Uroobovella phoenicicola, Kontschán & Tambe & Riolo, 2012
|
publication ID |
https://doi.org/ 10.5733/afin.053.0205 |
|
persistent identifier |
https://treatment.plazi.org/id/03EF87D9-E27C-6520-FE5F-FEBFF6A34F07 |
|
treatment provided by |
Felipe |
|
scientific name |
Uroobovella phoenicicola |
| status |
sp. nov. |
Uroobovella phoenicicola sp. n.
Figs 1–17 View Figs 1–3 View Figs 4–9 View Figs 10–13 View Figs 14–17
Etymology: The name of the new species refers to the name of the host species on which the specimens were found.
Description:
Female.
Length of idiosoma 460–490 μm, width 370–400 μm (n=4). Body shape oval, colour reddish brown.
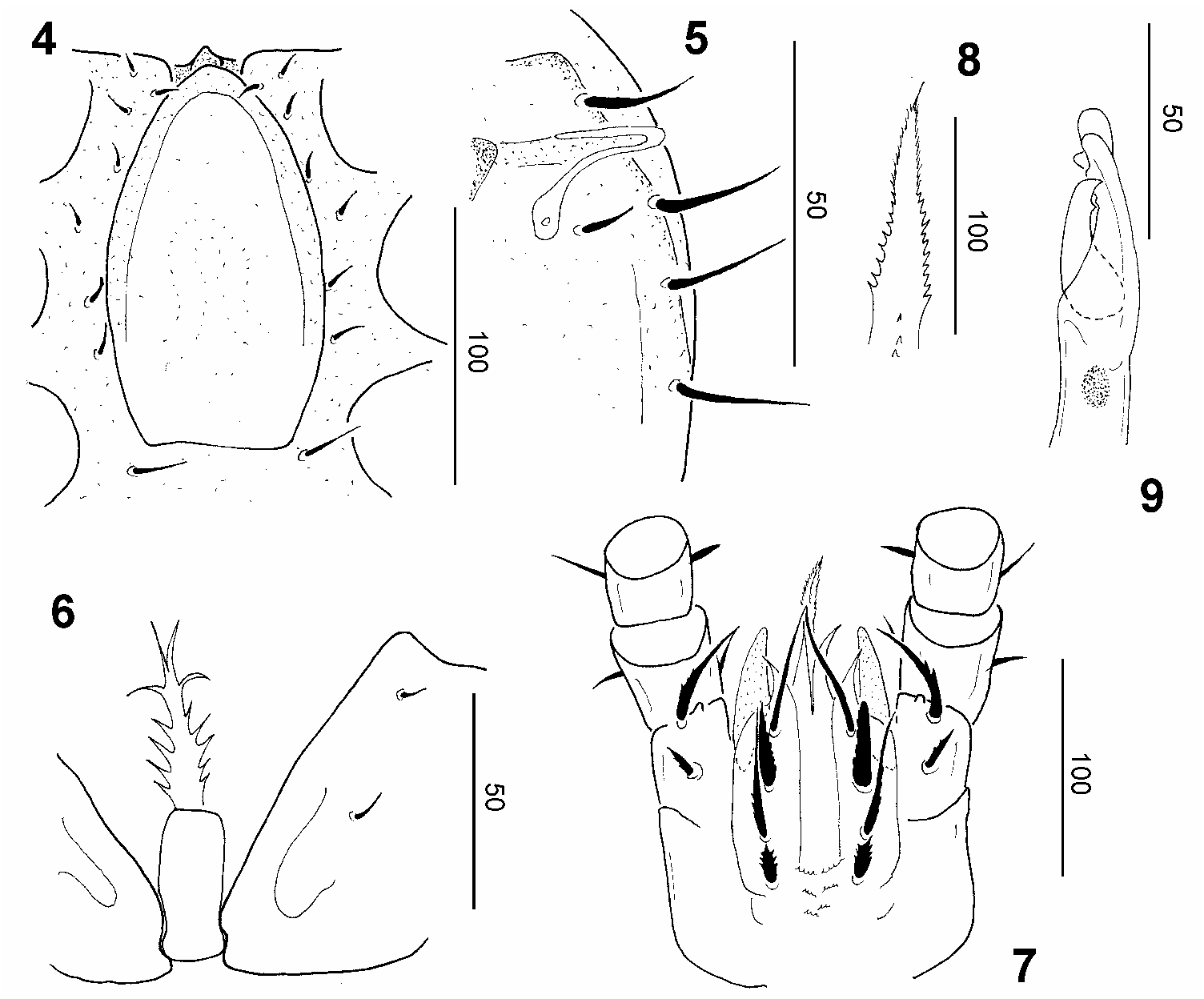
Dorsal idiosoma ( Fig. 1 View Figs 1–3 ): Dorsal and marginal shields fused anteriorly. All dorsal setae smooth and needle-like, but differ in size; most setae long (ca 30–40 μm), but several central setae short (ca 14–15 μm). Marginal shield smooth and bearing long (ca 30–35μm) and needlelike setae ( Fig. 2 View Figs 1–3 ). Dorsal shield smooth, muscle scars present on caudal part of central area.
Ventral idiosoma ( Fig. 3 View Figs 1–3 ): Sternal shield smooth, all sternal setae needle-like, St1–St7 short (ca 11–12 μm), St8 long (ca 22 μm). St1–St3 situated near anterior margin of genital shield, St4 at level of posterior margin of coxae II, St5 at level of anterior margin of coxae III, St6 at level of central area of coxae III, St7 at level of anterior margin of coxae IV, St8 placed near basal edges of genital shield ( Fig. 4 View Figs 4–9 ). Ventral shield smooth, several oval pits situated at level of anal opening. Ventral setae smooth and needle-like, V1, V3 and setae around anal opening short (ca 13–17μm), other setae on ventral idiosoma long, postanal (ca 41 μm) and two posterior setae (ca 50–55 μm) longer than other ventral setae (ca 35–40 μm). Two pairs of lyriform fissures present near pedofossae of leg IV. Stigmata situated between coxae II and III. Peritremes hook-shaped, poststigmatid part short ( Fig. 5 View Figs 4–9 ). Genital shield of female scutiform, placed between coxae II and IV, without sculptural pattern and anterior process ( Fig. 4 View Figs 4–9 ). Pedofossae deep, their surface smooth, with separate furrows for tarsi IV, metapodal lines absent. Base of tritosternum narrow, its laciniae marginally serrate and apically divided into four branches ( Fig. 6 View Figs 4–9 ).
Gnathosoma ( Fig. 7 View Figs 4–9 ): Corniculi horn-like, internal malae smooth and as long as corniculi. Labrum marginally pilose. Hypostomal setae: h1 long (ca 61 μm), smooth and setiform; h2 short (ca 39 μm), robust and marginally serrate; h3 long (ca 57 μm) and basally serrate; h4 short (ca 20 μm), robust and marginally serrate. Epistome basally serrate and apically pilose ( Fig. 8 View Figs 4–9 ). Chelicerae with internal sclerotised nodes, fixed digit with apical sensory organ and longer than movable digit, both digits bearing a single tooth ( Fig. 9 View Figs 4–9 ).
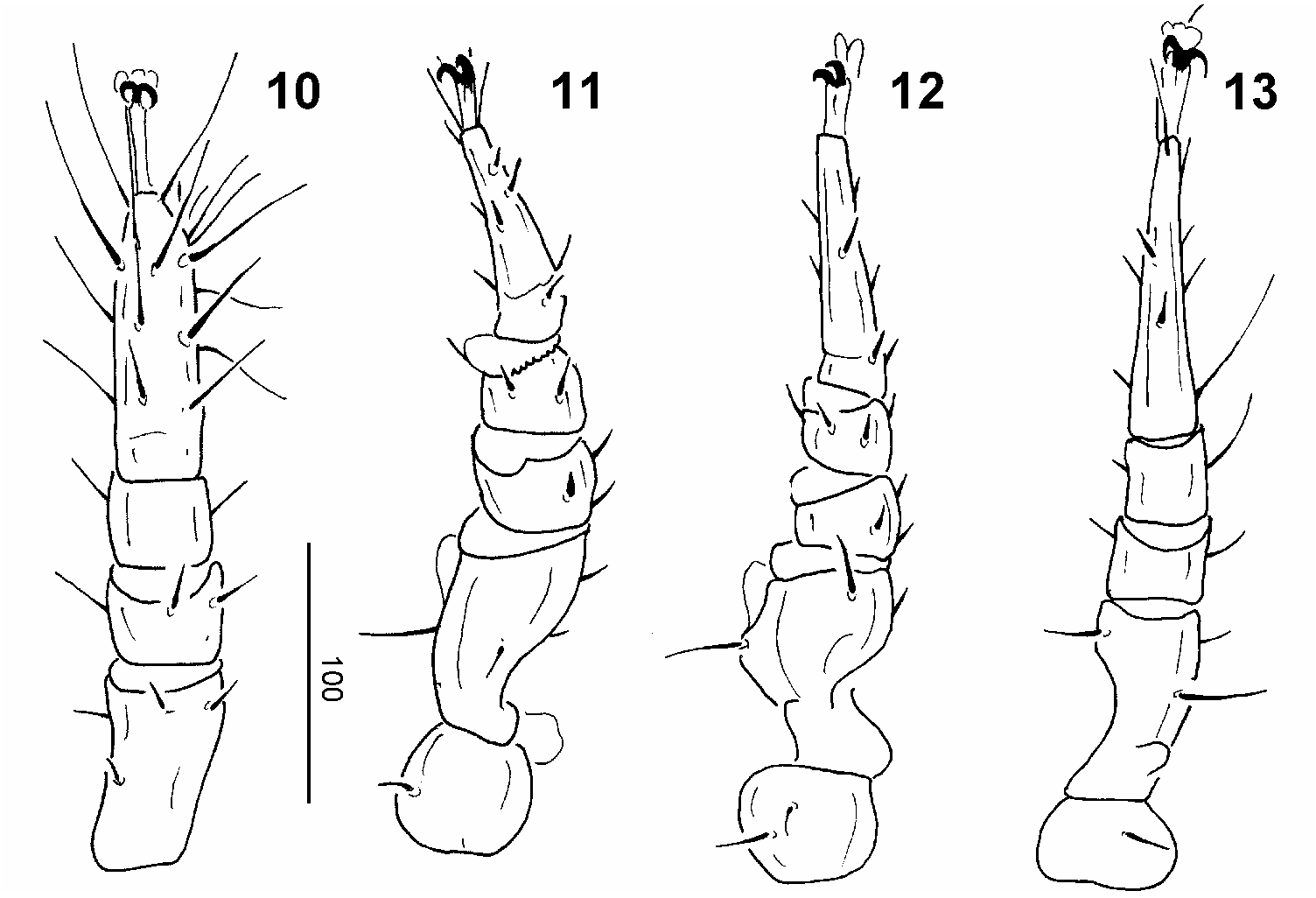
Legs ( Figs 10–13 View Figs 10–13 ): All legs bear claws on tip of tarsi and have needle-like setae.
Male.
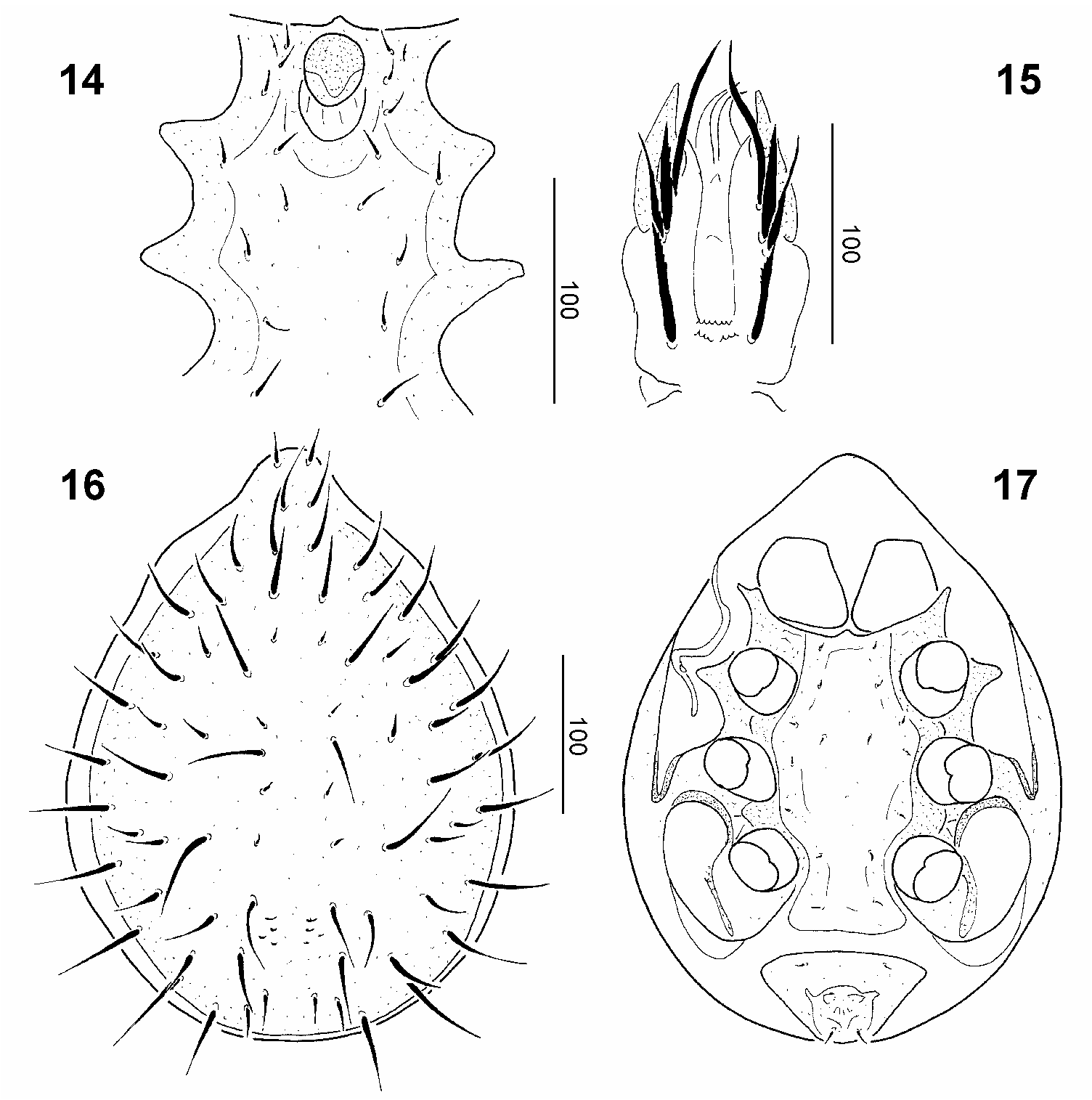
Length of idiosoma 460–470 μm, width 340–360 μm (n=4). Shape of idiosoma, ornamentation and chaetotaxy of dorsal parts as in female. Sternal setae short (ca 12–13μm) and needle-like, St1–St4 situated around margins of genital opening, St5 at level of anterior margin of coxae III, St6 posteriorly to St4 and at level of central area of coxae III, St7 at level of central area of coxae III, St8 at level of anterior margin of coxae IV, St9 at level of central area of coxae IV. Sternal shield smooth and bearing one pair of lyriform fissures near base of St1. Genital shield oval and located between coxae II ( Fig. 14 View Figs 14–17 ). Ventral setae and ornamentation similar to those of female.
Gnathosoma ( Fig. 15 View Figs 14–17 ): Corniculi horn-like, internal malae smooth and as long as corniculi. Hypostomal setae: h1 long (ca 74 μm), smooth and setiform; h2 (ca 50 μm) robust and marginally serrate; h3 (ca 46 μm) and h4 (ca 43 μm) marginally serrate. Other parts and processes similar to those of female.
Deutonymph.
Length of idiosoma 370–380 μm, width 270–280 μm (n=2). Dorsal idiosoma ( Fig. 16 View Figs 14–17 ): All dorsal setae smooth and needle-like, but differing in size: most setae long (ca 17– 20μm), but several setae in central area short (ca 9–10 μm). Dorsal shield without sculptural pattern, several muscle scars present on caudal part of central area.
Ventral idiosoma ( Fig. 17 View Figs 14–17 ): Sternal setae short (ca 5–6 μm) and needlelike, St1 situated near anterior margin of sternal shield, St2 at level of anterior margin of coxae II, St3 at level of posterior margin of coxae II, St 4 in central area of sternal shield, St5 at level of anterior margin of coxae III, St6 at level of posterior margin of coxae III, St7 at level of central area of coxae IV, St8 near posterior margin of sternal shield. Sternal shield smooth, without sculptural pattern. Ventrianal shield smooth, with two pairs of setae (ca 5–8 μm). Peritremes long and Sshaped.
Larva and protonymph. Unknown.
Holotype: ♀ REPUBLIC OF CAMEROON: South West Region, Manyu Division, Ashum Village , from cocoons formed by Rhynchophorus phoenicis weevils, 25.iv.2011, J. Tanyi Tambe ( HNHM, in alcohol).
Paratypes:All with same data as holotype : 1♀ 1♂ 1 nymph ( NHMG, in alcohol) ; 2♀ 3♂ 1 nymph ( HNHM, in alcohol) ; 1♀ ( HNHM, on slide) .
Note: The adults are from one Rh. phoenicis cocoon, and nymphs are from the body of the weevil.
Remarks: The new species is placed in the Uroobovella minima -group ( Hirschmann 1989) on the basis of the shape of the idiosoma and peritremes, number of sternal setae, and the presence of claws on the tip of leg I. Up until now, 23 species have been described in this group, one of which is known only from the deutonymph stage ( U. inhaerens (Vitzthum, 1921)) . The sternal shield of this species is ornamented by oval pits, but it is smooth in the new species. Only male specimens of U. vietnamvarians Hirschmann, 1981 are known. The genital shield of this species is situated between coxae III and IV, whereas that of the male of the new species is between coxae II. The characteristic differences between the new species and females of the Uroobovella minima -group are given in Table 1.
Biology: Adult mites were obtained from cocoons formed by Rhynchophorus phoenicis Fabricius, 1801 weevils, while deutonymphs were collected from the body of this weevil. The deutonymphs are either attached to the body of the weevils or move freely under their elytra. Not much is known about the biology of the mite, but since the adults were collected in cocoons, we assume that the species is a predator and feeds on the eggs and/or other life stages of the weevils. It is also possible that the mites prey on nematodes, maggots, juvenile slugs, fungi and/or organic matter that clusters on and around the cocoons, and use the weevils as a means of transportation only.
NOTES ON POSSIBLE ROLE IN BIOLOGICAL CONTROL
The African palm weevil is a large, reddish brown to black weevil that is distributed throughout tropical Africa and is a true pest of members of Arecaceae (= Palmae) ( Gries et al. 1994) and Poaceae (= Gramineae). In Cameroon, where the study was carried out, the larvae are commonly known as ‘tumbu’ or ‘tumbu for palm tree’ in pidgin English, but have different appellations according to the 270 spoken dialects. These larvae are eaten as a delicacy when seasonally available. They have a high fatty acid content, and are a source of proteins and carbohydrates ( Ekpo & Onigbinde 2005) as well as essential amino acids. Moreover, medicinal value has been ascribed to the larvae ( Nzikou et al. 2010; Zaid 1999). The pest status of the African palm weevil has already been established ( Bong et al. 2008). The weevils cause severe damage as a result of active feeding on the palms but also serve as a vector for plant parasitic nematodes ( Kanzaki et al. 2009). They therefore play a major role in crop damage and yield loss. Several strategies have been tried and implemented for the control of these weevils, such as felling of diseased palms, and the use of pheromone baits and traps. However, the only natural method in trial so far is the use of entomopathogenic nematodes ( Zaid et al. 2002). If the new mite species is indeed an obligate predator on the eggs and of developmental instars of this beetle, it will be interesting to determine if in the future the mite can be used as a biological control agent.
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.