Ichnea plumbea Gorham, 1877
|
publication ID |
https://doi.org/10.1649/0010-065X-64.mo4.1 |
|
persistent identifier |
https://treatment.plazi.org/id/317D6408-DC67-FFE8-AB46-FA46FC87FAD6 |
|
treatment provided by |
Carolina |
|
scientific name |
Ichnea plumbea Gorham, 1877 |
| status |
|
( Figs. 108, 109 View Figs , 152 View Figs , 164 View Figs , 176 View Figs , 198 View Figs ; Map 5 View Map 5 )
Ichnea plumbea Gorham, 1877: 413 . Lectotype ♀. Herein designated. Amazon, Bates (BMNH). (Specimen pin mounted on polyethylene foam with gender label; round type label; round syntype label; locality label; collection label; identification/ locality label; BMNH acronymic label; lecotype label.) Lohde 1900: 102; Schenkling 1903: 102, 1910: 123; Blackwelder 1945: 389; Corporaal 1950: 271.
Ichnea helvolicollis Corporaal, 1950: 94 . New synonymy. The specific epithet is a replacement name for a junior homonym ( I. roseicolle Kuwert ) whose specimen it represents is a color variant of I. plumbea . Lohde 1900: 102; Schenkling 1903: 102, 1910: 123; Corporaal 1942: 143, 1950: 270.
138) Ichnea praeusta ; 139) Ichnea aterrima .
Paralectotypes. Seven specimens. Brazil: Amazonia : Amazon, Bates ( BMNH, 6; MNHN, 1) .
Diagnosis. The members of this species are easily separated from other species in this genus by the extensive width of the vertex, which is much wider than the width of the eyes in dorsal view.
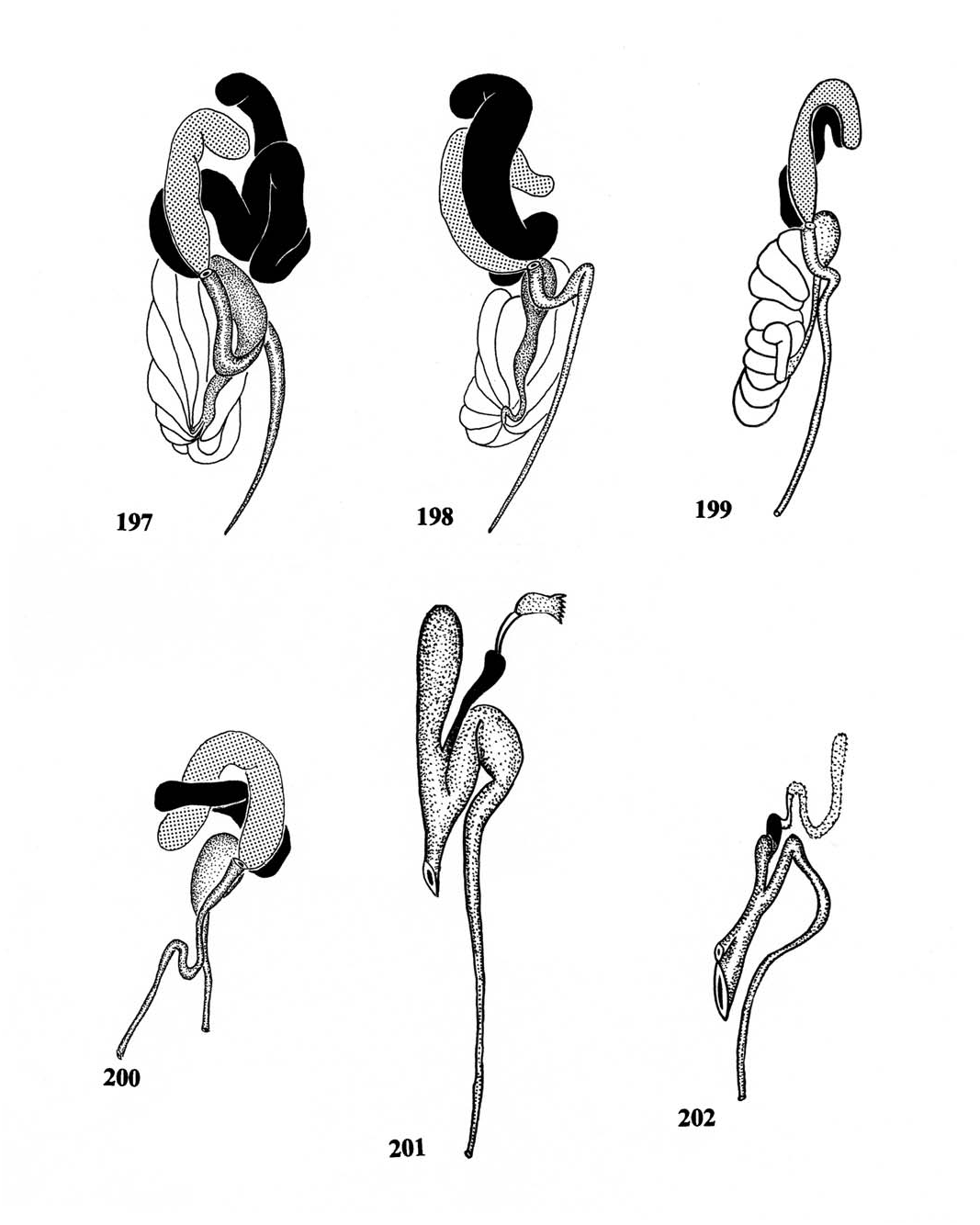
Description. Size: Length 8.1 mm; width 2.5 mm. Form ( Fig. 152 View Figs ): Oblong subovoid. Integumental color ( Figs. 108, 109 View Figs ): Cranium mostly yellow, frons dark brown, antennal mostly dark brown, apex yellow; pronotum mostly brown, with yellow lines at sides; elytra mostly brown, epipleural and sutural margins yellow; femora yellow, tibia and tarsi brown. Head: HW/PW 1.0; frons indented; EW/VW 1.5; antenna as in Fig. 164 View Figs , funicle not compressed. Thorax: PL/PW 0.95; elytra broadly
141) Ichnea dimidiatipennis .
143) Ichnea digna ; 144) Ichnea acanthomelina .
subovoid, EL/PL 4.1; EL/EW 1.9; anterior margin of protibia with 14 spines. Male genitalia: Aedeagus as in Fig. 176 View Figs . Male mesodermal internal reproductive organs: Testes comprised of 12 follicles.
Variation. Size: length 6.1–10.2 mm; width 2.0– 4.8 mm. The pronotum may be roseate. The antennal apex is yellow or brown, and there is considerable variation in the width of the yellow stripe along the epipleural margin. In a few specimens, the yellow epipleural and sutural marginal stripes are only faintly expressed.
Natural History. Specimens have been collected throughout the year at altitudes ranging from 30 to 350 m. Some were captured within a Malaise trap in a tropical rainforest and others on felled mahogany logs.
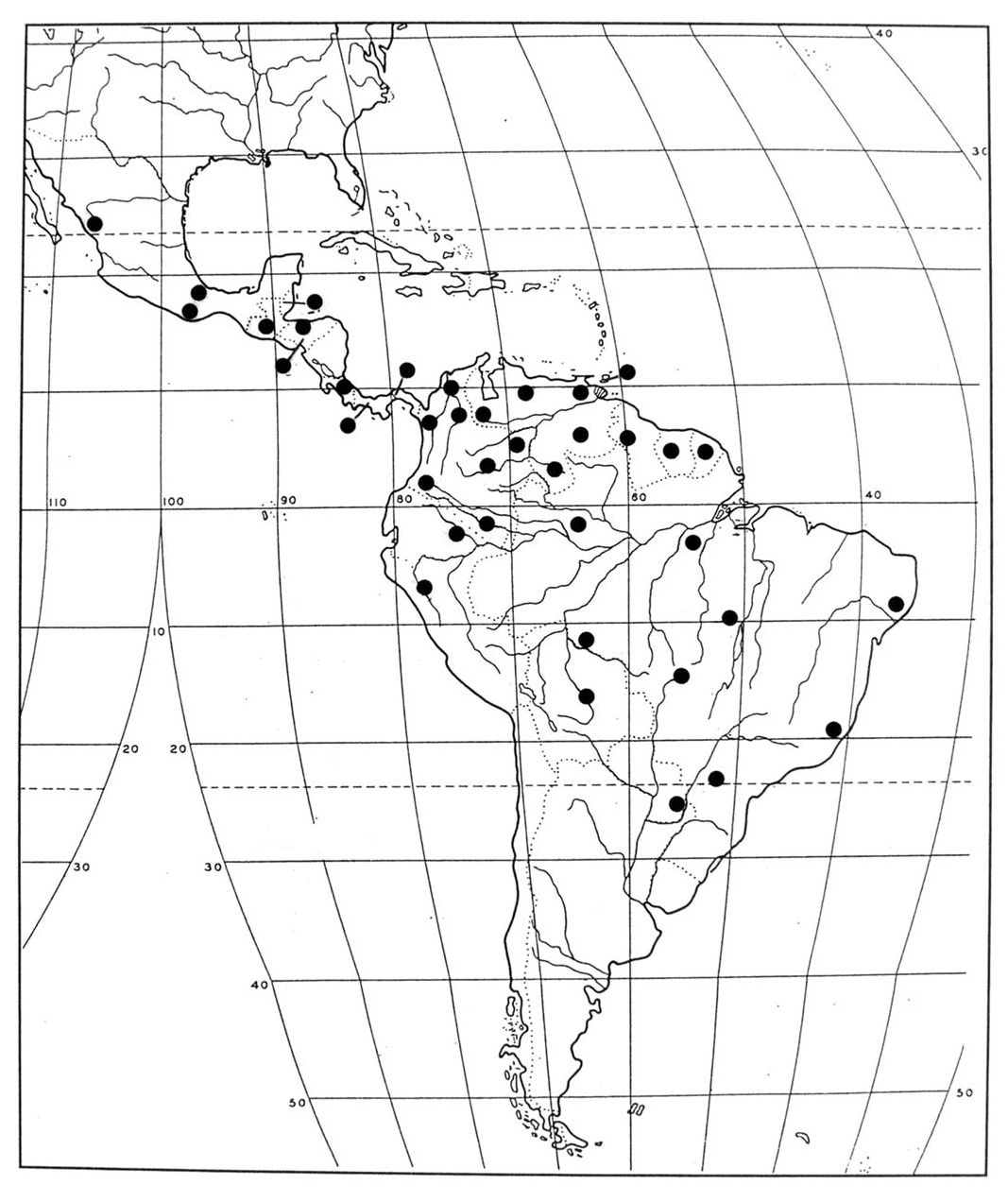
Distribution. Distributed from Panama to Brazil ( Map 5 View Map 5 ).
Specimens Examined. 77. PANAMA: Panamá, Fort Clayton, Panama Canal Zone, 21-VI-1972, on mahogany log, H. Stockwell; Cerro Azul, 29-V- 1983, J. E. Wappes; Bayano District, 5 km S Ipiti, 24-V-1992, J. E. Wappes; Cerro Campana, 29-VI- 1974, 350 m, beating underside of palm leaves, Erwin & Whitehead; Pipeline Road k 1-12, 26-30-VI-1997, Wappes and Morris; 6–10 km N El Llano, 26-IV-4- V-1992, E. Giesbert; Gamboa road, 7-V-1984, E. Giesbert. COLOMBIA: Caquetá, Chiribiquete, Rio Cuñaré, bosque tierra firme, 0°31′N 72°38′W, 29-X-12-XI- 2000, 300 m, Malaise trap, M. Ospina & E. Gonázlez: Putumayo: La Paya Manansoys, Tierra Alta, 0°6′S 74°58′W, 22-26-IX- 2001, 350 m Malaise trap, D. Campos; La Paya Fca. Charapa, 0°8′S 74°57′W, 22-26-IX-2001, D. Campos; La Paya Fca, Charapa, 0°08′S 74°57′W, 26- III-10IV- 330 m Malaise trap, R. Cobete; La Paya Finca. Charapa, 0°8′S 74°57′W, 28-IV-14-V- 2002, 330 m, Malaise trap, R. Cobete: Amazonas, Amacayacu, Matamata, 3°41′S 70°15′W, 8-14-VIII- 2000, 150 m, Malaise trap, A. Parente; Amacayacu, Camino a San Martin, 3°41′N 70°15′W, 1-10- III- 2004, 150 m, Malaise trap, T. Pape & D. Arias. TRINIDAD: Arima Borough, Arima, Arena forest reserve, 13-VI-8-VIII-1993, 80 m, rainforest Malaise trap, S. & J. Peck. ECUADOR: Orellana, Tiputini Biodiversiy Station near Yasuni National Forest, 00°37′S 76°08′W, 7-II-1999, 220– 250 m, T.L. Erwin: Napo, 24 km E Atahualpa, 16-23-X- 1995, 480 m, E. & V. Giesbert; 24 km E Atahualpa, 1-13-X- 1996, 450 m, E. Giesbert; Yasuni Reserve Station, 19-30-X- 1998, 250 m, W. J. Hanson; 28 km NE San Pedro de Avajuno, 15-30-III-2005, F. T. Hovore. PERU: Loreto, Yanamono Lodge, Quebrad Yanamono, 1 km N Rio Amazonas, 3°22′S 72°47′W, 28-VI-1978, H. A. Hespenheide; Chambireyacu Yurimaguas, 1885, M.de Mathan. BOLIVIA:
152) Ichnea plumbea ; 153) Ichnea divisa .
La Paz: Tumupasa , XII , W. M. Mann. BRAZIL: Amazonas, 1894; Manaus , VIII-1959, C. Ellas: Pará: Santarem, Rondônia: 62 km SE Ariquemes, 7-18- XI-1995, W. J. Hanson ; idem, 5-16-XI-1996, W. J. Hanson ; idem, 22-31-X-1997, W. J. Hanson; 62 km SE Ariquemes 1-14-XI-1997, B. Dozier; 62 km SW Ariquemes, Rancho Grande , 20-31-X-1997, B. K. Dozier ; idem, 1-17-XI-1997, B. K. Dozier; 62 km SW Ariquemes, Rancho Grande, 16-XI-1994, C. W. & L. B. O’ Brien; 62 km SW Ariquemes, Rancho Grande, 4-16-XI-1997, J. E. Eger; Vilhena, XI- 1973, M. Alvarenga: Mato Grosso, Sinop , X- 1975, M. Alvarenga: Specimens are deposited in BMNH, CHAH, CMNC, EMUS, FSCA, IAVH, JEWC, JNRC, OXUM, STRI, USNM , and WOPC.
155) Ichnea marginella ; 156) Ichnea atra .
EVOLUTIONARY CONSIDERATIONS
There is substantial interspecific and intraspecific color variation among the taxa under study. Unfortunately, characteristics involving integumental color are minimally valuable for proposals of species-level phylogeny. Therefore, this treatise of epiphloeine evolution deals with genus-level relationships among Acanthocollum , Stegnoclava , and Ichnea , and among the species-groups of Ichnea . The three aforementioned genera comprise a monophyletic group within Epiphloeinae .
Two major lines of evolution are apparent in the comparatively speciose genus Ichnea . Although these two lines are readily discernible at the species level, the magnitude of their difference does not justify their separation into two genera, when compared with the magnitude of genus-level morphological differences in Epiphloeinae . Both lines concern the conformation of the elytra, with one line having developed a wedge-shaped body and the other a more ovoid body form. The shapes of the pronotum are also useful in the character phylogeny that follows, as is the proximity of the compound eyes to each other. Lastly, the surface character of the frons, whether planar or excavated, showed significance in hypotheses of species-group phylogeny.
PHYLOGENETIC INTERPRETATION
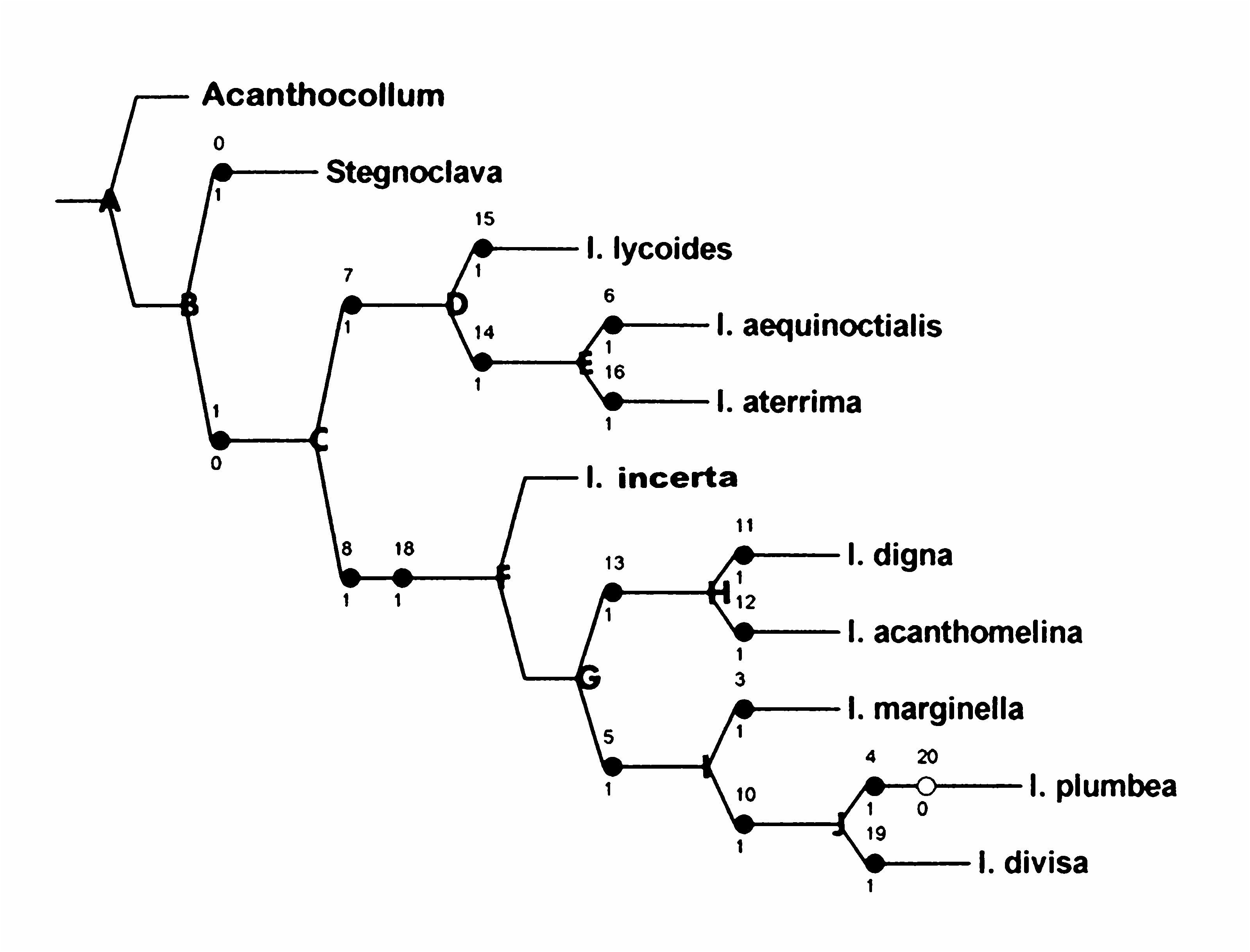
My current understanding of the phylogenetics of the taxa under consideration are depicted in Fig. 203 View Fig . The phylogeny was generated by computer via NONA (Goloboff 2003) in combination with Winclada version 1.00.08 (Nixon 2002). The analysis resulted in a single tree (L = 19, Ci = 100, Ri = 100).
Mimicry is a synapotypy (Kavanaugh 1978; Opitz 2007) for the generic grouping comprised of Acanthocollum , Stegnoclava , and Ichnea . It is postulated that the progenitor of these three genera evolved in South America. This conjecture is based on the distributional parameters of the extant taxa: Acanthocollum is exclusively South American, whereas Stegnoclava and Ichnea are predominantly South American. The aforementioned progenitor evolved a transverse pedicel and large number of protibial spines. In time, this ancestral species (stem species A) yielded two major lines of evolution, one led to Acanthocollum , which evolved a pronotum laden with linear aggregates of setae. The complementary stock (ancestral species B) led to Stegnoclava and Ichnea . In the former lineage, the antennal funicle became reduced in length, whereas in the latter lineage, which evolved into ancestral species C, the scape carina was lost and there developed a triangular, depigmented area on the distal margin of the pygidium.
Ancestral species C generated two main branches of Ichnea evolution. One branch, which led to ancestral species D, developed a line of epiphloeines that closely approximates members of the beetle family Lycidae . Ancestor D generated a lycoides stock in which the pronotum became distinctly trapezoidal. The complementary stock led to ancestor E, which evolved into sister groups aequinoctialis and aterrima . In the former lineage, the vertex became very narrow, and in the later line the pronotum narrowed variably in its anterior half.
Ancestor F had evolved away from the oblongrectangular body form to approximate a more lampyroid, oblong-suboval body form, and the epipleuron became explanate. This progenitor yielded the monotypic incerta lineage and its complementary stock that eventually evolved into ancestor G. The later promulgated two lines of evolution, one of which established ancestors H and I. Ancestor H yielded the digna lineage, which evolved a red-colored pronotum, and the acanthomelina line, which is characterized by having mostly black pronotal coloration. Ancestor I evolved three lines of evolution, one of which involves the highly vagile and monotypic marginella group, which is characterized by nearly contiguous eyes when viewed dorsally. The complementary lineage led to ancestral species J in which the pronotum became roseate in color. Finally, ancestor J diversified to yield the plumbea line and the divisa line, the former characterized by a very wide frons and the latter by having an expanded yellow marking on the medial aspect of the epipleuron.
ZOOGEOGRAPHIC CONSIDERATIONS
It is likely that insect groups whose species are widely distributed, and who are involved in comprehensive mimetic complexes, have had an extensive evolutionary history. During that history, considerable alteration of structure and functions would have taken place to approximate model species and to parallel geographically the model distributional character. The known distribution of Ichnea species , in which mimicry is extensively
193) I. aterrima , male; 194) I. aterrima , female; 195) I. callanga , male; 196) I. callanga , female.
engrained, is very scattered at both species and species-group levels, which is presumed to be a manifestation of Batesian polymorphism (Joron and Mallet 1998).
The distributions of Acanthocollum , Stegnoclava , and Ichnea are depicted in Maps 1–6 View Map 1 View Map 2 View Map 3 View Map 4 View Map 5 View Map 6 and are summarized in Table 2. Table 2 reviews the pres- ence or absence of the taxa under consideration among Middle American and South American clerofaunal areas of endemism, which were defined by Opitz (2005).
What is most apparent when one views the contents of Table 2 is that the distributional patterns of Ichnea species traverse nearly all the South American clerofaunas. Only two species, I. gremia and I. dimidiatipennis , are known only from one clerofaunal region, which is likely to be an artifact of collection, since the known distribution of these two species is based on one specimen each. More- over, of the 23 taxa involved in Table 1, four have traversed into Insular Central America (Opitz 2005) , two into Nuclear Central America, and two into Mexo-America. The widespread distribution of I. aequinoctialis and I. aterrima , which are among the most recently derived species (see phylogeny, Fig. 203 View Fig ), in conjunction with the extensive intraspecific variation in elytral color within those species, lends support to the hypothesis that ancestral Ichnea proliferated in South America and generated ancestral stocks that be- came widely distributed northward after the early closure of the Panamanian portal, about 25 million years ago (Ford 2006). A few of these ancestral stocks would also have traversed the Nicaraguan Depression (Whitehead and Ball 1997) of Central America ( sensu Opitz 2005) .
2005). + indicates overlap of species into more than one area of endemism.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |