Mesochaetopterus xerecus, Petersen & Fanta, 1969
|
publication ID |
https://doi.org/ 10.1111/j.1096-3642.2007.00342.x |
|
DOI |
https://doi.org/10.5281/zenodo.5747132 |
|
persistent identifier |
https://treatment.plazi.org/id/03FD7535-FFDF-FFB2-60B3-18EA3BC8F9DB |
|
treatment provided by |
Carolina |
|
scientific name |
Mesochaetopterus xerecus |
| status |
|
MESOCHAETOPTERUS XERECUS PETERSEN & FANTA, 1969 View in CoL
( FIGS 7–11 View Figure 7 View Figure 8 View Figure 9 View Figure 10 View Figure 11 )
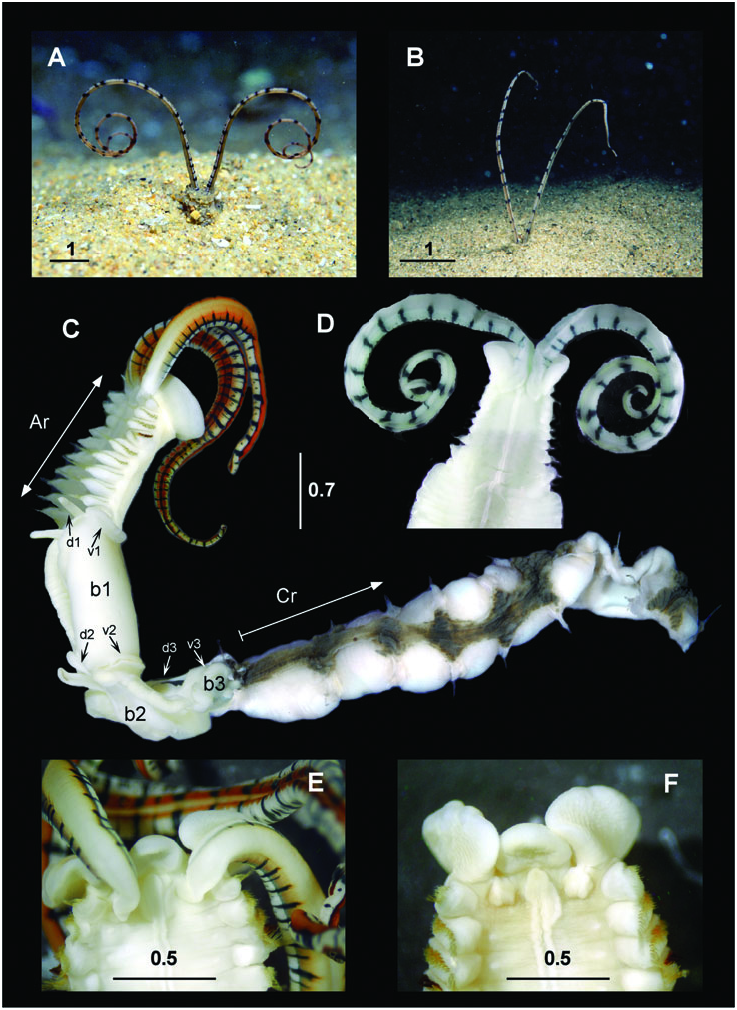
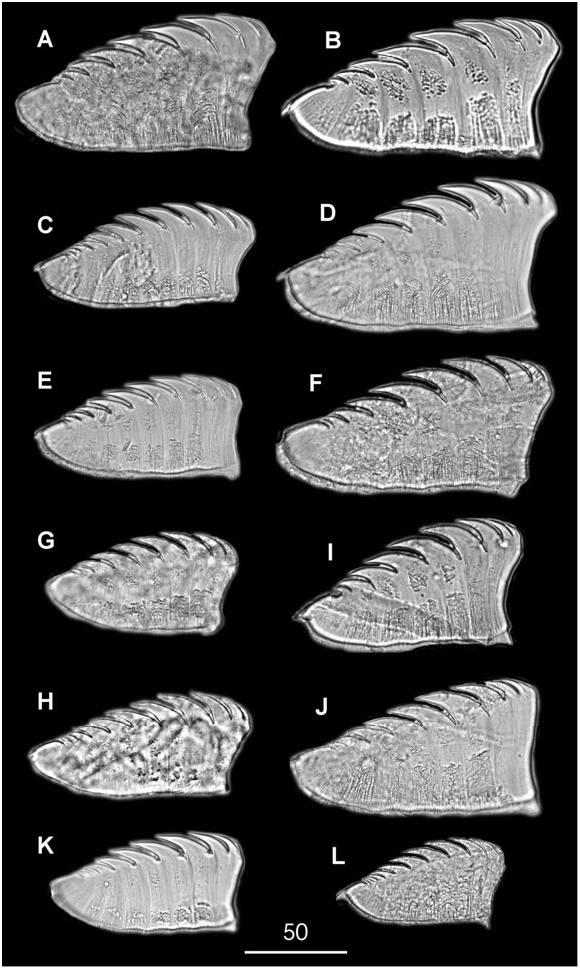
Diagnosis: Based on Petersen & Fanta (1969) and on observations of newly collected specimens. Large chaetopterid (reaching up to 60-cm long ‘ in vivo ’) with a well-developed peristomium (about one-fifth of the length of region A) surrounding the prostomium, and a pair of long peristomial palps up to twice as long as region A in preserved worms. Palps with transversal dark pigmented rings (dark-orange to greenishbrown) of different widths, without a clear alternating pattern, and a pair of longitudinal ciliated grooves (one dorsal and one ventral). Second pair of small antennae or palps absent. Eyes present between insertion of the palps and the peristomium. Dorsal ciliated faecal groove running from mouth to posterior end (anus), along the median body line. Ventral plastron occupying the whole of region A and up to one third of the first segment of region B.
Region A 1–1.5-cm long, with 8–14 chaetigerous segments (typically 11 or 12). Parapodia uniramous, notopodial lobes short; ventral glandular shield long, uniformly greenish in colour. Region B about 3-cm long, with four (but up to seven) elongate segments, and with associated feeding organs or cupules usually in segments 2–4 (but found up to segment 7). Parapodia biramous, as a flat plate-like region, with glandular lateral epithelium. Notopodia poorly developed, with a nearly triangular or wing-like shape. Uncinigerous neuropodia unilobed in segment 1, and bilobed in segments 2 and 3. Region C up to 55-cm long for 90–120 segments. Parapodia biramous, with unilobed notopodia and bilobed uncinigerous neuropodia. All segments similar, except for some in the posteriormost pygidial region, which are shorter and have reduced parapodia.
Tube longer than 1 m, parchment-like, externally covered by grains of sand (inconspicuous in worms from muddy sandy bottoms), vertical or J-shaped, with a transverse partition with three perforations in the lower part, closed at the lower extremity, and ending blindly in a nearly rounded apex.
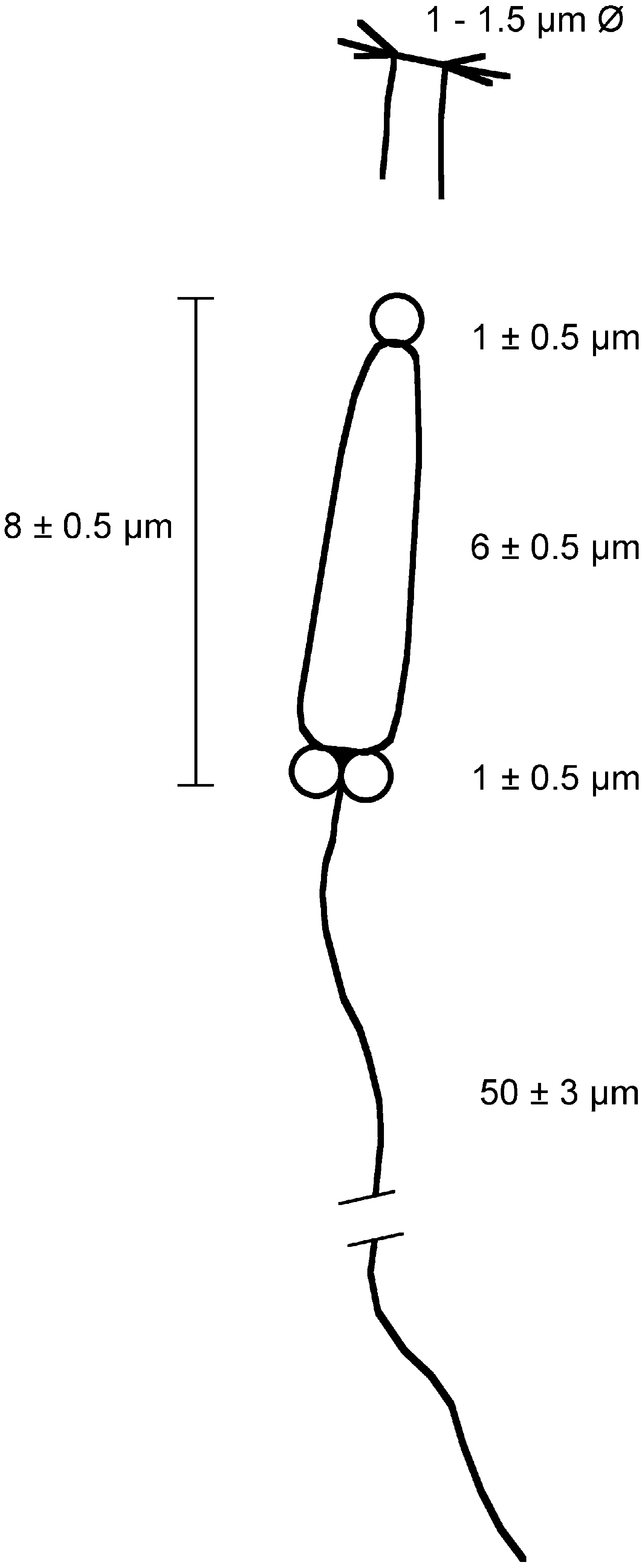
Males with elongate sperm having a long flagellum. Females with oocytes of about 200 Mm in diameter, which are present in all segments of region C. Population densities at Ilha do Mel reach about 100 individuals m-2.
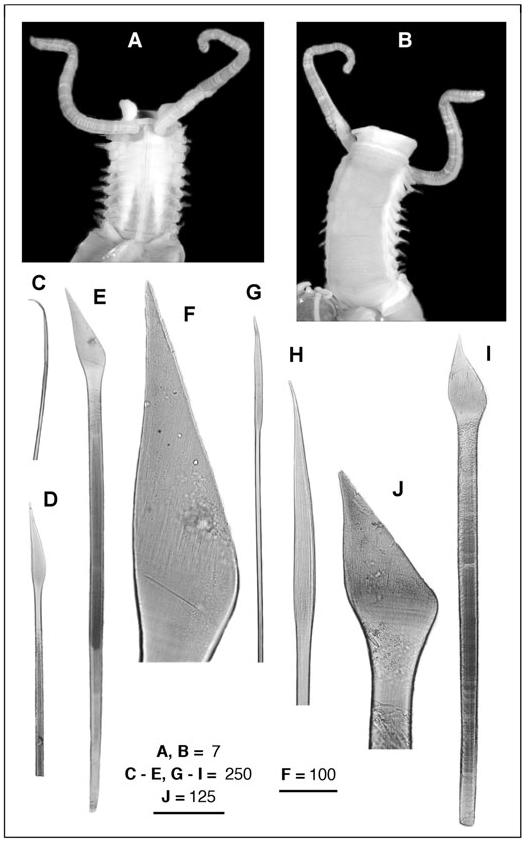
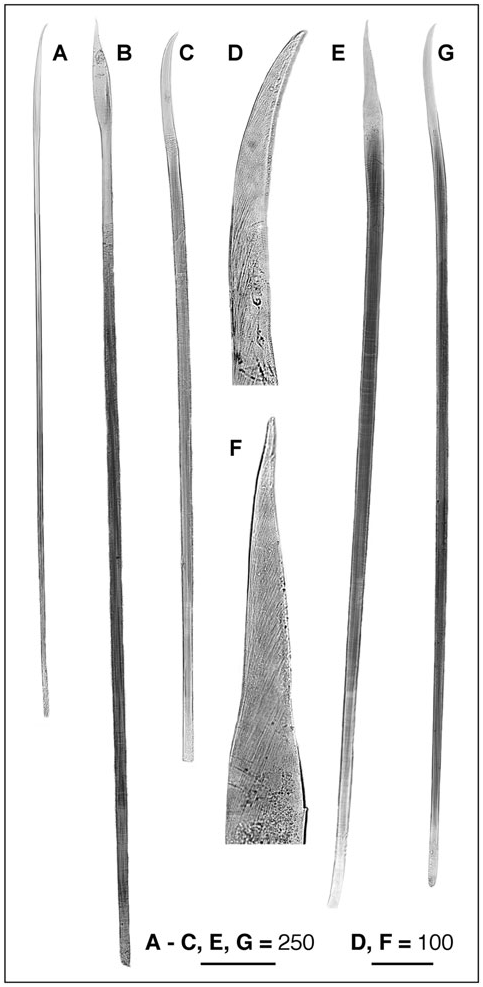
Region A: Between 9 and 14 segments, more frequently 9 (N = 3) or 13 (N = 3). Parapodia all uniramous, with notopodia only. Chaetae yellowish to pale orange (dark brown in A4), occurring dorsolaterally in several irregular, question mark shaped rows (a single row in A4). Chaetal arrangement changing in segments A1, A2–A6 (except the A4), A7–A9, and A10–A14; notopodia of Al with two types of chaetae, up to 25 very long and fine lanceolate to capillary dorsal chaetae, becoming finer as progressing to the dorsum, and about 30 small lanceolate, oar-like lateroventral chaetae; notopodia of A2–A14 with up to 30 very long and fine lanceolate dorsal chaetae, their flattened ends becoming shorter when more dorsal; notopodia from A2–A3 and A5–A6 with up to 35 oar-like lateroventral chaetae, the ventralmost chaetae twice as wide and long as the lateral chaetae; notopodia from A7 to A9, with up to 30 bayonet ventral chaetae and a few hooked lateroventral chaetae; the bayonet chaetae becoming smaller and progressively replaced by the hooked ones in the posteriormost segments; from A10 to A14, the hooked chaetae fully replacing the bayonet chaetae.
A4 notopodia with up to 15 yellowish, transparent, finely pointed lancet-like dorsal chaetae (d); five yellowish, transparent, knife-like chaetae more than twice as wide as the lancet-like ones (ld1); between two and five dark-yellow to brownish knife-like chaetae, stouter than the previous ones, with the tip of the curved edge slightly serrated (ld2, ld3); 12–14 (typically 11) asymmetrical, knob-like, stout modified chaetae having serrated tips, the ventral chaetae (v) smaller than the lateral ones (lv1–lv3). Modified chaetae dark brown, often partly embedded in the notopodia.
Region B: Usually with four (up to seven) segments having biramous parapodia. Between 12 and 16 extremely long, thin, notochaetae, scarcely protruding from the tip of the notopodia; about 12 with flattenedto-lanceolate tips, and two or three with pointed tips. Neuropodia unilobed in segment B1, and bilobed in B2 and B3, with several hundreds of uncini irregularly disposed in two or three rows, roughly triangular, with seven (between six and ten) long curved teeth plus a few small ones (often difficult to distinguish) in the anterior and posterior ends of the serrated edge. All uncini of B1 similar in size; dorsal uncini of B2 and B3 smaller than ventral uncini.
Region C: Incomplete GoogleMaps , with more than 50 segments, all biramous. Notopodia GoogleMaps with about ten long and thin chaetae (shorter than the notochaetae of B region), scarcely protruding from the tip of the notopodia, with flattened to lanceolate tips. Neuropodia GoogleMaps all bilobed, with uncini irregularly disposed in two or three rows, similar in shape and teeth arrangement to those of region B. Uncini differing in size both dorsoventrally and from anterior (smaller) to posterior (larger) sections of each neuropodia, clearer in anteriormost segments, and progressively less evident in the posteriormost segments, becoming basically similar in size around segment C30.
Material examined: Ten incomplete specimens (reaching up to 55 segments in region C) from a low intertidal sandy beach, Ilha do Mel, Paranaguá Bay (Paraná, Brazil), 25°34 ′ S, 48°20 ′ W, October 30 2001, collected by V. Radashevsky, MNCN 16.01/10148.
Habitat, behaviour and population density of Mesochaeopterus rogeri sp. nov.
Mesochaetopterus rogeri sp. nov. commonly inhabits fine to coarse grain sandy bottoms (of 400–600 Mm in grain diameter) between 5- and 30-m deep (but most commonly between 6- and 15-m deep). These locations are often subject to medium-speed currents, which may facilitate the suspension-feeding behaviour of the worm. Different kinds of particles (possibly potential food) transported by the water currents were observed ‘ in situ ’ when they came into contact with the tentacles: they remained attached to the surface of the tentacles and began to be transported towards the mouth with the help of the ciliated grooves.
When undisturbed, the tentacles protruded from the tube opening and formed a V, with both tips arranged in a spiral ( Fig. 1A View Figure 1 ). Changes in current speed or direction did not trigger any response from the worm, except for the modification of the tentacle position induced by the new conditions, which may lead toward a less regular tentacle arrangement in the case of stronger currents ( Fig. 1B View Figure 1 ). Any contact with the surrounding sediment induced the worm to retract inside the tube. A slow constant retraction occurred after a subtle contact. If the contact was not repeated, the worm stopped the reaction and re-acquired the typical position after a few minutes. Either repeated or violent contacts with the surrounding sediment caused a fast retraction inside the tube. However, after several minutes, the worm protruded again to adopt its usual position.
The worms were able to expel both introduced liquids and small particles (such as sand grains) from the interior of their tubes, by means of a powerful exhalation current. In normal conditions, with the worms completely inside the tube, there was a regular exhalation/inhalation water flow, probably generated by peristaltic movements of the segments in region C. This flow seemed similar to those described as a part of the feeding mode and ventilation system for other chaetopterid polychaetes, including Mesochaetopterus ( Barnes, 1965; Sendall, Fontaine & O’Foighil, 1995).
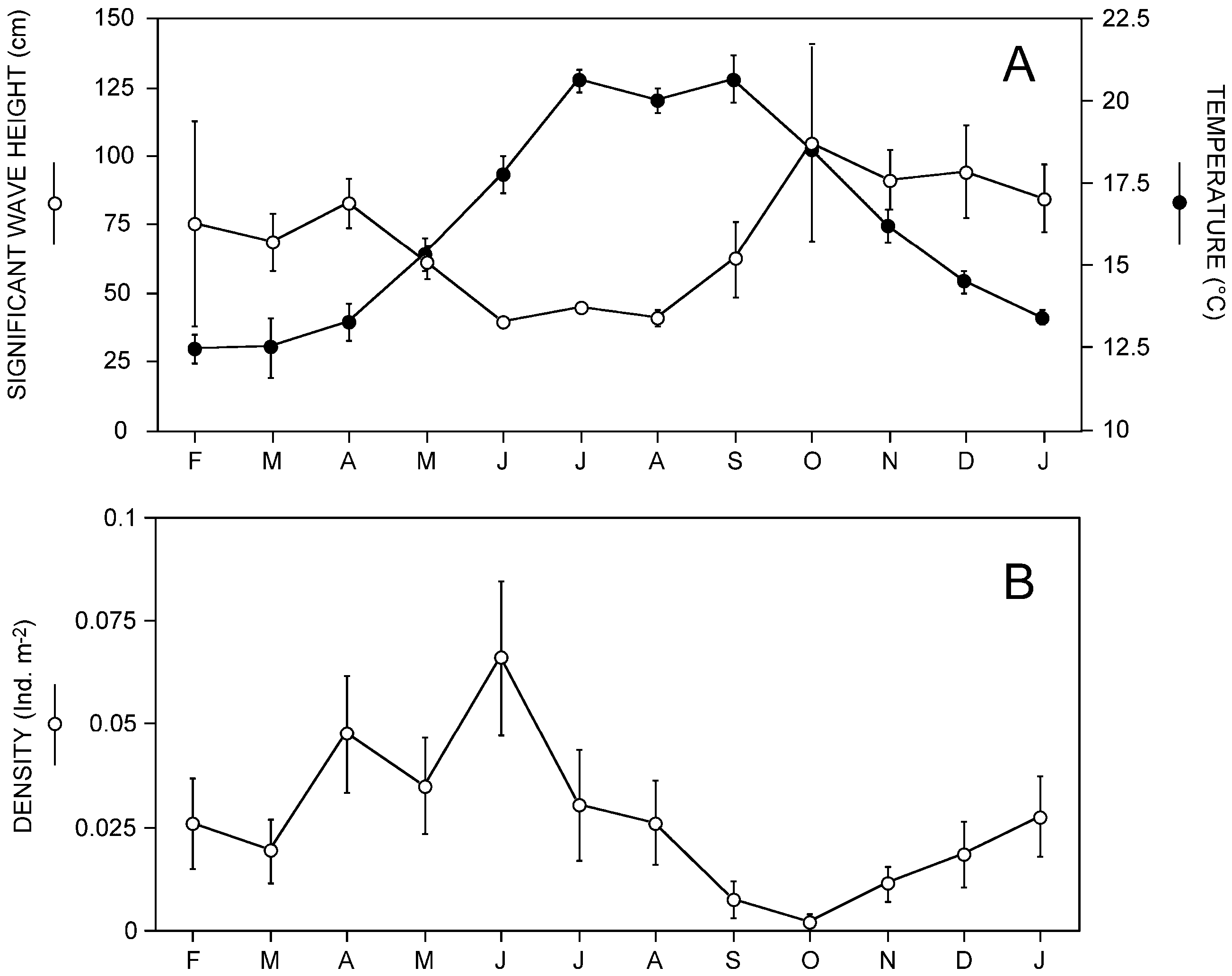
The population density ranged between 1 and 3 individuals 40 m-2, but may reach up to 1 individual 10 m-2 ( Fig. 12B View Figure 12 ). However, they were frequently found in clusters of 2 or 3 individuals m-2, separated from their neighbours by large unoccupied areas. Maximum densities tended to occur from April to June, together with rising temperatures, and could perhaps be related to recruitment events, as proposed for Mediterranean soft-bottom invertebrates ( Sardá et al., 1995, Sardá, Pinedo & Martin, 1999). However, the seasonal pattern of this species differed from the general pattern proposed by Sardá et al. in that remarkable minimum densities occurred between September and November (particularly in October). This could not be explained by an increasing mortality after recruitment events leading to basal adult densities, as previously proposed ( Sardá et al., 1995, 1999). In M. rogeri sp. nov., the density decrease did not seem to be related to mortality, but to an adaptation to survive under the increasing instability of the sediment during the stormy autumnal period characteristic of a Mediterranean environment ( Fig. 12A View Figure 12 ). In fact, the extraordinary length of the tube, as well as the ability to quickly retract inside, could represent an adaptation to such an unstable sedimentary regime. Together with the increase of instability of the water column (as expressed by the increase in wave height, Fig. 12A View Figure 12 ), the sediments tended to become more and more mobile, to the extent that the worms were not able to protrude above the surface, and survived by hiding inside the tube, at a depth where the sediment was stable. This could explain the autumnal decrease in density ( Fig. 12B View Figure 12 ), as the divers could not count the worms that did not protrude from the sediment surface. Moreover, this could also explain the occasional presence of empty ramifications and the huge number of empty tubes found during the excavation collection. Accordingly, these were probably functional tubes, abandoned by the worms either during their normal growth or as a result of the aforementioned adaptive process. Once the storms ended, the worms built new tube sections to reach the surface, so that there would be as many empty tubes as stormy events during the lifetime of each worm.
This mode of life supports the potential influence of M. rogeri sp. nov. in structuring the surrounding sediments, as a sediment stabilizer (dense populations inside extremely long tubes, palisades of empty tubes). Also, the presence of a cylinder of compact sediment surrounding the tube all along its length (i.e. more than 2.5-m deep into the sediment, and reaching up to 6 cm in diameter) in M. rogeri sp. nov. (also reported for M. taylori ) has been attributed to an ‘aerobic halo’ generated by the presence of the tube ( Sendall et al., 1995). Therefore, a possible significant bioturbating potential can also be attributed to the M. rogeri sp. nov. populations.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
SubOrder |
Spionida |
|
Family |
|
|
Genus |