Terebellides africana Augener, 1918
|
publication ID |
https://doi.org/ 10.11646/zootaxa.4771.1.1 |
|
publication LSID |
lsid:zoobank.org:pub:A46FAF72-6F95-4DA3-A41D-FE770D6EDF1F |
|
DOI |
https://doi.org/10.5281/zenodo.3816151 |
|
persistent identifier |
https://treatment.plazi.org/id/03BF4239-FFF3-FFC5-EAF8-C10C9B87FD84 |
|
treatment provided by |
Plazi |
|
scientific name |
Terebellides africana Augener, 1918 |
| status |
|
Terebellides africana Augener, 1918 View in CoL stat. prom.
Figures 2A View FIGURE 2 , 3−7 View FIGURE 3 View FIGURE 4 View FIGURE 5 View FIGURE 6 View FIGURE 7 , 11A View FIGURE 11 , 37 View FIGURE 37 ; Tables 1, 2
Terebellides Stroemi View in CoL var. africana Augener, 1918 View in CoL . Augener (1918): 555 –560, Plate 7, fig. 245, text fig. 96.
Material examined. Type material. Two syntypes ( ZMH V1709 , ZMH V1712 ) (Table 1) ; lectotype: V1709 (female with intra-coelomic oocytes) ; paralectotype: V1712 (see Discussion) . Non-type material. Seven specimens (NHMD-231436, NHMD-231441, NHMD-231453, NHMD-231455, NHMD-231458); Atlantide and Galathea expeditions ( Kirkegaard, 1959) (Table 1).
Diagnosis. Body large (20–30 mm in length). Branchial anterior lobe present; posterior ventral lobes much smaller than dorsal ones, without terminal filament and emerging after fusion line of dorsal ones. TC1 notopodia and notochaetae as long as following. Thoracic neurochaetae with rostrum / capitium length about 3/1, and capitium with a first row of 3(4) medium-sized teeth, followed by progressively smaller teeth.
Description of lectotype
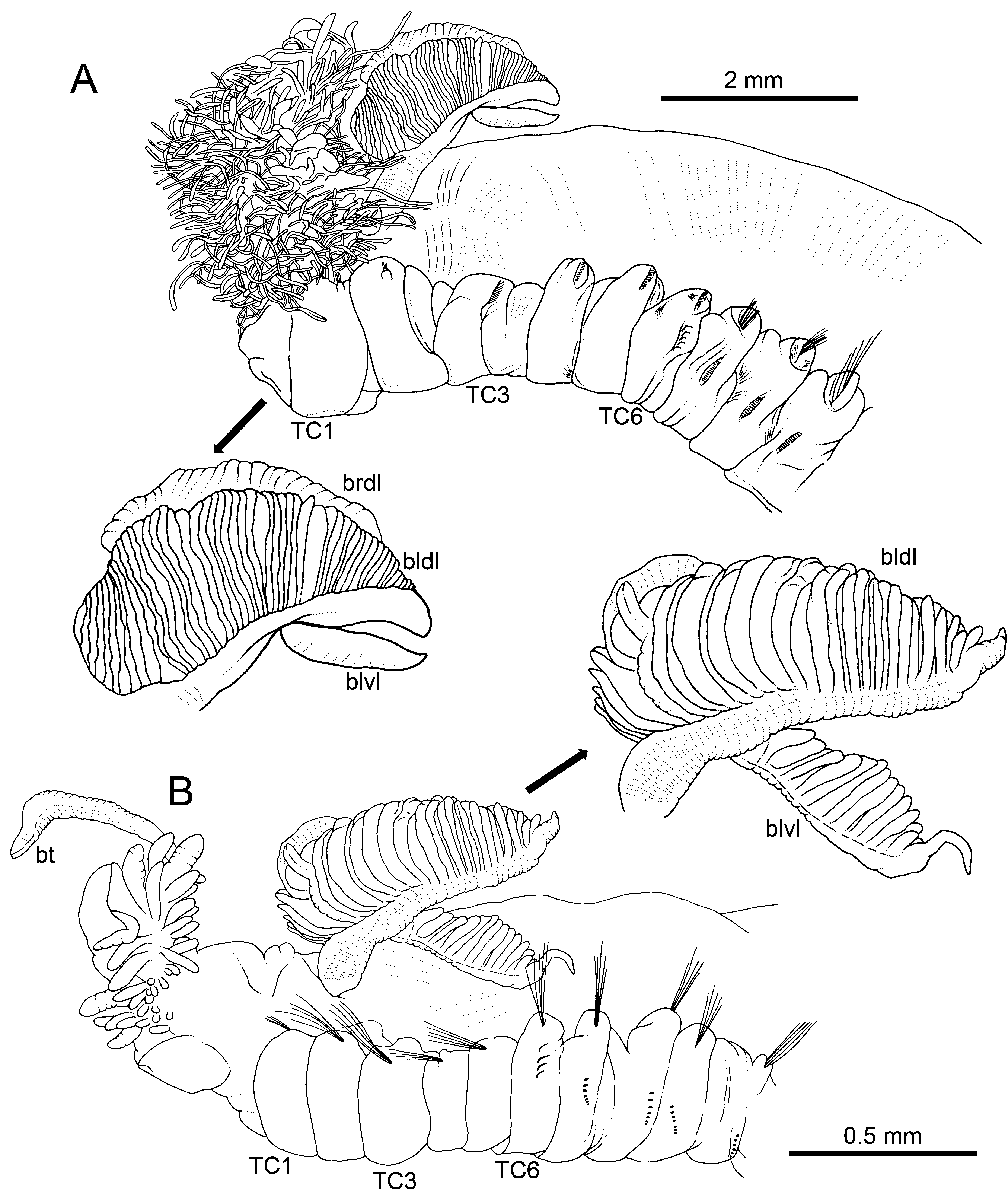
Measurements and general body features. Complete specimen, 32 mm long and 3.0 mm wide ( Fig. 2A View FIGURE 2 , 3 View FIGURE 3 A−C), body tapering posteriorly, with segments increasingly shorter and crowded towards pygidium. Prostomium compact, large tentacular membrane with typical buccal tentacles with expanded tips. SGI forming an expanded lower lip below tentacular membrane ( Fig. 3 View FIGURE 3 E−F). Lateral lappets not conspicuous on SGIII–VII (TC1–5), larger in SGIII–V, with low dorsal projections in TC1−3 ( Fig. 5A View FIGURE 5 ); TC2 dorsally elevated in relation to TC1 and TC3 ( Fig. 2A View FIGURE 2 , 5A View FIGURE 5 ). Oval-shaped glandular region in CH 3 not observed.
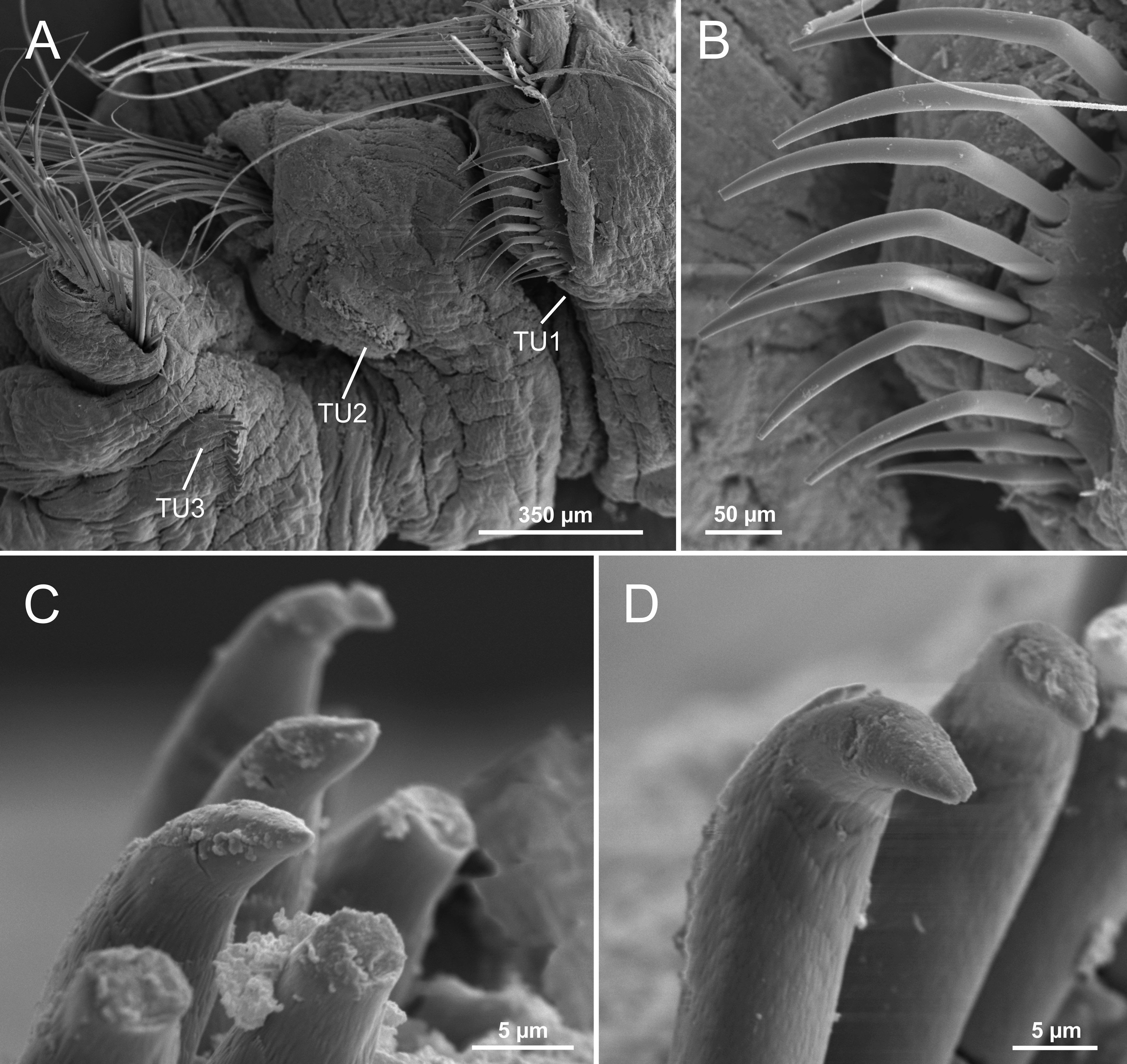
Branchiae. Branchiae arising as single structure from SGIII, with a single, short, stalked, mid-dorsal branch, typically comma-shaped and reaching TC3 ( Fig. 2A View FIGURE 2 , 3 View FIGURE 3 A−C). A short anterior lobe (see below) and two pairs of pos- terior lobes: one pair dorsal (upper), with lobes 1+2 fused along most of their length, one pair ventral (lower), with lobes 3+4 much shorter than dorsal ones, not fused together, near distal end and after a long fusion line of dorsal ones. Anterior projection of dorsal lobes (lobe 5) short, about ¼ of dorsal lobes’ length ( Fig. 2A View FIGURE 2 , 3 View FIGURE 3 C−D, 4B). Pointed projection of posterior region of both upper and lower lobes short, a bit longer in lower ones but never forming a long terminal filament. Branchial lamellae with well-developed ciliary rows and ciliated papillae on lamellar outer edge ( Fig. 5B View FIGURE 5 ).
Thorax. Eighteen pairs of notopodia (SGIII−XX), that of TC1 about as long as following ones ( Fig. 3E View FIGURE 3 , 5A View FIGURE 5 ), with dorsal projection; notochaetae as simple capillaries as long as following ones. TC2 and TC3 with dorsal projection similar to TC1 ( Fig. 5A, C View FIGURE 5 ). Notopodia of TC2 shifted dorsally in some specimens ( Fig. 5A View FIGURE 5 ). Neuropodia as sessile pinnules from TC6 (SGVIII) to pygidium, with uncini in single rows starting from TC7 (SGIX) throughout. First neuropodia at TC6, with five sharply bent, acute tipped, geniculate chaetae in left torus ( Fig. 6 View FIGURE 6 A−B); right side torus lost, presumably removed by H. Augener ( Fig. 3C View FIGURE 3 ). From TC7, neuropodia with up to 18–22 uncini per torus in one row, with long shafted denticulate hooks, rostrum about three times longer than capitium, with three large teeth above main fang, and surmounted by an upper crest of several smaller denticles ( Fig. 7 View FIGURE 7 A−C), dorsalmost hooks in anteriormost uncinigers showing a very short rostrum and a less angled capitium in relation to chaetal main axis, resembling aciculae ( Fig. 6 View FIGURE 6 C−D).
Abdomen. Abdomen as long as thorax. Thirty-two to thirty-four pairs of neuropodia as erect pinnules with about 25−30 uncini per torus; uncini with about five teeth above main fang, surmounted by a upper crest of 2 teeth and a variable number of smaller denticles (?) (see Fig. 2E View FIGURE 2 in Augener, 1918).
Other body features. Single, well-developed nephridial papilla behind TC1 notopodia and nephridial openings dorsal to TC4−5 notopodium ( Fig. 5A, C View FIGURE 5 ). Pygidium as blunt, funnel-like depression.
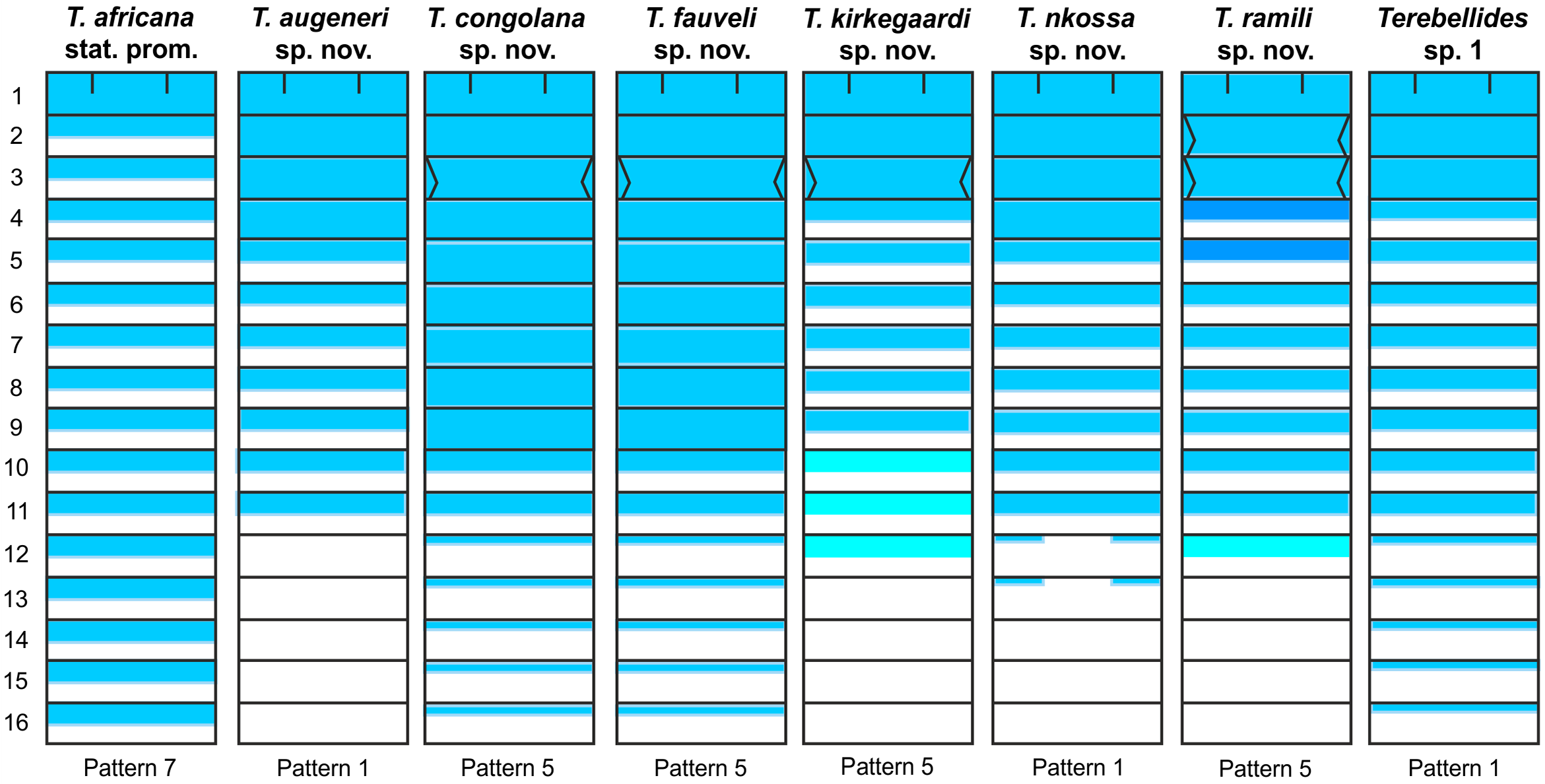
Methyl green staining pattern. Striped through the entire thorax; pattern 7 of Schüller & Hutchings (2010) ( Fig. View FIGURE 37
37).
Variability. Type specimens differing in size (paralectotype: 22.0 mm long, 2.0 mm wide), but showing no significant differences in relevant morphological characters. Branchiae clearly smaller in lectotype than in non-type specimens identified by Kirkegaard (1959), likely due to a longer preservation in alcohol.
Type locality. Equatorial Guinea; 0−30 m depth ( Table 1). The location corresponds to the lectotype (V-1709), while Augener (1918) simply refers to “West Africa” for the paralectotype (V-1712) .
Distribution and bathymetry. The revision of material from the Atlantide expedition, originally identified as T. stroemii by Kirkegaard (1959), expands the distribution of T. africana stat. prom. to the Gold Coast and Nigeria, from 17 to 66 m depth ( Fig. 11A View FIGURE 11 ). The specimen NHMD-231441, collected during the Galathea expedition (75 m depth, off Angola), mostly agrees with the diagnosis of T. africana stat. prom., but its very poor condition prevented fully confirming its identity. The specimen NHMD-231455 ( Fig. 4C View FIGURE 4 ) shows two filiform structures protruding dorsally from TC3 that might correspond to a parasitic copepod.
Nomenclatural note. Following Alonso-Zarazaga (2007), the term stat. prom. (Lat. status promotus; Eng. promoted status) is used here to qualify T. africana instead of the classical stat. nov. (Lat. status novus; Eng. new status) for referring to the rising of a subspecies (meaning of “variety” following the International Code for Zoological Nomenclature-ICZN, article 45.6.4.) to the species rank. This author also proposed stat. dem. (Lat. status demotes; Eng. demoted status) instead of stat. nov. (i.e., upgrading an infrasubspecific entity to specific or subspecific status, ICZN, article 16A) .
Remarks. Even though type specimens were not examined under SEM to avoid damage, this species is clearly distinguished from the other described here due to its large body size, a TC1 similar in size to following thoracic notopodia, and branchiae with very short ventral lobes emerging close to the distal end of the dorsal ones.
Terebellides africana stat. prom. resembles T. stroemii as redescribed by Parapar & Hutchings (2015) in size and general body shape, typical comma-shaped branchiae, and appearance and size of TC1, and mostly differs in branchial features: 1) ventral and anterior lobes clearly smaller, the former showing a different position, and 2) lobes bearing ciliated papillae ( Fig. 5 View FIGURE 5 A−B) (absent in T. stroemii, Parapar & Hutchings, 2015 : Fig. 6 View FIGURE 6 E−F).
Terebellides africana stat. prom. resembles T. stroemii sensu Caullery (1944) from Indonesia (St. 271 of Siboga Expedition ) in branchiae size and shape, which reach TC3 ( Caullery, 1944: Fig. 147A). However, their ventral posterior lobes are much different, and resemble those of a specimen from St. 311 ( Caullery, 1944: Fig. 148A) that has also larger anterior branchial lobes. Therefore, we suspect that each of them might correspond to undescribed species.
Rullier (1964a) reported T. stroemii from Cameroon, near the type locality of T. africana stat. prom. ( Equatorial Guinea), which were not assigned to the variety africana due to the shape of TC6 geniculate chaetae (not illustrated in his paper). Even though we were not able to examine this material, deposited in the Muséum national d’Histoire naturelle (Paris), it seems it does not correspond to any of the species described here. Two species described here, T. kirkegaardi sp. nov. and T. longiseta sp. nov., are geographically close to T. africana stat. prom., but they clearly differ in the shape of TC6 geniculate chaetae.
Terebellides persiae Parapar, Moreira, Gil & Martin, 2016 overall resembles T. africana stat. prom., but differs in having much larger branchiae (particularly the anterior lobe) and a characteristic thoracic dorsal hump (cfr. Fig. 2B View FIGURE 2 & 3A View FIGURE 3 in Parapar et al., 2016b) absent in T. africana stat. prom. Terebellides parvus Solís-Weiss et al., 1991 ( Solís-Weiss et al., 1991: Fig. 2a View FIGURE 2 ) has also similar branchiae, but differs by having fewer abdominal chaetigers (25 vs. 32−34 in T. africana stat. prom.).
The relative size and position of the postero-ventral branchial lobes in relation to the dorsal ones in T. africana stat. prom. resembles those of T. mediterranea Parapar, Mikac & Fiege, 2013 , T. guangdongensis Zhang & Hutchings, 2018 and T. yangi Zhang & Hutchings, 2018 . However, T. mediterranea has much larger ventral lobes, a more developed branchial stem, and a larger TC1 with longer notochaetae; T. guangdongensis shows smaller anterior branchial lobes and a very small TC1, and T. yangi clearly differs in having much-developed anterior lobes, similar to those of T. longiseta sp. nov. (see below).
Terebellides africana stat. prom. resembles T. augeneri sp. nov., but differs in body size (22−32 mm vs. 7−19 mm in T. augeneri sp. nov.) and in the position of the branchial ventral lobes (arising from the base of the dorsal ones in T. augeneri sp. nov.).
Two more species here described occur in shallow waters: T. congolana sp. nov. and T. kirkegaardi sp. nov. The former resembles T. africana stat. prom. in body size and shape and in the position of branchial ventral lobes but differs in having much longer branchial dorsal lobes that reach TC6 and a TC1 notopodium proportionally larger compared to thorax size. On the other hand, T. kirkegaardi sp. nov. differs from T. africana stat. prom. in having a distinctly smaller body and branchiae with a very long branchial stem, an anterior lobe about half the length of posterior ones, and ventral lobes originating from the base of dorsal ones.
| ZMH |
Zoologisches Museum Hamburg |
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |
Terebellides africana Augener, 1918
| Parapar, Julio, Martin, Daniel & Moreira, Juan 2020 |
Terebellides
| Augener, H. 1918: 555 |