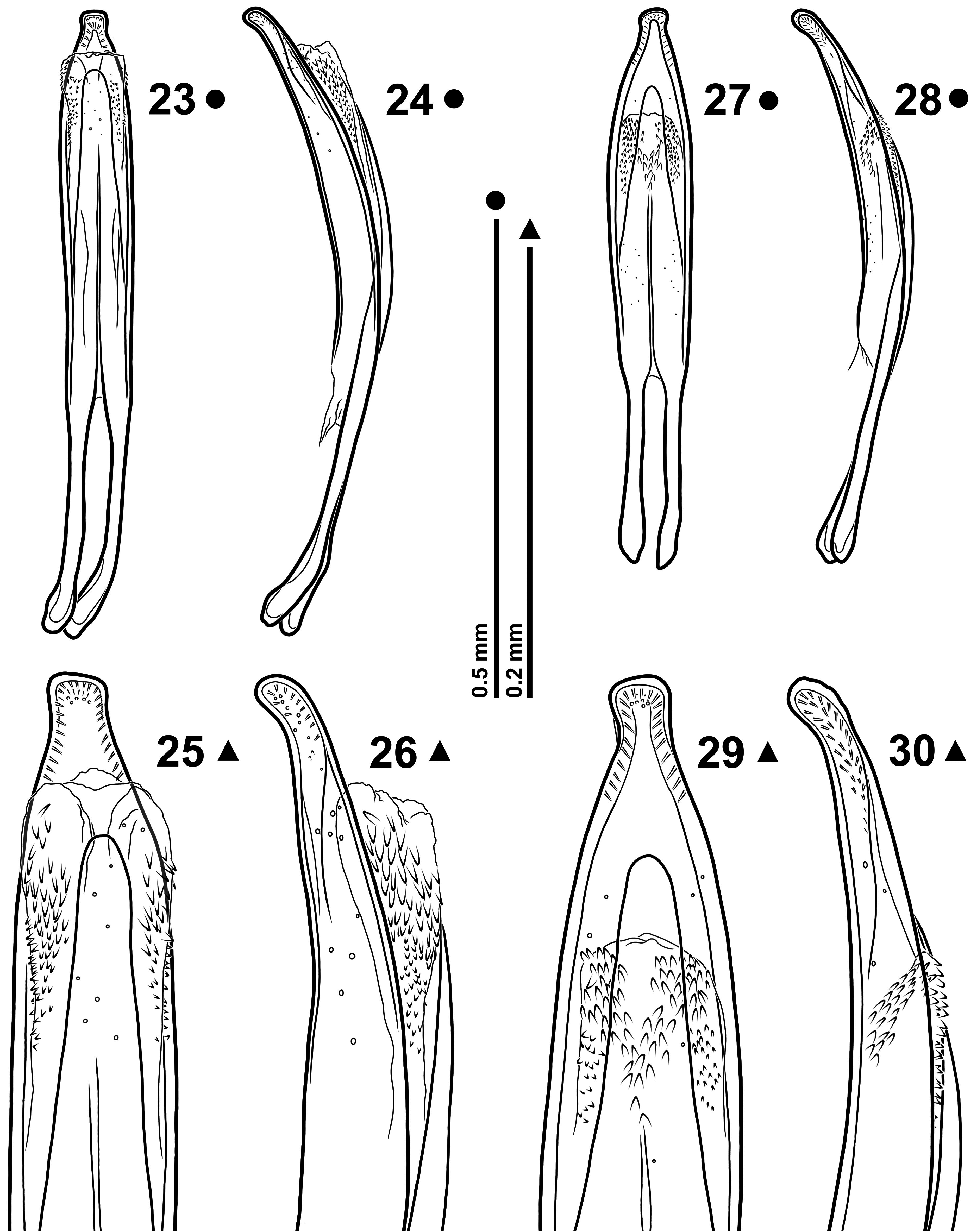
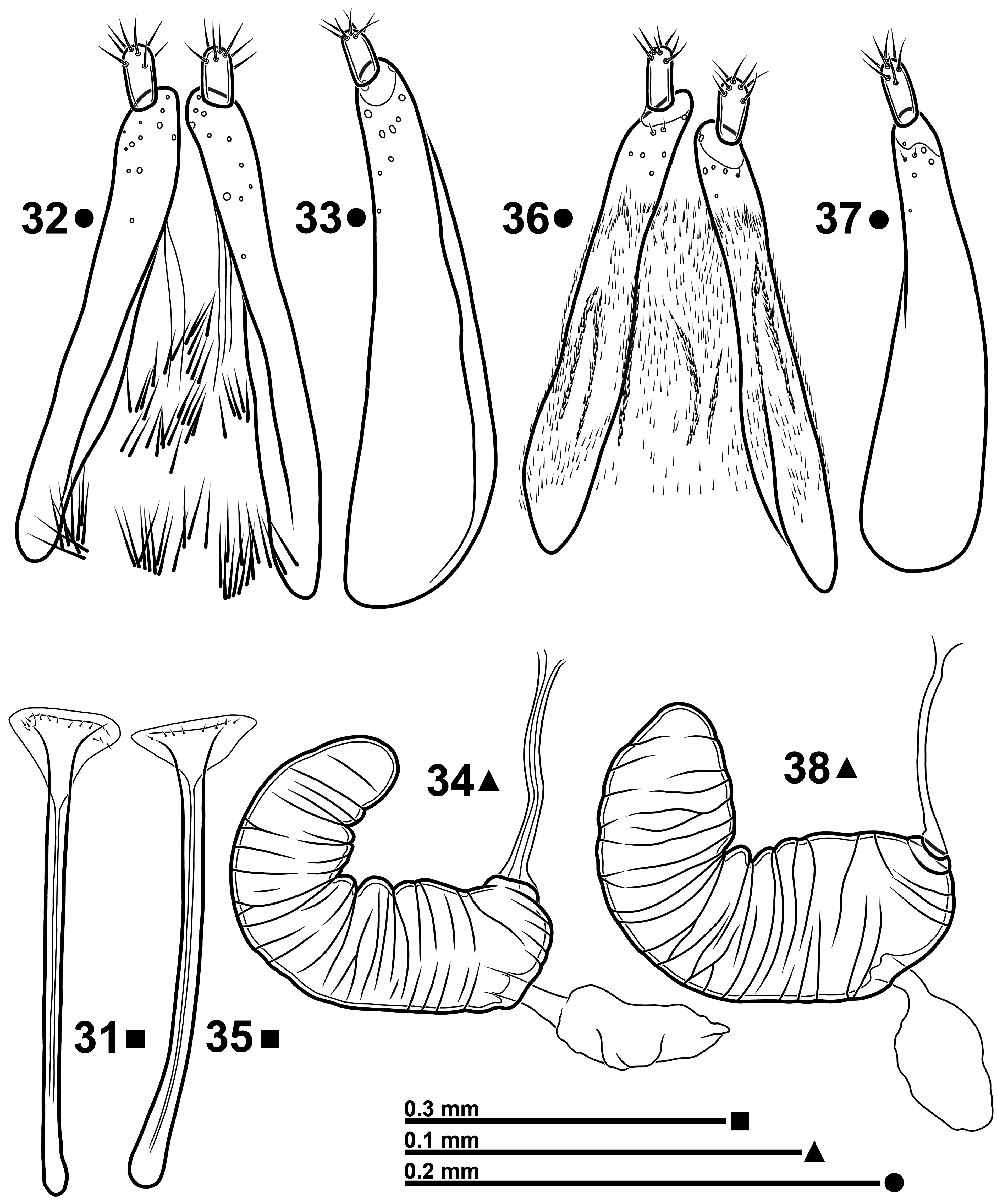
( Figs 1–2 View FIGURES 1–4 , 5–8 View FIGURES 5–12 , 13–17 View FIGURES 13–22 , 23–26 View FIGURES 23–30 , 31–34 View FIGURES 31–38 )
http://zoobank.org/ urn:lsid:zoobank.org:act:
Type locality. Genova-Quinto al Mare: Rio San Pietro (Genoa Province, Liguria, Italy).
Diagnosis. A Squamapion species belonging to the “ S. vicinum group” as defined by Wanat (1997). It differs from all other Palaearctic Squamapion in having the following combination of characters: stocky general appear- ance; legs robust; vestiture dense, especially on sides of body, composed of narrowly lanceolate white scales, on elytral intervals arranged in 2 rows; rostrum long and thin, particularly in female; mesorostral dilatation dentiform in both sexes.
Type material. Holotype (Ƌ): ITALY — Liguria: Genoa Province : “Genova-Quinto al / Mare: Rio San Pietro / 100 m ca. 6.V.2020 / C. Giusto leg.” // “ on Thymus vulgaris L.” ( BMNH).
Paratypes (329 ƋƋ, 410 ♀♀): FRANCE— Provence-Alpes-Côte d’Azur: Alpes-Maritimes Department: Gourdon , env. S, 700 m a.s.l., 21.V.2013, C. Giusto, G. Gardini & S. Zoia leg., on Thymus vulgaris L. ( 1 ♀ CG) ; Tende , Vallon du Réfrei, 30.X.2015, H. Pierotti leg. ( 1 ♀ LD) ;— Bouches-du-Rhône Department: Aix-en-Provence , 3.XI.1968, G. Bartoli leg., on Thymus vulgaris L. ( 4 ♀♀ MSNG) ;— Hautes-Alpes Department: Garde-Colombe, Saint-Genis: Gorges du Riou, 7.VII.2015, R. Casalini leg. (1 Ƌ, 1 ♀ CG; 1 Ƌ, 1 ♀ RC);— Var Department: Callas , env. N, 460 m a.s.l., 23.IV.2011, Ch. Germann leg. ( 1 ♀ CGE) .
ITALY— Abruzzo: Pescara Province: Popoli, Sorgenti del Pescara, 250–300 m a.s.l., 22.VI.2017, C. Giusto & G. Gardini leg., on Thymus vulgaris L. (1 Ƌ CG);— Emilia-Romagna: Piacenza Province: Ottone, Losso, 550 m a.s.l., 21.VI.2020, C. Giusto leg., on Thymus vulgaris L. ( 1 ♀);— Liguria: Genoa Province: Bogliasco, 9.VI.1961, G. Bartoli leg. (7 ƋƋ, 2 ♀♀ MSNG); Bogliasco, Bric Gianesi, slopes S, 600 m a.s.l., 7.I.2018, C. Giusto leg., on Thymus vulgaris L. ( 14 ♀♀ CG); Bogliasco, Monte Santa Croce, slopes S, 500 m a.s.l., 12.I.2020, C. Giusto leg., on Thymus vulgaris L. ( 3 ♀♀ CG); Camogli, Monte di Portofino, slopes W, 11.IV.1968, G. Bartoli leg., on Thymus vulgaris L. ( 1 ♀ MSNG); Camogli, Monte Tocco, 470 m a.s.l., 25.V.2020, C. Giusto leg., on Thymus vulgaris L. (3 ƋƋ CG); Camogli, San Rocco, Mortola, env. E, 350 m a.s.l., 25.V.2020, C. Giusto leg., on Thymus vulgaris L. (4 ƋƋ, 1 ♀ CG); Chiavari, Monte Castello, slopes S, 250 m a.s.l., 27.III.1984, C. Giusto leg., on Thymus vulgaris L. ( 1 ♀ CG); Crocefieschi, 20.IX.1905, A. Dodero leg. ( 2 ♀♀ MSNG); Crocefieschi, 700 m a.s.l., 16.II.2020, C. Giusto leg., on Thymus vulgaris L. ( 10 ♀♀ CG); Davagna, Canate di Marsiglia env., 450 m a.s.l., 25.IV.2016, C. Giusto leg., on Thymus vulgaris L. (25 ƋƋ, 10 ♀♀ CG); Genova, VI.1911, C. Mancini leg. (1 Ƌ, 1 ♀ MSNG); Genova-Apparizione, 250 m a.s.l., 30.V.2020, C. Giusto leg., on Thymus vulgaris L. (3 ƋƋ, 1 ♀ CG); Genova-Ba- vari env., 300 m a.s.l., 31.V.2013, C. Giusto, G. Gardini & S. Zoia leg., on Thymus vulgaris L. (27 ƋƋ, 19 ♀♀ CG); Genova-Bavari, VI.1907, C. Mancini leg. ( 1 ♀ MSNG); Genova-Bavari env., 300 m a.s.l., 7.VI.2017, C. Giusto & G. Gardini leg., on Thymus vulgaris L. (10 ƋƋ, 4 ♀♀ CG); Genova-Bavari env., 250 m a.s.l., 22.V.2020, C. Gi- usto leg., on Thymus vulgaris L. (13 ƋƋ, 10 ♀♀ CG); Genova-Chiappeto, 2.VI.1953, F. Capra leg. ( 1 ♀ MSNG); Genova-Granarolo, 21.VII.1961, G. Bartoli leg. (3 ƋƋ, 5 ♀♀ MSNG); Genova-Granarolo, 4.IV.1967, G. Bartoli leg., on Thymus sp. ( 2 ♀♀ MSNG); Genova-Molassana, 21.V.1944, G. Binaghi leg., on Thymus vulgaris L. (11 ƋƋ, 7 ♀♀ MSNG); Genova-Molassana, 28.III.1976, G. Gardini leg. ( 1 ♀ MSNG); Genova-Molassana, 11.V.1982, C. Giusto leg., on Thymus vulgaris L. (5 ƋƋ CG); Genova-Molassana, 16.VIII.1962, L. Briganti leg. ( 1 ♀ CG); Genova-Molassana, V.1944, A. Baliani leg. (1 Ƌ CG); Genova-Quezzi, 19.XI.1967, G. Bartoli leg., on Thymus vulgaris L. ( 5 ♀♀ MSNG); Genova-Quezzi, 29.V.1963, G. Bartoli leg., on Thymus sp. ( 1 ♀ MSNG); Genova-Quinto al Mare, Rio San Pietro, 200 m a.s.l., 15.III.2020, C. Giusto leg., on Thymus vulgaris L. ( 3 ♀♀ CG); Genova-Quinto al Mare, Rio San Pietro, 70 m a.s.l., 28.IV.2019, C. Giusto leg., on Thymus vulgaris L. (13 ƋƋ, 11 ♀♀ CG); Genova- Quinto al Mare, Rio San Pietro, 100 m a.s.l., 6.V.2020, C. Giusto leg., on Thymus vulgaris L. (3 ƋƋ, 3 ♀♀ BMNH; 39 ƋƋ, 43 ♀♀ CG; 3 ƋƋ, 3 ♀♀ CMNC; 3 ƋƋ, 3 ♀♀ EC; 3 ƋƋ, 3 ♀♀ MNHN; 3 ƋƋ, 3 ♀♀ MSNG; 3 ƋƋ, 3 ♀♀ MSNM; 3 ƋƋ, 3 ♀♀ MW; 3 ƋƋ, 3 ♀♀ NMW; 3 ƋƋ, 3 ♀♀ RC; 3 ƋƋ, 3 ♀♀ SDEI; 3 ƋƋ, 3 ♀♀ ZMHB); Genova-Righi, 30.IV.1976, G. Bartoli leg., on Thymus vulgaris L. (1 Ƌ, 1 ♀ MSNG); Genova-Righi, 5.VI.1980, G. Bartoli leg., on Thymus vulgaris L. ( 1 ♀ MSNG); Genova-San Desiderio, 16.IV.1961, G. Bartoli leg., on Thymus sp. ( 6 ♀♀ MSNG); Genova-San Desiderio, 8.IV.1962, G. Bartoli leg., on Thymus sp. ( 4 ♀♀ MSNG); Genova, Monte Fasce, 830 m a.s.l., 11.VII.2020, C. Giusto leg., on Thymus vulgaris L. ( 1 ♀ CG); Genova, Monte Fasce, slopes S, 700 m a.s.l., 11.VII.2020, C. Giusto leg., on Thymus vulgaris L. (2 ƋƋ, 1 ♀ CG); Genova, Monte Fasce, slopes S, 300 m a.s.l., 26.X.2014, C. Giusto leg., on Thymus vulgaris L. (5 ƋƋ, 20 ♀♀ CG); Genova, Monte Fasce, slopes W, 300 m a.s.l., 18.XI.2017, C. Giusto leg., on Thymus vulgaris L. ( 2 ♀♀ CG); Genova, Monte Fasce, slopes S, 400 m a.s.l., 6.V.2020, C. Giusto leg., on Thymus vulgaris L. (14 ƋƋ CG; 3 ƋƋ, 3 ♀♀ CGE; 3 ƋƋ, 3 ♀♀ FS; 3 ƋƋ, 3 ♀♀ KS; 3 ƋƋ, 3 ♀♀ LD; 3 ƋƋ, 3 ♀♀ MK; 3 ƋƋ, 3 ♀♀ TAUI; 3 ƋƋ, 3 ♀♀ TLFN; 3 ƋƋ, 3 ♀♀ ZMUH); Genova, Monte Fasce, 16.IX.1967, G. Bartoli leg., on Thymus vulgaris L. ( 1 ♀ MSNG); Genova, Monte Fasce, 25.VIII.1884, Caneva leg. ( 1 ♀ MSNG – Apion atomarium , det. J. Desbrochers des Loges); Genova, Monte Gazzo, slopes SE, 200–300 m a.s.l., 1.I.2019, C. Giusto leg., on Thymus vulgaris L. ( 13 ♀♀ CG); Genova, Monte Ratti, slopes S, 500 m a.s.l., 14.I.2018, C. Giusto leg., on Thymus vulgaris L. ( 5 ♀♀ CG); Montoggio, Creto, 11.VII.1961, G. Bartoli leg. (1 Ƌ MSNG); Montoggio, Creto, 4.X.1968, G. Bartoli leg., on Thymus vulgaris L. ( 3 ♀♀ MSNG); Montoggio, Creto, 600 m a.s.l., 16.II.2020, C. Giusto leg., on Thymus vulgaris L. ( 4 ♀♀ CG); Montoggio, Creto, 600 m a.s.l., 28.IX.2014, C. Giusto leg., on Thymus vulgaris L. (1 Ƌ, 2 ♀♀ CG); Montoggio, Creto, env. E, 650 m a.s.l., 22.V.2020, C. Giusto, G. Gardini & P. Gardini leg., on Thymus vulgaris L. (7 ƋƋ, 10 ♀♀ CG); Rapallo, Monte Caravagli, slopes, 400 m a.s.l., 16.I.1993, C. Giusto leg., on Thymus vulgaris L. ( 2 ♀♀ CG); Rapallo, Monte Pegge, slopes S, 700 m a.s.l., 1.I.2020, C. Giusto leg., on Thymus vulgaris L. ( 4 ♀♀ CG); Rapallo, Passo della Crocetta, 550 m a.s.l., 20.VI.1992, C. Giusto leg., on Thymus vulgaris L. (1 Ƌ, 4 ♀♀ CG); Sestri Levante, Trigoso, 75 m a.sl., 1.XI.2020, C. Giusto leg., on Thymus vulgaris L. ( 4 ♀♀ CG); Sori, 9.III.1969, G. Bartoli leg., on Thymus vulgaris L. ( 1 ♀ MSNG); Sori, Canepa env., 350 m a.s.l., 9.VI.1991, C. Giusto leg., on Thymus vulgaris L. (17 ƋƋ, 10 ♀♀ CG); Sori, Canepa env., 350–400 m a.s.l., 31.XII.2016, C. Giusto leg., on Thymus vulgaris L. ( 9 ♀♀ CG); Vobbia, 470 m a.s.l., 16.II.2020, C. Giusto leg., on Thymus vulgaris L. ( 2 ♀♀ CG);— Imperia Province: Ventimiglia, Capo Mortola, 50 m a.s.l., 10.V.2019, C. Giusto leg., on Thymus vulgaris L. (1 Ƌ CG); Ventimiglia, Villatella, 400–500 m a.s.l., 10.V.2019, C. Giusto leg., on Thymus vulgaris L. ( 2 ♀♀ CG);— La Spezia Province: Deiva Marina, Ca’ Marcone env., 475 m a.s.l., 1.XI.2020, C. Giusto leg., on Thymus vulgaris L. (1 Ƌ, 2 ♀♀ CG); Deiva Marina, Passo del Bracco, 7.VI.2006, L. Diotti leg. (1 Ƌ, 3 ♀♀ LD); Portovenere, Monte Muzzerone, slopes NW, 220 m a.s.l., 22.II.2020, C. Giusto leg., on Thymus vulgaris L. ( 5 ♀♀ CG);— Savona Province: Albenga, 25.IV.1901, A. Dodero leg. ( 1 ♀ MSNG); Albenga, Bastia, 31.III.1968, G. Binaghi leg. ( 1 ♀ MSNG); Albissola Marina, 20.X.1926, F. Capra leg. ( 1 ♀ MSNG); Bergeggi, Monte Mao, 400 m a.s.l., 10.IV.2017, C. Giusto, G. Gardini & A. Trotta leg., on Thymus vulgaris L. ( 1 ♀ CG); Boissano env., 150 m a.s.l., 15.V.1994, C. Giusto leg., on Thymus vulgaris L. (12 ƋƋ, 8 ♀♀ CG); Celle Ligure, Bric dei Corvi, 250 m a.s.l., 7.V.1995, C. Giusto leg., on Thymus vulgaris L. (2 ƋƋ CG); Cisano sul Neva, Poggio Croce Ceresa, 700 m a.s.l., 10.VII.2002, C. Giusto leg., on Thymus vulgaris L. (2 ƋƋ CG); Finale Ligure, Finalmarina, 22.V.1899, A. Dodero leg. (14 ƋƋ, 5 ♀♀ MSNG); Noli, Capo Noli, 3.IV.1966, G. Bartoli leg., on Thymus sp. ( 1 ♀ MSNG); Toirano, Salto del Lupo, 180–200 m a.s.l., 15.VI.2016, C. Giusto leg., on Thymus vulgaris L. (6 ƋƋ, 11 ♀♀ CG); Villanova d’Albenga, Coasco, 54 m a.s.l., 21.XI.2015, C. Giusto leg., on Thymus vulgaris L. ( 5 ♀♀ CG);— Piedmont: Alessandria Province: Dernice env., 21.IV.2016, L. Diotti leg. ( 1 ♀ CG); Dernice, Costa di Montebore, 680 m a.s.l., 21.VI.2020, C. Giusto leg., on Thymus vulgaris L. (8 ƋƋ, 8 ♀♀ CG); Dernice, Montebore, 20.I.2018, L. Diotti leg. ( 1 ♀ CG; 1 ♀ LD); Dernice, Montebore, 29.VIII.2014, L. Diotti leg. ( 2 ♀♀ LD); Dernice, Montebore, 28.IX.2016, L. Diotti leg. (2 ƋƋ, 2 ♀♀ LD); Dernice, Montebore, 24.VI.2020, L. Diotti leg. (4 ƋƋ, 8 ♀♀ LD); Dernice, Montebore, 24.VI.2020, M. Tedeschi leg. (3 ƋƋ, 3 ♀♀ MT).
SPAIN— Community of Madrid: Madrid Province: Santa María de la Alameda, La Paradilla , 1,100 m a.s.l., 27.VI.1991, C. Giusto leg., on Thymus vulgaris L. ( 2 ♀♀ CG) .
Etymology. It is a real pleasure and an honor for me to dedicate the new taxon to Giulio Gardini, pseudoscorpion specialist, friend, mentor and research partner in my first 40 years of entomology.
Description (Ƌ ♀). Body stocky; Lb: Ƌ 1.20–1.77 mm [ 1.64 mm] ( 1.67 mm), ♀ 1.30–1.79 mm [ 1.60 mm]. Body integument black; femora black; tibiae and tarsi dark brown to nearly black; in male, antennae testaceous with darker club and sometimes with darkened distal funicular segments; in female, antennae dark brown to nearly black. Vestiture dense, composed of white scales, rarely white-greyish; hair-like on prorostrum and ventrite 5; narrowly lanceolate on metarostrum, pronotal disc, elytra and on body venter; lanceolate, with truncate apex, around eyes, on body sides and on legs; on elytral intervals, scales, only just longer than width of intervals, arranged in two quite irregular rows, partially overlapping; at base of intervals 2–4, scales more numerous forming short multiple rows; at base of interval 3, scales, with truncate apex, arranged in a more or less condensed patch; interstrial scales similar to those on intervals, barely separated from each other ( Figs 1–2 View FIGURES 1–4 ).
Rostrum rather long; in dorsal view, obtusely dentiform at antennal insertion, narrowed from mesorostrum to the half of prorostrum, then cylindrical up to apex; in lateral view, sometimes feebly bent at antennal insertion and with prorostrum regularly curved, less in male, more in female, narrowing at apex in male or equally high on whole length in female ( Figs 5–8 View FIGURES 5–12 ); rostral surface shagreened, almost matt in male, weakly shining in female; shallowly punctate; in male, up to half prorostrum with sparse, whitish, hair-like scales and then with microscopic scales up to apex; in female almost whole prorostrum clothed with microscopic and sparse scales. Head transverse; eyes, in male, moderately large and regularly convex, in female distinctly smaller; frons flat, strongly punctate and microsculptured with almost indistinct median fovea in some specimens. Antennae ( Figs 13–14 View FIGURES 13–22 ) inserted at basal 0.13–0.19 of rostrum, slender and with white and weakly erect setae.
Prothorax ( Figs 1–2 View FIGURES 1–4 ) in dorsal view transverse, campanulate, moderately rounded at sides, widest at base with prominent hind angles; disc weakly convex, densely punctate; punctures round, 16–22 µm in diameter, separated by less than one third of their diameter; interspaces strongly microreticulate; prescutellar fovea as broad as discal punctures or weakly broader, not longer than three punctures. Scutellum triangular with curved sides, weakly de- pressed on disc and with two rudimentary keels at base, glabrous, microsculptured. Elytra ( Figs 1–2 View FIGURES 1–4 ) in dorsal view subrectangular, with slightly curved sides in male, more rounded in female; intervals 1.4–1.8 times as wide as striae, flat, rugosely microsculptured and with two rows of shallow punctures; striae sharply margined with septa between punctures distinctly deepened. Legs robust; in male, hind femora thickened; tibiae straight, relatively short, strongly widened from base to apex; protarsi robust; 1 st segment 1.27–1.50 times as long as wide; 2 nd 1.00–1.21 times as long as wide, 0.80–0.87 times as long as 1 st; 3 rd 0.72–0.86 times as long as wide, 0.84–0.93 times as long as 2 nd, with narrow lobes; onychium robust, 1.09–1.33 times as long as 2 nd.
Sternite IX as in Fig. 15 View FIGURES 13–22 . Tegmen ( Figs 16–17 View FIGURES 13–22 ); phallobase Y-shaped with tegminal apodeme slightly longer than forked basal piece; parameral plate with median carinae moderately separated, vanishing just behind the supra- fenestral sclerites; each suprafenestral sclerite bears 7–10 pores; apical membranous lobes well developed, united, margined with microchaetae; whole folded margins of parameral plate with very fine and dense longitudinal cari- nae. Penis ( Figs 23–26 View FIGURES 23–30 ) in dorsal view more or less slender, variably narrowed in distal half and ending in a slightly asymmetrical blunt tip; endophallus with a more or less defined M-shaped aggregation of spines in orificial region; basal spines small, distal ones bigger. Female sternite VIII as in Fig. 31 View FIGURES 31–38 . Spermatheca ( Fig. 32 View FIGURES 31–38 ) C-shaped with cornu more or less recurved; spermathecal gland inserted on a just sketched ramus; nodulus slightly protruding. Ovipositor as in Fig. 33 View FIGURES 31–38 ; foretube densely clothed with long microtrichia; gonocoxites ( Figs 33–34 View FIGURES 31–38 ) about 4 times as long as wide; styli inserted latero-apically, cylindrical, 2.25–2.42 times as long as wide, with 7–8 apical setae.
Distribution. Spain, France, Italy.
Host plant. Thymus vulgaris L. ( Figs 40–42 View FIGURES 39–45 ).
Bionomy. Squamapion gardinii prefers the garrigue and the arid meadows of the Mediterranean calcareous or siliceous/calcareous slopes, approximately from sea level ( Italy, Ventimiglia, Capo Mortola, 50 m a.s.l.) up to 1,100 m a.s.l. ( Spain, Santa María de la Alameda, La Paradilla); it is particularly abundant between 100 and 500 m above sea level ( Fig. 39 View FIGURES 39–45 ).
Plants that host S. gardinii show little damage caused by the adults: small damage is visible on the leaves while it is possible to observe the galls—inside which the insects develop—on the stems ( Figs 41–45 View FIGURES 39–45 ). No galls were found on roots.
Adults are present on the host plant all year round, but they are particularly numerous from April to June and again quite frequent from October to December.
Remarks. Some authors had already noted in the past that the southernmost populations of S. atomarium were a little different from those of central Europe. Indeed, it is worth remembering what was written by Sainte-Claire Deville (1924): “J’ai capturé çà et lâ l’A. atomarium dans des montagnes sèches du Midi de la France où le Thymus serpyllum n’existait pas et où par contre Ie T. vulgaris était abondant; les individus de cette provenance sont plus robustes et leur pubescence est plus apparente”, observations also quoted later by Hoffmann (1929, 1958). Just two years before, Schatzmayr (1922) had already observed some differences within different populations of S. atomarium underlining the extraordinary body length of the specimens from Montoggio ( Liguria, Italy), ”sino a 2,4 mm”.
At first glance, thanks to its dense vestiture, S. gardinii appears whitish whilst, probably due to its stocky body, it seems much larger than it really is. Measurements specified in Table 1, in fact, show that S. gardinii on average is just a little bit larger than S. atomarium and that dimensions of the two species are rather similar (cfr. Figs 1–2 View FIGURES 1–4 with Figs 3–4 View FIGURES 1–4 ). The analysis of rostral morphology reveals the most useful characters to discriminate S. gardinii from S atomarium : both sexes of the new species show—on average—a longer and thinner rostrum and a dentiform mesorostral dilatation, whereas the rostrum of S. atomarium is thicker and the mesorostral dilatation is dentiform in males and obtusely rounded in females (cfr. Figs 5–8 View FIGURES 5–12 with Figs 9–12 View FIGURES 5–12 ).
Among the other species of Squamapion , only Squamapion solariorum (Wagner, 1908) n. stat. (see below), Squamapion bifarium (Balfour-Browne, 1944) , Squamapion kandarense ( Hustache, 1946) and Squamapion terraesanctae Friedman & Freidberg, 2007 share with S. gardinii the long and thin rostrum. Despite the small number of specimens examined of some of these species (see Appendix 1) and the fact that their measurements and their ratios overlap abundantly, it is easy to observe that, regardless of the curvature of rostrum—more recurved in S. solariorum , S. bifarium and S. kandarense , less in S. terraesanctae —all their female mesorostra are obtusely rounded in dorsal view. Moreover, their vestiture differs from that of S. gardinii being composed of piliform scales ordered in a single row on elytral intervals, sometimes forming 2–3 short confused rows at base of intervals 2–4, and often arranged in a poorly condensed patch at base of interval 3. The body outlines of all of them are stout, but less than in S. gardinii ; their humeral calli are more prominent, their pronotal surfaces are slightly less densely punctate and the interspaces are microreticulate and weakly shiny.
A further, although partial, aid for recognizing these species comes from the knowledge of their host plants: S. atomarium is oligophagous on some Thymus species, mainly on taxa belonging to “ Thymus serpyllum L.” and “ Thymus pulegioides L.” groups; also S. gardinii is associated to Thymus , but it is monophagous on Thymus vulgaris L. Squamapion terraesanctae lives on Satureja thymbra L. and Origanum syriacum L. Raf. whilst Mentha longifolia typhoides (Briq.) Briq. (= Mentha longifolia cyprica (Heinr. Braun) Harley ) is doubtfully quoted as the host plant of S. bifarium by Friedman & Freidberg (2007). Unfortunately, no data about host plants of S. solariorum and S. kandarense are available, even though their association with Thymus is predictable.
The ecology and the phenology of S. gardinii and S. atomarium are also different: the latter species inhabits arid meadows on hills and mountains with calcareous or siliceous soils from 250 to 2,450 m a.s.l. being particularly common from 500 to 1,500 m a.s.l. with adults on the host plants from April to December, but the peak attendance is recorded between June and September (especially in September).
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.