Vaejovis miscionei Myers & Ayrey, 2021
|
publication ID |
https://doi.org/10.5281/zenodo.7162607 |
|
publication LSID |
lsid:zoobank.org:pub:43A2A40F-53F9-43E9-8571-E7643B6AD7CF |
|
persistent identifier |
https://treatment.plazi.org/id/A03E87B1-AA27-FFFF-F217-3F4FFB32F887 |
|
treatment provided by |
Felipe |
|
scientific name |
Vaejovis miscionei Myers & Ayrey |
| status |
sp. nov. |
Vaejovis miscionei Myers & Ayrey View in CoL , sp. n.
( Figures 1–15 View Figures 1–4 View Figure 5 View Figure 6 View Figures 7–12 View Figures 13–14 View Figure 15 ; Tables 1–2) http: //zoobank. org/urn: lsid: zoobank. org: act: 966D5326-
857B-4D02-B3C3-6743662FC692
TYPE LOCALITY AND TYPE REPOSITORY. USA, Arizona, Cochise County, Mule Mountains , 31.45870°N 109.94304°W, 1834 m a. s. l. GoogleMaps ; USNM.
TYPE MATERIAL. USA, Arizona, Cochise County, Mule Mountains , 31.45870°N 109.94304°W, 1834 m a. s. l., leg GoogleMaps . R. F. Ayrey, 4 September 2018, 1♀ ( holotype, #RA2079) , USNM, 1♂ ( paratype, #RA2076), 7 October 2018, 1♂ ( paratype, #RA2081), 1 April 2019, 2♀ ( paratypes, #RA2071, #RA2074), RFA .
The type specimens were found with a blacklight at night. The vegetation type is mesic, mixed evergreen oak woodland ( Figs. 16–17 View Figures 16–17 ). Centruroides sculpturatus was found syntopically with V. miscionei sp. n. during 4 field trips to the Mule Mountains above Bisbee, Arizona.
ETYMOLOGY. The species is named in honor of Tom Miscione who informed the authors of the locality.
DIAGNOSIS. Small (ave. 23.79 mm) scorpions. Color is dark brown, lighter on the legs, with underlying mottling on carapace and mesosoma (see Figure 1 View Figures 1–4 ). Pedipalp movable finger with 6 ID denticles and fixed finger with 5. Carapace of female is longer than the fifth metasomal segment. Pectinal tooth count for females 11.0 [n=6], males 12.0 [n=4]. Small, nearly obsolete subaculear tubercle.
DESCRIPTION. Based on holotype female, unless otherwise noted, see Figure 3 View Figures 1–4 for dorsal and ventral views.
Color ( Fig. 3 View Figures 1–4 ). Color is dark brown, lighter on the legs, telson red. Faint underlying mottling on the carapace and mesosoma. Carapace ( Fig. 5 View Figure 5 ). Anterior margin of carapace moderately emarginated, posterior margin slightly emarginated. Carapace moderately granular with fine granules interspersed. Three lateral eyes on each side, with two large median eyes positioned on anterior 1/3. Median furrow moderate and traverses entire length of carapace. Eight setae present on anterior portion of carapace. One pair of setae present along lateral edge of carapace directly behind lateral eyes. One pair of setae found in front of median eyes, and one pair behind. One pair of setae positioned roughly 10% from posterior edge. Ratio of median eyes location from anterior edge/carapace length 0.35; carapace length/width at median eyes 1.36. Carapace of female is longer than metasomal segment V.
Mesosoma ( Figs. 3 View Figures 1–4 , 5 View Figure 5 ). Tergites finely granular with coarse granules throughout. One pair of setae situated approximately 10% from posterior edge of each tergite. Tergite VII finely granular with large granules throughout, with strong dorsal lateral and lateral supramedian granular carinae and with large granules on posterior edge. Sternites III-VI finely granular and without carinae. Sternite VII with granular ventral lateral carinae on posterior half. Presternites smooth. Spiracles ovoid with median side rotated 35 degrees from posterior sternite margin. Sternites with variable number of microsetae.
Sternum ( Fig. 5 View Figure 5 ). Sternum is type 2.
Genital Operculum ( Fig. 5 View Figure 5 ). Sclerites separated on posterior one-fifth.
Pectines ( Fig. 5 View Figure 5 ). Pectinal tooth counts 11/11 [n=3] with a mean of 11.0 [n=6] for females and 12/12 [2] with a mean of 12.00 [n=4] for males. All pectinal teeth have exterodistal angling with large sensorial area. Middle lamellae 7/7. Fulcra are present. Each fulcrum with 5-10 central setae.
Metasoma ( Fig. 13 View Figures 13–14 ). Carapace of female is longer than the metasomal segment V. Ratio of segment I length/width 0.81; of segment II length/width 1.07; of segment III length/width 1.18; of segment IV length/width 1.69; of segment V length/ width 2.25. Segments I-IV: dorsolateral carinae strong and granular with distal denticle of I-IV enlarged and spinoid. Lateral supramedian carinae I-IV strong and granular with enlarged spinoid distal denticle. Lateral inframedian carinae moderately granular on segment I, posterior 3/5 of II, 4/5 of III, and weak on 2/5 of IV. Ventrolateral carinae I weak and granular; on II-III moderate, granular; on IV strong, granular. Ventral submedian carinae moderate to strong on segments I-IV. Dorsal and lateral intercarinal spaces very finely granular. Segment I-IV ventral submedian setae 3/3. Segment V: Dorsolateral carinae moderate, distally crenulate, basally granular. Lateromedian carinae weak and granular on basal 3/5, obsolete on distal 2/5. Ventrolateral and ventromedian carinae strong. Intercarinal spaces finely granular. Segment V ventrolateral setae 4/4.
Telson. Smooth with 4 pairs of large setae on the ventral surface, 3 large setae along both lateral edges of the vesicle and numerous smaller setae. Small, poorly-developed subaculear tubercle present. Lateral aculear serrations present in all specimens studied. Female teeth number 5-7 (n=3), male paratypes with 5-6 teeth (n=2).
Chelicerae. Typical for genus. Dorsal edge of movable cheliceral finger with two subdistal ( sd) denticles. Ventral edge is smooth, with well-developed serrula on distal half. Fixed cheliceral finger with four denticles: basal, median, subdistal and distal. Basal and median denticles forked.
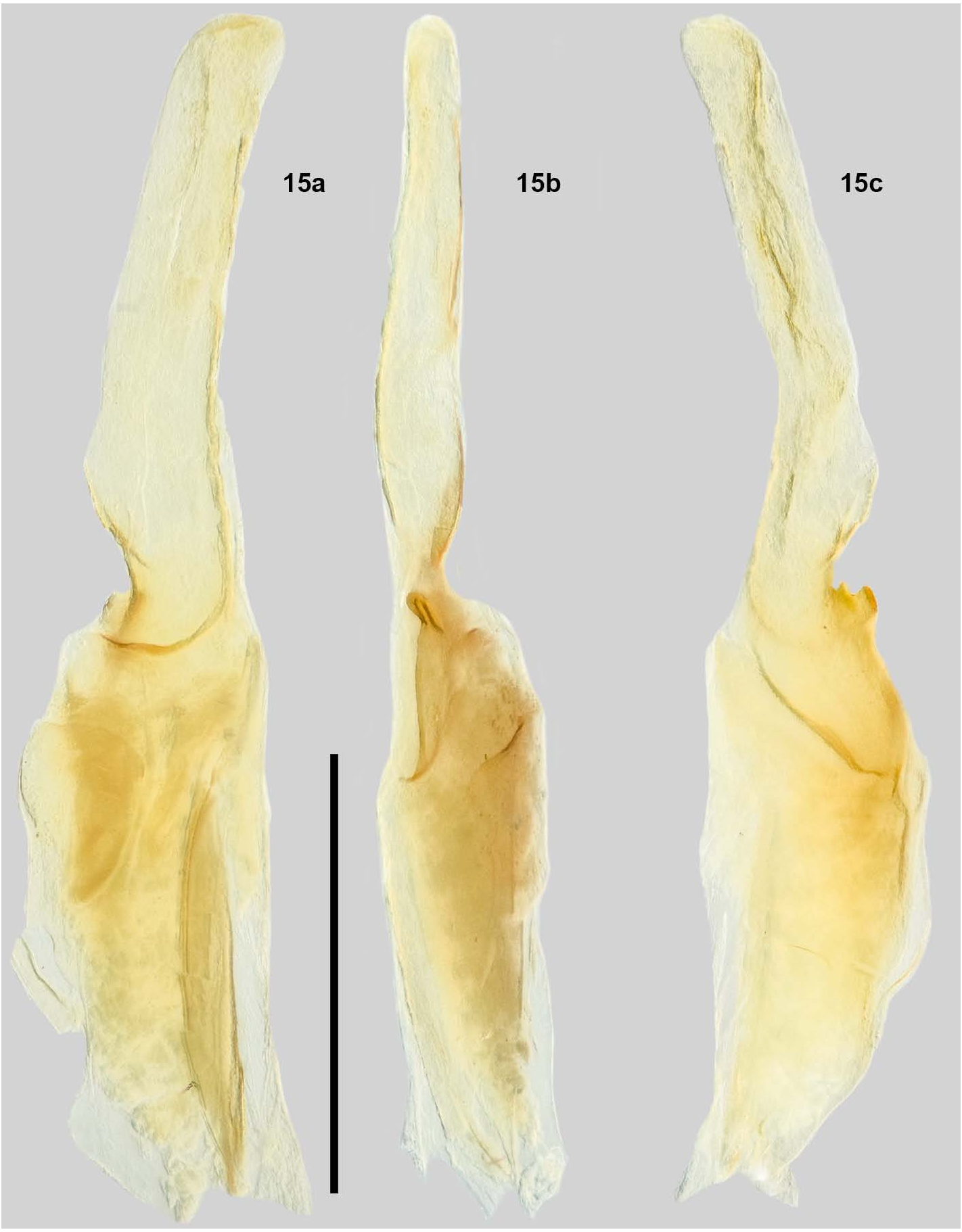
Pedipalps. Trichobothrial pattern type C (Vachon, 1974). Trichobothria ib/it at base of fixed finger. Pedipalp ratios: chela length/width 3.89; femur length/width 3.19; patella length/width 3.22; fixed finger length/carapace length 0.64. Fixed finger median (MD) denticles aligned and divided into 6 subrows by 5 outer (OD) denticles and 5 ID denticles. Mating Plug: Both left and right hemispermatophores were Movable finger with 6 subrows, 5 OD denticles and 6 ID dissected from two different males. After studying four denticles (Soleglad & Sissom, 2001). Chela and femur with total hemispermatophores, no mating plug could be found carinae moderate. Patella with carinae strong, internal surface embedded. While we assume the mating plug morphology is with very large granules on the DPSc carina. similar to the other species of “vorhiesi” group, we cannot be Legs. Ventral surface of tarsomere II with single median row sure until a successful dissection has been made.
of spinules terminating distally with one spinule pair.
REPRODUCTION. Several adult females were kept alive to Variability. Unlike in all other Vaejovis “vorhiesi” group determine 1st instar behavior. Of those, 6 gave birth with species examined, no variability of male or female pectinal the average number of 1st instars being 17.0 (6), standard tooth counts was found. All females had 11/11 and all males deviation 2.09762. The 1st instar behavior was as previously were 12/12. See Pectines section. described ( Hjelle, 1974; Ayrey, 2012, 2013; Ayrey & Myers, Hemispermatophore ( Fig. 15 View Figure 15 ). Wide hemispermatophore 2019). The birthing process was captured on camera. In Figure trunk with a well-defined truncal flexure; the dorsal trough 18, several first instar juveniles can be seen in the typical is shallow, with its base terminating at the distal end of the “birthing basket” and several others can be seen in the process truncal flexure and tapers posteriorly; the lamellar hook is of climbing up from the ventral surface of the female, behind relatively large and strongly bifurcated at the distal tip, and the pedipalps to her dorsal surface.
also possesses a strong groove and slight basal constriction.
Measurements (mm) are as follows based on right COMMENTS. In their description of Vaejovis islaserrano , hemispermatophore (see Soleglad & Fet, 2008: 30, fig. 40): Barrales-Alcalá et al. (2018) made note of a “glandular area Total length, 2.84; lamina length, 1.78; trunk width, 0.44; lamina on dorsal face present on distal third, and longer than wide”. width, 0.27; lamellar hook length, 0.42; trough difference, 0.25. This visible dorsal gland is evident in the male specimens of Morphometric ratios: trough difference/lamellar hook length, other species of scorpions from multiple families (Williams, 0.60; lamellar hook length/lamina length, 0.24. 1970a, 1970b; González-Santillán & Prendini 2013; Teruel et visible difference when comparing the closest related species. AFFINITIES. Comparisons are made to all species of the Vaejovis V. deboerae , V. grahami , V. troupi and V. vorhiesi all have “vorhiesi” group, with emphasis on V. deboerae , V. grahami , chela that is more than four times longer than wide. The chela V. tenuipalpus , V. troupi and V. vorhiesi due to recent DNA in V. miscionei sp. n. is more stout, with the ratio of length to work (Bryson et al., 2013). The type locality for V. miscionei width being 3.52-3.89. sp. n. is the same locality as the Mule Mountains population discussed in Bryson et al. (2013) (see their figs. 2-4). In that DISTRIBUTION. Known only from the type locality, Mule study (figs. 3-4), the authors outlined six different geographical Mountains above Bisbee, Cochise County, Arizona, USA. clades. V. miscionei sp. n. and one other undescribed species from the Dragoon Mountains form Clade 2, which is separate Acknowledgments from all the species discussed below. Vaejovis bigelowi , V. crumpi , V. elii , V. grayae , V. jonesi , The second author would like to thank his wife Melinda V. lapidicola , V. paysonensis , and V. trinityae : all exhibit seven DeBoer-Ayrey for joining him on four field trips to the Mule inner denticles ( ID) on the chelal movable finger while V. Mountains. We would also like to thank Eric Stetson for his miscionei sp. n. exhibits 6. All seven species are also widely assistance on one field trip and two anonymous reviewers for allopatric with V. miscionei sp. n. their comments. Vaejovis bandido , V. cashi , V. electrum , V. feti , V. halli , V. islaserrano , V. patagonia , and V. stetsoni are all widely allopatric with V. miscionei sp. n. Each of these species has 6 References inner denticles on the fixed finger and 5 inner denticles on the moveable finger, like most of the more southern species. AYREY, R. F. 2009. Sky island Vaejovis : A new species Vaejovis tenuipalpus has 6 ID denticles on both the fixed ( Scorpiones : Vaejovidae ). Euscorpius , 86: 1–12. and movable fingers while V. miscionei sp. n. has 5 ID denticles on the fixed finger. V. tenuipalpus is also widely allopatric with AYREY, R. F. 2012. A new Vaejovis from the Mogollon V. miscionei sp. n., separated by more than 500 km. Highlands of northern Arizona ( Scorpiones : Vaejovidae ). Vaejovis deboerae , V. grahami and V. troupi exhibit Euscorpius , 148: 1–13. 8 morphometric ratios, which do not overlap with V. miscionei sp. n. AYREY, R. F. 2013. A new species of Vaejovis from the Vaejovis vorhiesi exhibits 6 morphometric ratios which Mogollon Rim of northern Arizona ( Scorpiones : do not overlap with V. miscionei sp. n. Vaejovidae ). Euscorpius , 166: 1–15. AYREY, R. F. 2018. A new species of Vaejovis from the Patagonia Mountains , southern Arizona ( Scorpiones : Vaejovidae ). Euscorpius , 183: 1–13.
AYREY, R. F. & B. MYERS. 2019. A new “ vorhiesi ” group species of the genus Vaejovis from the Galiuro Mountains, southern Arizona ( Scorpiones : Vaejovidae ). Euscorpius , 284: 1–14.
AYREY, R. F. & M. E. SOLEGLAD. 2011. A new species of Vaejovis from Prescott, Arizona ( Scorpiones : Vaejovidae ). Euscorpius , 114: 1–15.
BARRALES-ALCALÁ, D. A., O. F. FRANCKE, T. R. VAN DEVENDER & G. A. CONTRERAS-FÉLIX. 2018. A new Sky Island species of Vaejovis C. L. Koch, 1836 from Sonora, Mexico ( Scorpiones , Vaejovidae ). ZooKeys, 760: 37–53.
BRYSON, R. W. JR., B. R. RIDDLE, M. R. GRAHAM, B. T. SMITH & L. PRENDINI. 2013. As old as the hills: montane scorpions in Southwestern North America reveal ancient associations between biotic diversification and landscape history. PLoS ONE, 8: 1–11.
CONTRERAS-FÉLIX,G. & O. F. FRANKE. 2019.Taxonomic revision of the “mexicanus” group of the genus Vaejovis C. L. Koch, 1836 ( Scorpiones : Vaejovidae ). Zootaxa, 4596(1):1–100.
GONZÁLEZ-SANTILLÁN & PRENDINI, L. 2013. Redefinition and generic revision of the North American vaejovid scorpion subfamily Syntropinae Kraepelin, 1905 , with descriptions of six new genera. Bulletin of The American Museum of Natural History, 382:1-71.
GRAHAM, M. R. 2007. Sky island Vaejovis: two new species and a redescription of V. vorhiesi Stahnke (Scorpiones: Vaejovidae). Euscorpius, 51: 1–14.
HJELLE, J. T. 1974. Observations on the birth and post-birth behavior of Syntropis macrura Kraepelin (Scorpiones: Vaejovidae). Journal of Arachnology, 1: 221–227.
LOWE, G. 2018. The genera Butheolus Simon, 1882 and Xenobuthus gen. nov. (Scorpiones: Buthidae) in Oman. Euscorpius, 261: 1–73.
LOWE, G., E. A. YAĞMUR & F. KOVAŘÍK. 2014. A review of the genus Leiurus Ehrenberg, 1828 (Scorpiones: Buthidae) with description of four new species from the Arabian Peninsula. Euscorpius, 191: 1–129.
PRENDINI, L. 2003. Discovery of the male of Parabuthus muelleri, and implications for the phylogeny of Parabuthus (Scorpiones: Buthidae). American Museum Novitates, 3408: 1–24.
SOLEGLAD, M. E. & V. FET. 2008. Contributions to scorpion systematics. III. Subfamilies Smeringurinae and Syntropinae (Scorpiones: Vaejovidae). Euscorpius, 71: 1–115.
SOLEGLAD, M. E. & W. D. SISSOM, 2001. Phylogeny of the family Euscorpiidae Laurie, 1896: a major revision. Pp. 25–111 in Fet, V. & P.A. Selden (eds). Scorpions 2001. In memoriam Gary A. Polis. Burnham Beeches, Bucks: British Arachnological Society.
STAHNKE, H. L. 1971. Scorpion nomenclature and mensuration. Entomological News, 81: 297–316.
TERUEL, R., G. LOWE, V. FET & K. A. DANIEL. 2015. Occurrence of a telson gland in the genus Superstitionia Stahnke, 1940 (Scorpiones: Superstitioniidae). Euscorpius, 204: 1−5.
WARSHALL, P. 1995. The Madrean sky-island archipelago. Pp. 6-18 in: Biodiversity and Management of the Madrean Archipelago: the Sky-islands of Southwestern United States and Northwestern Mexico. (LF DeBano, PF Folliott, A. Ortega-Rubio, GJ Gottfried, RH Hamre & CB Edminster, eds.). Fort Collins, Colorado: USDA Forest Service Rocky Mountain Forest and Range Experiment Station.
WILLIAMS, S. C. 1970 a. A systematic revision of the giant hairy scorpion genus Hadrurus. Occasional Papers of the California Academy of Sciences, 87: 1–62.
WILLIAMS, S. C. 1970 b. A redescription of Hadrurus pinteri Stahnke based on the adult (Scorpionida: Vejovidae). The Wasmann Journal of Biology, 28 ( 1): 169–174.
VACHON, M. 1974. Etude des caractères utilizés pour classer les familles et les genres de Scorpions (Arachnides). 1. La trichobothriotaxie enArachnologie.Sigles trichobothriaux et types de trichobothriotaxie chez les Scorpions. Bulletin du Museum National d’Historie Naturelle, Paris, (Ser. 3), 140 (Zool. 104): 857–958.
VOLSCHENK, E. S. 2005. A new technique for examining surface morphosculpture of scorpions. The Journal of Arachnology, 33: 820–825.
| USNM |
Smithsonian Institution, National Museum of Natural History |
| R |
Departamento de Geologia, Universidad de Chile |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |