Chelidoperca barazeri Lee, Lee, Matsunuma & Chen, 2019
|
publication ID |
https://doi.org/ 10.11646/zootaxa.5189.1.19 |
|
publication LSID |
lsid:zoobank.org:pub:DDDB8C91-9AD8-47F9-A88E-331E510DA29B |
|
DOI |
https://doi.org/10.5281/zenodo.7125677 |
|
persistent identifier |
https://treatment.plazi.org/id/03DCE14D-FF9A-FFC5-E994-46CEFD7BFE49 |
|
treatment provided by |
Plazi |
|
scientific name |
Chelidoperca barazeri Lee, Lee, Matsunuma & Chen, 2019 |
| status |
|
Chelidoperca barazeri Lee, Lee, Matsunuma & Chen, 2019
Figures 1‒3 View FIGURE 1 View FIGURE 2 View FIGURE 3 , 5‒6 View FIGURE 5 View FIGURE 6 ; Tables 1‒3
Chelidoperca barazeri Lee, Lee, Matsunuma & Chen, 2019 : fig. 7; supplementary figs. 1A, 2A‒B, 3A (type locality: Gazelle Channel, New Ireland, Bismarck Sea ).
Chelidoperca tosaensis not of Matsunuma et al., 2018: Koeda & Ho, 2019: 668, figs.1‒2 (Ke-tzu-liao, southwestern Taiwan, misidentification).
Type series. NTUM15639 , holotype, 32.2 mm SL ; NTUM15635 , paratype, 32.0 mm SL, sta. CP4262, 2˚4’S, 151˚07’E, 150– 160m. Gazelle Channel, New Ireland, Bismarck Sea, R / V Alis , French beam trawl, MADEEP expedition, 25 Apr. 2014. Non-type specimens. All from southwestern Taiwan : KAUM –I. 115150, 65.2 mm SL, 115151, 45.6 mm SL, Ke-tzu-liao, Kaoshiung , southwestern Taiwan, coll. by K. Koeda et al., bottom trawl, 8 May 2018; KUN-P 55609, 57.8 mm SL, 55610, 40.9 mm SL, 55611, 59.0 mm SL, off Kaoshiung, southwestern Taiwan, coll. by H.-C. Ho, bottom trawl, 16 Sept. 2019 ; NMMB-P35231 (CO1: ON797433 View Materials ), 53.1 mm SL, 35232 (CO1: ON797434 View Materials ), 50.8 mm SL, Ke-tzu-liao, Kaoshiung , southwestern Taiwan, ca. 100m, bottom-trawl, 24 Jan. 2021, coll. by C.-N. Tang ; NMMB-P35233 (CO1: ON797435 View Materials ), 67.7 mm SL, 35234 (CO1: ON797436 View Materials ), 66.1 mm SL, 35235 (CO1: ON797437 View Materials ), 64.4 mm SL, 35236 (CO1: ON797438 View Materials ), 59.6 mm SL, 35237 (CO1: ON797439 View Materials ), 54.1 mm SL; Ke-tzu-liao, Kaoshiung , southwestern Taiwan, ca. 100m, bottom-trawl, 31 Jan. 2021, coll. by C.-N. Tang .
Diagnosis. A species of Chelidoperca distinguished from congeners by the following combination of characters: three scale rows between lateral line and base of 6th dorsal-fin spine (two full-sized plus a dorsalmost half-sized scales); pored lateral-line scales 34‒37 (modally 36); scale rows in longitudinal series 35–39 (modally 38); developed gill rakers 1‒2+7‒8 (modally 2+7); cheek scale rows 4–7 (modally 5); dentary surface of lower jaw naked; interorbital region with a single row of cycloid scales extend to or slightly beyond the mid-orbit level; outermost row of teeth of the upper jaw enlarged, antrorse and caniniform; outermost row of teeth of the lower jaw at the lateral side near symphysis with ca. 3‒4 enlarged antrorse canines, innermost row of teeth enlarged and caniniform; penultimate and the last dorsal- and anal-fin rays in adult not elongated; a red ocellus on opercular membrane between the spines; soft dorsal fin with series of large yellow spots; anal fin with a yellow margin; caudal fin with yellow spots along the basal-half median fin rays; and the presence of a faint and discontinuous stripe formed by clusters of melanophores along the mid-lateral body when preserved.
Description. The following values are provided for the holotype with the paratype and non-types in parentheses.
Dorsal-fin elements X, 10; anal-fin elements III, 6; caudal fin slightly rounded, total caudal-fin rays - (30‒33), principal rays - (9+8), branched rays - (8+7); upper/lower procurrent caudal-fin rays -/- (6–9/6‒7). Scale rows above lateral line to the dorsal-fin origin 3 (3‒4); scale rows below lateral line 9 (9–10); cheek scale rows 4 (4–7); predorsal-fin scale rows 7 (6‒8); circumpeduncular scales 17 (17‒18).
First gill arch with 17 (15‒18) total rakers, including 3 (3–5) (rudimentary) +2 (1‒2) (developed) rakers on the upper limb and 7 (7–8) (developed)+5 (2–5) (rudimentary) rakers on the lower limb. Pseudobranchial filaments 12 (11–18). Branchiostegal rays 7. Vertebrae 10 (precaudal)+14 (caudal)= 24; supraneurals 3; formula for the configuration of supraneural spines and anterior dorsal pterygiophores 0/0/0+2/1+1/1; ribs present on the 3rd vertebrate to the 10th. Terminal dorsal pterygiophore in interneural space 16; terminal anal pterygiophore in interhaemal space 4.
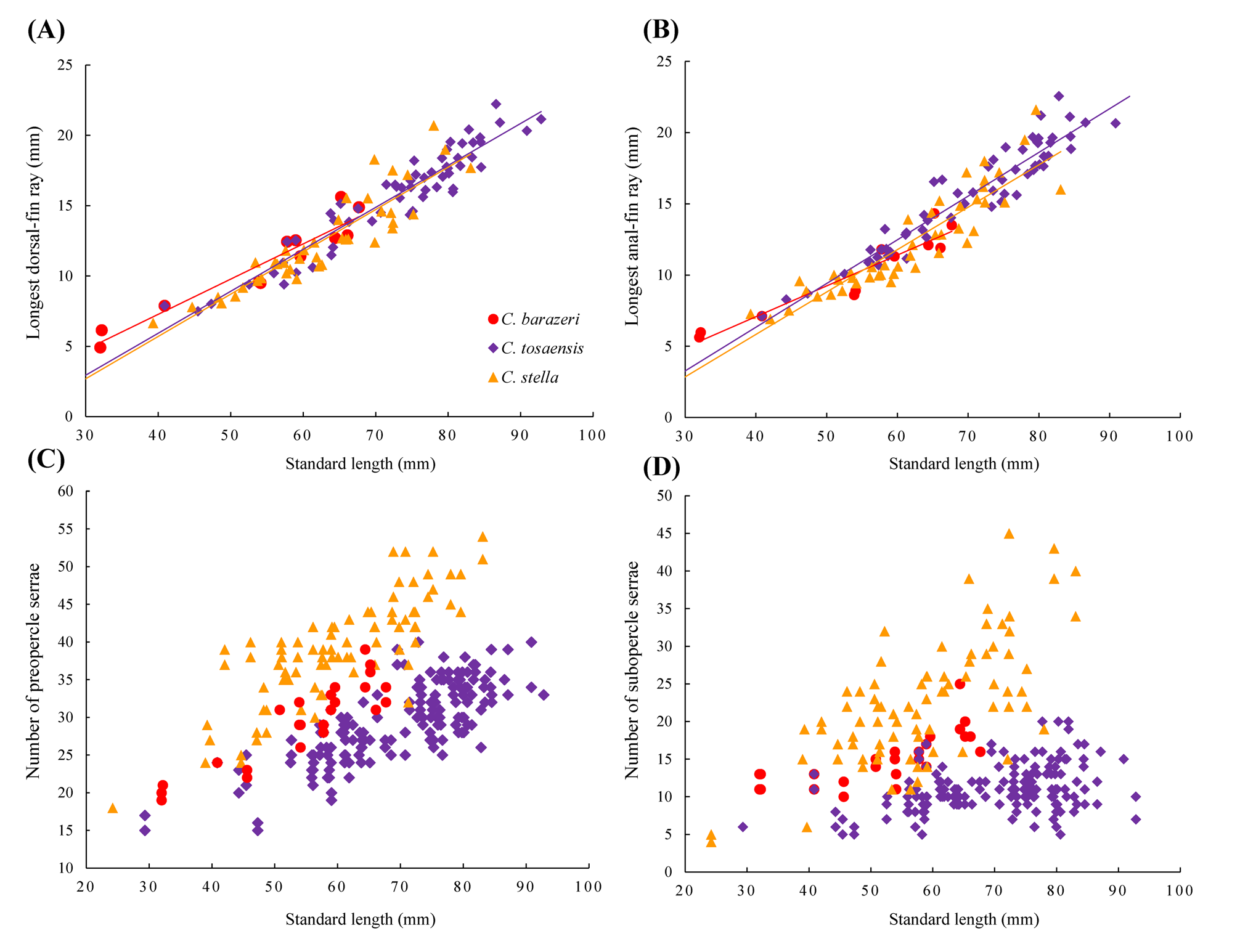
TABLE 1. Morphometric data and count of head serrae of Chelidoperca barazeri and two similar species. Means of head serrae were recorded to nearest integer. Data for comparison were adopted from Matsunuma et al. (2018). Data sources: A. Present study; B. Matsunuma et al. (2018). HT= holotype; D= dorsal; A= anal.
......Continued on the next page
Body elongated, cylindrical in section anteriorly, gradually becoming compressed posteriorly. Body depth and width 24.8% (22.1–26.9%) and 18.6% (16.6–20.1%) of SL, respectively. Head moderately large, its length 38.8% (37.1–38.8%) of SL, upper profile not steep. Snout moderately blunt, anterior margin forms an angle ca. 40‒50° with the upper jaw, its length 17.6% (15.6–21.3%) of HL, shorter than the eye diameter. Eye moderately large, its diameter 35.2% (29.9–36.6%) of HL; bony interorbital width narrow, 6.4% (5.9–7.5%) of HL. Mouth large and slightly oblique, the upper-jaw length 43.2% (42.7–46.7%) of HL. Postorbital length 50.4% (49.5–53.4%) of HL; predorsal length 38.2% (35.3–38.2%) of SL; preanal length 61.5% (60.0–63.1%) of SL; prepelvic length 32.6% (30.7–34.7%) of SL; caudal peduncle relatively long, length 63.6% (57.5–68.6%) of HL, the least caudal peduncle depth 32.0% (27.8–33.0%) of HL.
Dorsal-fin origin above a vertical through pectoral-fin base, the first spine shortest, 16.4% (11.8–19.4%) of HL; fourth spine longest, 44.5% (38.9–48.5%) of HL, length of spine gradually reducing after the fourth; the ninth spine 22.6% (14.9–23.0%) of HL, the last (tenth) spine longer than the ninth, 23.2% (19.6–25.6%) of HL; all soft rays branched, generally subequal in length, not elongated or filamentous, the penultimate (ninth) ray longest, its length 50.4% (40.0–62.5%) of HL. Anal-fin origin below the base of the second dorsal-fin ray; the third spine longest, its length 27.5% (16.7–27.8%) of HL; all soft rays branched, not elongated, the penultimate (fifth) ray longest, its length 49.3% (42.2–57.3%) of HL. Pectoral fin with uppermost two rays unbranched, remaining rays branched, its posterior tip do not reach the level of anal-fin origin; its length 72.0% (61.4–72.0%) of HL. Pelvic-fin origin below the pectoral-fin base; the spine covered with skin, its length 39.3% (27.8–40.5%) of HL; all soft rays branched, the second longest and elongate, 76.0% (61.3–76.0%) of HL, expanded distally, its tip pointed, reaching the anus when appressed. Caudal fin slightly rounded, its length 52.0% (52.0–75.0%) of HL.
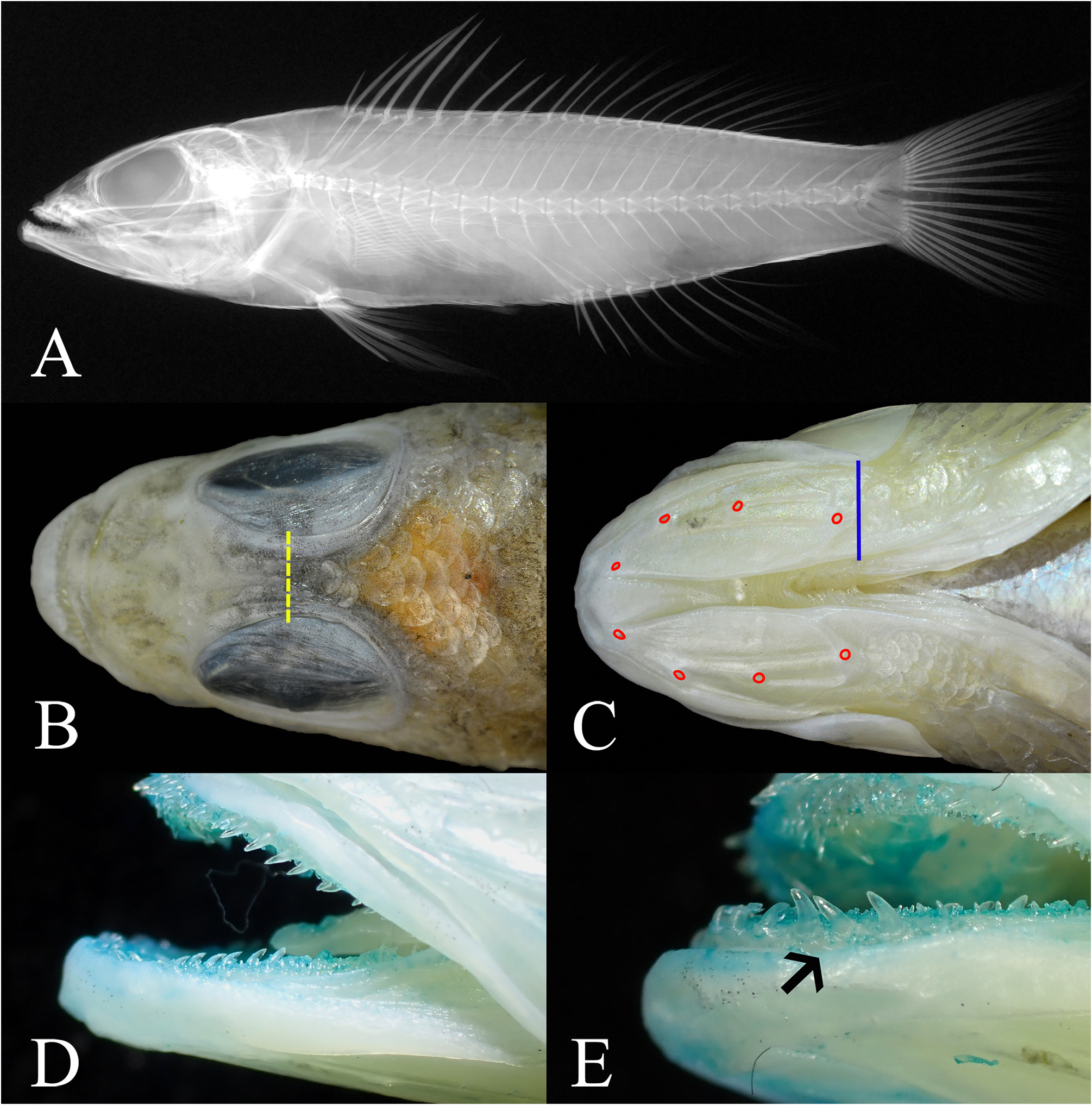
Anterior nostrils situated at the middle of snout, its opening with a small rounded flap rising from the posterior rim (or short membranous tube); posterior nostril an elliptical opening at anterior border of eye. Opercle with two flat prominent spines, the upper spine slightly longer than the lower spine, interopercular-spine width 12.8% (12.8‒16.3%) of HL; posterior edge to the angle of preopercle serrated, with 19/21 (19–39) well-developed serrae; upper distal edge of interopercle serrated, with 5/4 (4–18, one non-type without serrae on one side) weak, not welldeveloped serrae; lower distal edge of subopercle serrated, with 13/11 (10–25) weak serrae; posttemporal with a small bony crest with 2/2 (2–4) serrae tips at the beginning of the lateral line.
Maxilla with a low lateral ridge along the median level; ventral part of maxilla with a low skin fold extended from the premaxilla; lower jaw slightly protruding beyond the upper jaw when the mouth is closed. Bands of small conical teeth and enlarged canines on both jaws, vomer and palatine bands, teeth bands tend to be broader in larger specimens. The anterior (broadest) portion of tooth band on the upper jaw at symphysis with ca. 4–7 rows of small, sharp-tipped conical teeth, progressively longer towards the inner rows, the band becoming narrower posteriorly; outermost row of upper jaw with enlarged antrorse canines ( Fig. 3D View FIGURE 3 ); anterior (broadest) portion of lower jaw at symphysis with ca. 3‒4 rows of sharp-tipped conical teeth, progressively longer towards the inner rows, the outermost row of teeth at the symphysis stout and enlarged, the band becoming narrower posteriorly; the outermost row near symphysis at lateral side with ca. 3‒4 enlarged antrorse canines ( Fig. 3E View FIGURE 3 ), and enlarged canines along the innermost row. Vomer with a V-shaped band consisting ca. 3‒5 rows of small conical teeth at broadest part, a few enlarged canines at posterior end of each sides. Palatine band consisting ca. 2‒4 rows of small sharp-tipped conical teeth, the innermost row often enlarged.
Body generally covered with ctenoid scales, except for several regions with cycloid scales. Uppermost row of body scales along the dorsal-fin base about half size of the scales on body. Lateral line complete, slightly arched over pectoral fin and gradually descending, terminating at caudal-fin base. Pored lateral-line scales 35 (34–37). Scale rows in longitudinal series 37 (35–39). Pectoral fin with a scaly basal sheath. Scales absent on all rays and membranes of dorsal, anal and pelvic fins. Caudal fin with scaly basal sheath with ctenoid scales, scales extended to and covering one-third to half of the area from the fin base, scales on caudal fin ctenoid. Head generally covered with both ctenoid and cycloid scales; snout (including infraorbital), maxilla and ventral surface of dentary naked; ventral surface of the angular covered with ca. 2‒4 (more rows in larger non-type specimen, fewer in small specimens) rows of cycloid scales, not extended onto the dentary; cheek (preopercle) possesses both cycloid and ctenoid scales (larger non-type specimens with more ctenoid scales on the upper two-third area); scales on interopercle cycloid; scales on subopercle and opercle ctenoid; scales on interopercular membrane cycloid; interorbital with a single row of cycloid scale reaching or slightly anterior to the mid-orbit level (some specimens were judged by scale pockets due to scale loss). Prepelvic area possess mainly cycloid scales, except scale in the vicinity of pelvic-fin base with few and weak ctenii.
A pair of interorbital canals with numerous small pores running along outer margin of interorbital region, diverging outward anteriorly, and reaching between anterior and posterior nasal pores. Lower jaw with four, nonpaired large sensory pores on each side ( Fig. 3C View FIGURE 3 ), the anteriormost pore near symphysis, followed by two pores along the sensory canal of the dentary surface, the last one located between the junction of dentary and angular.
Fresh coloration. Based on adult specimens from Taiwan ( Fig. 1A‒C View FIGURE 1 ). Body pinkish red, becoming paler ventrally. Upper body darker, scales above lateral line with dark brown edges. Several small shiny white spots scattering along and above the lateral line. A faint reddish stripe (ca. 1.5 scale in width) running horizontally along the body axis. Chest shiny white. Lateral body with ca. 9 whitish or iridescent vertical rectangular bars along the ventral profile. Head (snout and jaws) generally reddish pink. Premaxilla with a white spot at the anterior part, becoming pale yellow posteriorly. Maxilla pale red. Lower jaw with a white spot near the symphysis (present on both sides). A small iridescent white stripe extends posteroventrally from the posterior edge of maxilla to the anteroventral margin of preopercle. Iris yellow with dusky reddish pigments. Upper rim of eye reddish. Cheek and opercle not uniformly red, region posterior to the maxilla sometimes more reddish (may varies due to condition of specimens). Opercular membrane between the spines with a distinct red spot (red ocellus), with a translucent white margin.
Spinous dorsal-fin membrane translucent white, with several irregular yellow spots, the distal margin of membrane yellow. A small group of dark reddish spot (ca. 4‒6) on the basal half of the 4 th to 6 th spine. Soft dorsalfin membrane translucent white, with 2‒3 rows of large yellow spot along the rays, upper distal margin translucent white. Anal fin translucent white with broad yellow margin, distal tip of soft rays sometimes reddish. Membrane between the branches of the last anal-fin ray creamy white, a pale yellow spot sometimes present on the mid-way. Caudal fin translucent white, becoming translucent yellow posteriorly. Two yellow spots near the base of the middle caudal-fin rays, a short vertical yellow bar at middle of the fin, several dusky reddish spots along dorsal and ventral margin. Pectoral fin yellow hyaline, an obvious reddish spot near the base of the middle rays. Pelvic fin with a creamy white anterior margin, remainder translucent yellow, distal tip of soft rays sometimes reddish.
Preserved coloration. Body pale white in general; upper body dusky, posterior field of scales with dark crescent in larger specimens ( Fig. 2A View FIGURE 2 ). A faint, discontinuous stripe formed by clusters of melanophores along the mid lateral body (more obvious in smaller specimens, see Fig. 2A‒B View FIGURE 2 ). Snout with a faint cluster of melanophores on each side. Dorsal fin with several irregularly clusters of melanophores along the base, spine and rays. Anal fin translucent white. Caudal fin dusky near the base. Pectoral fin base and distal tip of anal fin slightly dusky.
Distribution. Chelidoperca barazeri has only been recorded in its type locality, the Gazelle Channel of Bismarck Sea, Papua New Guinea ( Lee et al., 2019), and newly recognized in southwestern Taiwan. It is predicted that C. barazeri is a widespread species in the western Pacific.
Ecology. Chelidoperca barazeri is a relatively shallow water species compare to its congeners. It was trawled from seamount and bands at depth 150–160 m in its type locality ( Lee et al., 2019). In Taiwan, it is captured by bottom-trawl off southwestern Taiwan, the depth of the trawl operation is assumed to be rubble sandy bottom (topology of seafloor unknown) at depth ca. 100 m, judging from others by-catch species [e.g., Neomerinthe erostris ( Alcock, 1896) , Scorpaena miostoma Günther, 1877 ].
Size. Chelidoperca barazeri is recognized as a relatively small species. The largest specimen examined is 67.7 mm SL, which is a mature male individual (NMMB-P35233, Fig. 1A View FIGURE 1 ) with developed testes, several smaller specimens are mature females with ripe ovaries (NMMB-P35231, Fig. 1C View FIGURE 1 ; NMMB-P35237).
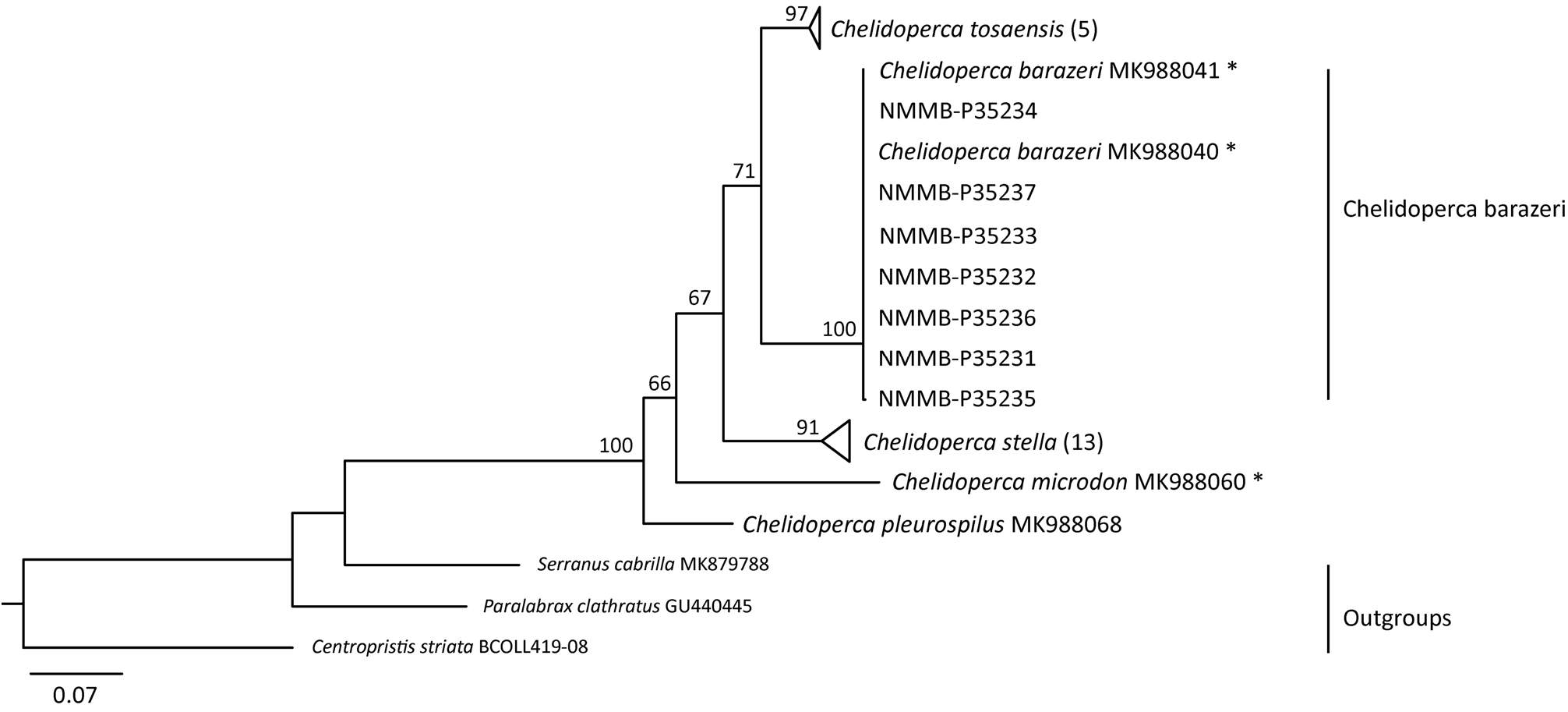
Remarks. Phylogenetic analysis based on the COI dataset ( Fig. 5 View FIGURE 5 ) highly support the identification of C. barazeri . The K2P genetic distance within C. barazeri from Taiwan and its type locality (northern Papua Guinea) is relatively small, ranging from 0.0‒0.2% within 9 specimens, compare to the mean interspecific distances ranging from 9.2‒16.9% calculated from the same dataset. This result is also expected at the range of intraspecific variation ( Ward et al., 2005; Holmes et al., 2009). In morphology, no significant difference was observed between the types and newly collected non-types in this study. In addition, C. barazeri is sister to C. tosaensis ( Fig. 4 View FIGURE 4 ) with medium support; their genetic distances are ranging from 8.8‒10.0%. Compare to another morphologically similar species, C. stella , their genetic distances are ranging from 11.6‒12.9%.
The fresh coloration of specimens of C. barazeri from type locality ( Fig. 1D View FIGURE 1 ) and Taiwan ( Fig. 1A‒C View FIGURE 1 ) are similar in general, but different in a few details. Types of C. barazeri possess four irregular dark pinkish red bands extend from dorsal profile to mid-body, each with longitudinally rectangular reddish blotches, and an oblique red stripe extended from infraorbital to the anteroventral margin of the opercle. These two characters are not obvious in the Taiwanese specimens. However, the differences in coloration may due to geographical or ontogenetic variation, or even the condition of the fresh specimens. Preserved coloration of types and Taiwanese specimens without distinctive difference, all these specimens possess a discontinuous line on mid lateral body formed by clusters of melanophores, fainter in larger specimens ( Fig. 2 View FIGURE 2 ).
| R |
Departamento de Geologia, Universidad de Chile |
| V |
Royal British Columbia Museum - Herbarium |
| KAUM |
Kagoshima University Museum |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Chelidoperca barazeri Lee, Lee, Matsunuma & Chen, 2019
| Tang, Chi-Ngai & Matsunuma, Mizuki 2022 |
Chelidoperca tosaensis
| Koeda, K. & Ho, H. - C. 2019: 668 |