Neoephydra ciligena (Rondani)
|
publication ID |
https://doi.org/10.11646/zootaxa.4116.1.1 |
|
publication LSID |
lsid:zoobank.org:pub:22D15539-E49E-4D6C-BFCF-D4DBC72BA640 |
|
DOI |
https://doi.org/10.5281/zenodo.6059431 |
|
persistent identifier |
https://treatment.plazi.org/id/967487E2-FFA6-FFAA-FF4D-98F9FE0BFABE |
|
treatment provided by |
Plazi |
|
scientific name |
Neoephydra ciligena (Rondani) |
| status |
|
16. Neoephydra ciligena (Rondani)
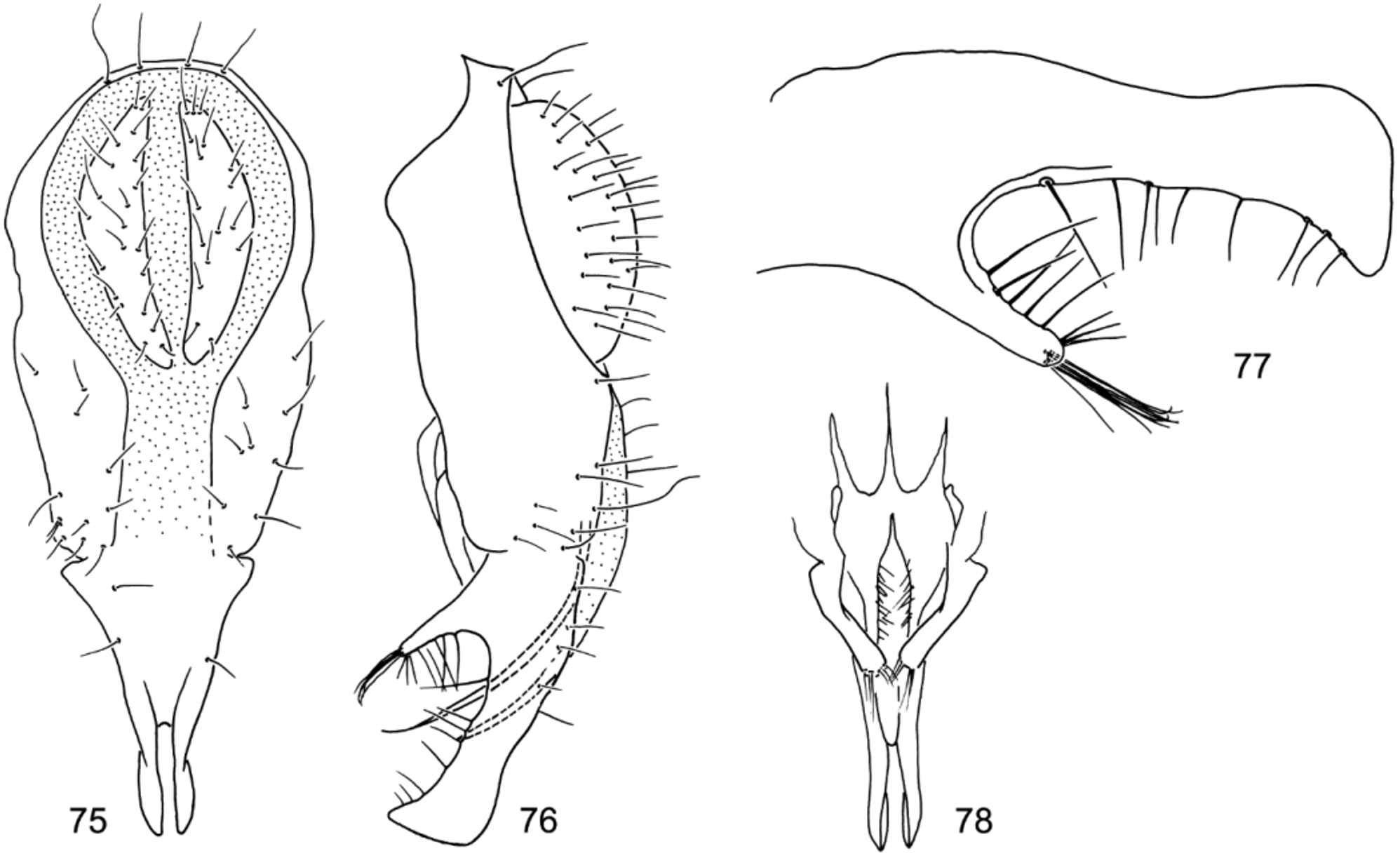
Figs. 75–78 View FIGURES 75 – 78 , Map 16
Ephydra ciligena Rondani 1868: 32 View in CoL .
Parydra ciligena .— Wirth 1968: 22 [generic combination, Neotropical catalog]. Dimecoenia ciligena View in CoL .— Clausen 1985: 382 [generic combination].— Mathis and Zatwarnicki 1995: 239 [world catalog]. Neoephydra ciligena .— Mathis 2008: 10 [generic combination].
Ephydra densepilosa Hendel 1930: 152 View in CoL . New synonym
Dimecoenia densepilosa View in CoL .— Hendel 1933: 223 [generic combination].— Wirth 1968: 23 [Neotropical catalog].—Lizarralde de Grosso 1989: 58 [list, Argentina].— Mathis and Zatwarnicki 1995: 239 [world catalog].
Neoephydra densepilosa .— Mathis 2008: 10 [generic combination].
Dimecoenia prionoptera View in CoL , in part [misidentification].— Cresson 1935: 350 [listed questionably in synonymy].
Dimecoenia lopesi Oliveira 1954b: 269 View in CoL .— Wirth 1968: 23 [Neotropical catalog].— Mathis and Zatwarnicki 1995: 239 [world catalog]. New synonym
Neoephydra lopesi .— Mathis 2008: 10 [generic combination].
Dimecoenia grumanni Oliveira 1954b: 272 View in CoL ; 1958: 167 [description and illustration of immature stages].— Wirth 1968: 23 [Neotropical catalog].— Mathis and Zatwarnicki 1995: 239 [world catalog]. New synonym
Neoephydra grumanni .— Mathis 2008: 10 [generic combination].
Diagnosis. Specimens of N. ciligena are distinguished from similar congeners of the araucaria group by the following characters: appearance variable, either generally dark, particularly the legs, or pale colored, with legs mostly reddish yellow; face moderately setose; gena high; and structures of male terminalia with distinctive conformation. Moderately large to large shore flies, body length 4.10–5.60 mm; generally dark colored, grayish brown to brown with some subshiny to shiny, golden brown or brown areas dorsally.
Head: Head ratio 0.67–0.70; frontal ratio 0.47–0.50; subquadrate mesofrons shiny with golden brown to greenish brown metallic luster, moderately pilose anteriorly, lateral margins becoming slightly narrower anteriorly; ocellar triangle and parafrons dull, microtomentose, brown to blackish brown; ocelli in isosceles triangle, distance between posterior ocelli less than between medial ocellus and either posterior ocellus; parafrons mostly blackish brown, lacking any subshiny area through insertions of fronto-orbital setae. Antenna mostly unicolorous blackish brown, densely microtomentose; arista thickened on basal 1/3, gradually tapered to style-like, bare tip, basal portion with macropubescent vestiture; aristal length equal to combined length of 1st 3 segments. Facial ratio of females 0.80–0.82, of males 0.93–0.96, densely microtomentose, mostly unicolorous, brown to golden brown; dorsum of interfoveal hump with small anterior area subshiny, golden brown; facial setae moderately well developed, particularly along dorsal slope and oral margin, larger setae along dorsal slope slightly anaclinate. Eye ratio 1.0; gena-to-eye ratio 0.46; gena moderately high, anterior portion concolorous with face becoming slightly more yellowish posteriorly.
Thorax: Generally brown; scutum mostly subshiny to shiny, microtomentose only along anterior and lateral margins; becoming shinier and darker posteriorly; scutellum concolorous with posterior portion of scutum. Pleural areas generally concolorous, dull, microtomentose, blackish brown to grayish brown, becoming only slightly paler toward venter. Wing very palely infuscate, pale grayish brown; wing length averaging 3.38–3.47 mm; costal vein ratio 0.25–0.28; M vein ratio 0.71–0.72. Legs mostly concolorous; femora variable, either concolorous with pleural areas, with tibiae blackish brown dorsally, ventral surface somewhat rufous to brownish orange, and tarsi blackish brown except at articulations which are brownish orange, or with legs mostly pale, reddish yellow.
Abdomen: Generally subshiny dorsally, becoming duller, more microtomentose toward lateral margins; first 4 tergites with large subshiny area toward anterior margin with dark, greenish brown, metallic luster, lateral margins becoming gradually duller and grayer; 5th tergite of male mostly brown, at most subshiny, sides tapered gradually to bluntly rounded apex. Male terminalia ( Figs. 75–78 View FIGURES 75 – 78 ): epandrium in posterior view becoming narrower and parallel sided below level of cerci; surstyli in posterior view with broadly rounded basal enlargement, becoming abruptly narrower along ventral projections; surstylus in lateral view with ventral projection curved at tip and slightly enlarged, lateral projection over 1/2 length of ventral projection, claviform.
Type material. The lectotype female of Ephydra ciligena Rondani (designated by Clausen 1985: 382) is labeled “ HOLOTYPE Ephydra ciligena Rondani P.J.Clausen, 1984 HOLOTYPE [red]/Correct genus now Dimecoenia P.J. Clausen, 1984 [handwritten, white with a black sub-border].” Although Clausen (1985) labeled this specimen as the “ HOLOTYPE,” Rondani's original paper is not specific about whether the type series is a single specimen or a series. Thus the primary type is better considered a lectotype rather than a holotype. The lectotype is directly pinned, is in poor condition (covered by fungal hyphae), and is deposited in the MZUF.
The lectotype male of Ephydra densepilosa Hendel , here designated to stabilize and make more universal the use of this name, labeled “Tapikiole-Arg[entina]. XII. 25–I.26 [ Dec 1925 – Jan 1926] Lind O. Chaco-Exped/ Ephydra densepilosa Ƌ Hendel F. Hendel det. [species name, sex symbol, and author’s name handwritten]/ Type Hendel 1930 [handwritten in red, black border]/ LECTOTYPE Ƌ Ephydra densepilosa Hendel By W. N. Mathis [handwritten, black sub-border].” The lectotype is double mounted (minute nadel in paper rectangle), is in good condition (the abdomen has been removed and dissected; the structures are in an attached microvial), and is deposited in the SMNS. A female paralectotype is also designated herein.
The holotype male of Dimecoenia lopesi Oliveira is labeled “[ Brazil] Recr. dos Band[eirantes]. 24-IV- 940 [ 24 Apr 1940] Lopes/ COL. INST. O. CRUZ NO. 714/ HOLOTYPE Dimecoenia lopesi Oliveira [red].” Allotype female and one male paratype are labeled with the same label data as the holotype except for the collection numbers, which in these specimens are 715 and 716 respectively. The type series is in the IOC. The holotype is directly pinned and is in excellent condition. The abdomens of the allotype and paratypes have been removed, presumably for purposes of illustration, and were not included with the pinned specimens that we examined.
The holotype male of Dimecoenia grumanni Oliveira is labeled “TORRES, 1. XI. 950 [ 1 Nov 1950] RIO GRANDE do SUL E. Gruman/ COL. INST. O. CRUZ NO. 723/ HOLOTYPE Dimecoenia grumanni Oliveira [red].” Allotype female (No. 739) and five paratypes (3Ƌ, 2♀) are labeled with the same label data as the holotype except for the collection numbers which are 722, 724, and 738 for the males, and 740 and 748 for the females. The type series is in the IOC. All specimens of the type series are double mounted on paper points and appear to have been collected in alcohol or a moist killing jar before being mounted. Consequently they are not in the best of condition. The holotype is the best preserved specimen.
Type locality. Argentina. Capital Federal: Buenos Aires ( 34°40'S, 58°24'W).
Additional specimens examined. ARGENTINA. Formosa: Estancia Tapikiolé ( 25°01'S, 59°19'W), E. Lindner ( 1♂, 1♀; SMNS). Jujuy: Yavi Chico ( 22°06'S, 65°28'W), Oct 1968, L. E. Peña (2♀; USNM). Mendoza: Mendoza ( 32°53'S, 68°49'W), Jan 1970, L. E. Peña (1♀; USNM). Tucumán: Dique de Cadillal ( 26°41'S, 65°16'W), 16–29 Jan 1951, R. Golbach (3♀; FML); Finca La Cavera (Tafí Viejo; 26°45'S, 65°16'W), 23–28 Nov 1951, M. L. Aczel, R. Golbach ( 7♂, 9♀; FML); Pacará ( 26°54'S, 65°08'W), 20–28 Nov 1947, R. Golbach ( 1♂; FML); Tacanas ( 27°08'S, 64°49'W), 20–28 Nov 1957, R. Golbach ( 1♂, 3♀; FML); Tafi del Valle ( 26°52'S, 65°41'W), Nov- 6-12 Dec 1947 –1970, M. L. de Grosso, R. Golbach ( 9♂, 6♀; FML).
BRAZIL. Rio Grande do Sul: Torres ( 29°21'S, 49°44'W), Jun 1965, N. Papavero ( 4♂, 2♀; USNM).
CHILE. Santiago: El Arbol-Aculeo ( 33°51'S, 71°01'W), Oct 1969, L. E. Peña (1♀; USNM); El Convento (coast of Santiago Province, south of Santo Domingo village, south of San Antonio, Luis E. Peña G., personal communication; 33°47'S, 71°38'W), 12 Oct 1963, L. E. Peña ( 77♂, 65♀; MZUSP, USNM); Santiago ( 33°27'S, 70°40'W), 10 Oct 1963, L. E. Peña (2♀; USNM).
Distribution (Map 16). Neotropical: Argentina (Jujuy, Mendoza, Tucumán), Brazil (Rio de Janeiro, Rio Grande do Sul), and Chile (Santiago), between 22°–34°S and 44°–71°W.
Remarks. The known distribution of N. ciligena is widespread, and there is considerable color polymorphism throughout. This is especially evident in the color of the legs, which vary from being mostly blackish gray to mostly reddish yellow. The structures of the male terminalia, however, are very similar, and based on them, we have determined that the various color morphs are conspecific.
MAP 16. Distribution map for Neoephydra ciligena (Rondani) .
| MZUF |
Museo Zoologico La Specola, Universita di Firenze |
| SMNS |
Staatliches Museum fuer Naturkund Stuttgart |
| COL |
Universidad Nacional de Colombia |
| IOC |
Colecao de Culturas de Fungos do Instituto Oswaldo Cruz |
| USNM |
Smithsonian Institution, National Museum of Natural History |
| FML |
Fundacion Miguel Lillo |
| MZUSP |
Museu de Zoologia da Universidade de Sao Paulo |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Tribe |
Ephydrini |
|
Genus |
Neoephydra ciligena (Rondani)
| Mathis, Wayne N. & Marinoni, Luciane 2016 |
Neoephydra densepilosa
| Mathis 2008: 10 |
Neoephydra lopesi
| Mathis 2008: 10 |
Neoephydra grumanni
| Mathis 2008: 10 |
Parydra ciligena
| Mathis 2008: 10 |
| Mathis 1995: 239 |
| Clausen 1985: 382 |
| Wirth 1968: 22 |
Dimecoenia lopesi
| Mathis 1995: 239 |
| Wirth 1968: 23 |
| Oliveira 1954: 269 |
Dimecoenia grumanni
| Mathis 1995: 239 |
| Wirth 1968: 23 |
| Oliveira 1954: 272 |
Dimecoenia prionoptera
| Cresson 1935: 350 |
Dimecoenia densepilosa
| Mathis 1995: 239 |
| Grosso 1989: 58 |
| Wirth 1968: 23 |
| Hendel 1933: 223 |
Ephydra densepilosa
| Hendel 1930: 152 |
Ephydra ciligena
| Rondani 1868: 32 |