Fridericia spelaeophila, Dózsa-Farkas & Nagy & Felföldi, 2019
|
publication ID |
https://doi.org/10.5852/ejt.2019.553 |
|
publication LSID |
lsid:zoobank.org:pub:568256D2-171B-4F15-82E8-5C33EA86F8CB |
|
DOI |
https://doi.org/10.5281/zenodo.3477384 |
|
persistent identifier |
https://treatment.plazi.org/id/03E4DB78-FF9C-FFDB-1F1E-FEE9FAA286A0 |
|
treatment provided by |
Plazi |
|
scientific name |
Fridericia spelaeophila |
| status |
sp. nov. |
Fridericia spelaeophila View in CoL sp. nov.
urn:lsid:zoobank.org:act:
Figs 1 View Fig , 4–5 View Fig View Fig
Diagnosis
The new species can be recognized by the following combination of characters: (1) large size (body length 17–29 mm in vivo); segments 48–73; (2) chaetae maximum 5–6 (7) per bundle, many detached chaetae in coelom; (3) clitellum girdle-shaped, weakly developed; (4) body wall thick (40–60 μm) and cuticle thin (1.5–2 μm) in vivo; (5) five preclitellar pairs of nephridia; (6) coelomo-mucocytes c-type of Möller (1971), lenticytes 5–10 μm; (7) oesophageal appendages long with some branches at the end; (8) dorsal vessel from 11/12 (peculiar character); (9) seminal vesicle in XI; (10) subneural glands absent; (11) sperm funnel rounded, about ¼ as long as body diameter, collar about as wide as funnel diameter, spermatozoa 150–170 μm long, heads 50–70 μm when fixed; (12) spermatheca with 10–15 sessile diverticula with sperm in them, ectal duct of variable length with 2–3 large ectal glands of variable size; proximal part of ampullae conspicuous, fused, with common opening into oesophagus dorsally.
Etymology
From the composition of spelaeo (Latin: spēlaeum, i) ‘cavern’ and phila (Latin: -philus, -phila) ‘lover’, as it was collected in a cave.
Material examined
Holotype HUNGARY • Kis-kőháti shaft (Bükk Mts) , coarse woody debris; 48°04′05.1″ N, 20°33′07.7″ E; 1 May 2018; L. Dányi & G. Balázs leg. (F.31. slide No. 2607. adult, stained whole-mounted specimen, last 11 segments used for molecular analysis, DNA ID number 1283); ELTE. GoogleMaps
Paratypes HUNGARY • 2 specimens; same data as for the holotype (P. 129.1–129.2 slide No. 2646, 2650, last 19 segments for molecular analysis, DNA ID number 1284) GoogleMaps .
HUNGARY • 3 specimens; Szepesi-Láner cave system (Bükk Mts): Láner Olivér cave , debris of decaying wood and clay; 48°05’59.8” N, 20°35’41.7” E; 16 Oct. 2014; D. Angyal, G. Balázs & L. Dányi leg. (P. 130.1–130.3 slide No. 2647a+b; 2667a+b; 2085a+b) GoogleMaps • 8 specimens; 27 Jan. 2015; D. Angyal, G. Balázs & L. Dányi leg. (P. 130.4–130.11 slide No. 2648; 2649a+b, 2651a+b; 2665a+b; 2668a+b; 2669a+b; 2670a+b; 2676a+b); ELTE .
Additional material
2 juvenile specimens only in vivo from Láner Olivér cave. Three adult specimens were fixed on site, but due to the improper fixation these were not suitable for detailed morphological analysis.
Description
MEASUREMENTS. Large, whitish worms. Holotype 59 segments, 23.5 mm long, 0.75 mm wide at VIII and 0.80 mm at the clitellum in vivo; fixed specimen: 12.7 mm long, 0.89 mm wide at VIII and 1.0 mm at the clitellum (later from this specimen, 10 segments were taken for molecular analysis, DNA ID number 1283). Body length of the paratypes 17–27 mm, width 0.60–0.78 mm at VIII and 0.77–0.90 mm at the clitellum in vivo; fixed specimens 10–29 mm long, width 0.75–1.2 mm at VIII and 0.90–1.25 mm at the clitellum; segments 48–73.
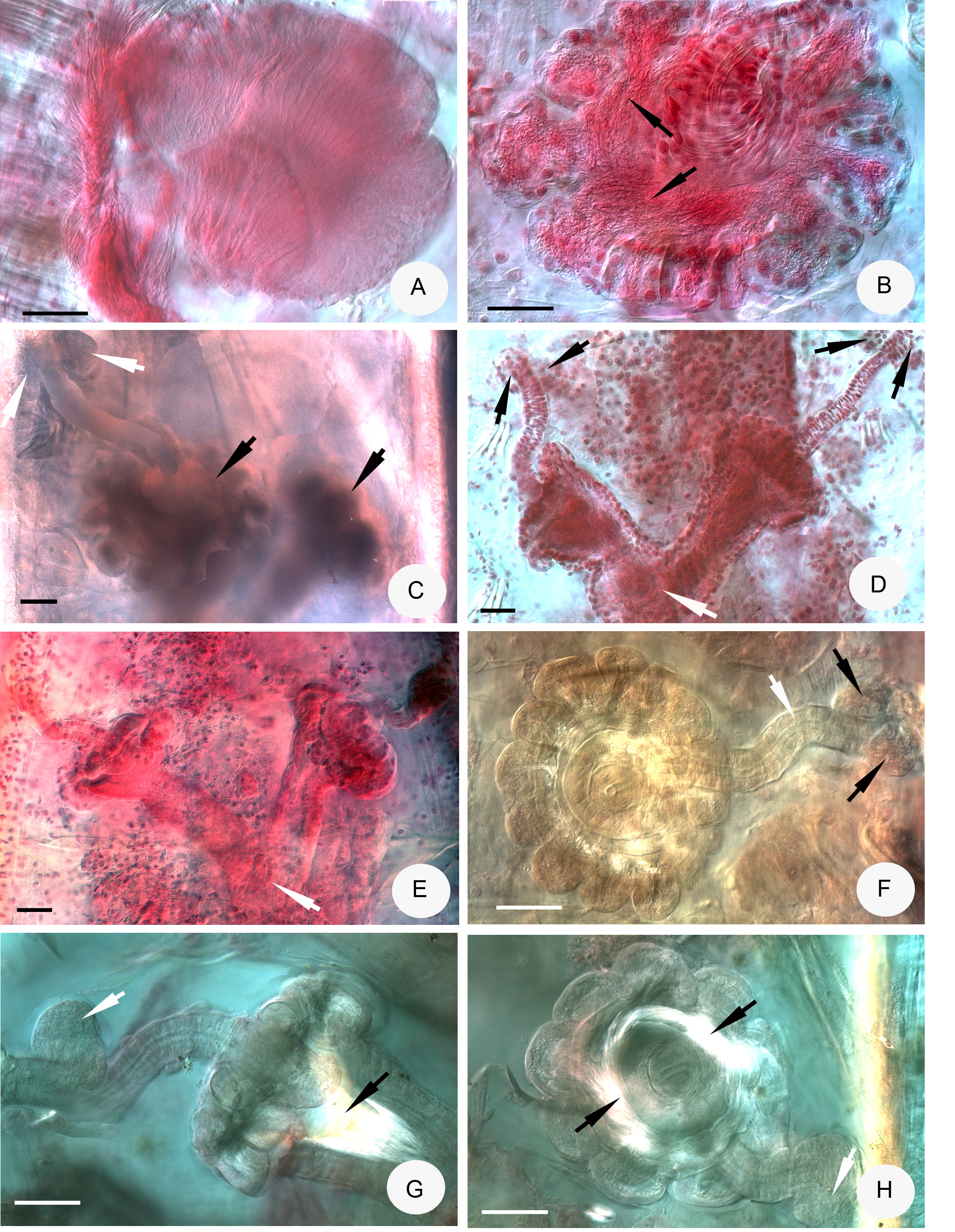
CHAETAE. Chaetal formula: 2,3,4,5,(6,7)–5,4,2: (2),4,5,6,(7)–(6),4,3,2. Mostly inner chaetae being slightly shorter and thinner than the outer ones: e.g. 50–70 × 5–7 μm and 74–100 × 9–11 μm ( Fig. 4C View Fig ), but sometimes placed irregularly (e.g. from one side to the other half in line: 78 × 8; 100 × 9; 100 × 9; 75 × 8, 93 × 9 μm) or almost equally long and thick in preclitellar bundles ( Fig. 4E View Fig ). Many detached chaetae in packages in coelomic cavity ( Fig. 4F View Fig ). In these packages the length of largest chaetae was 104 × 8–9 μm, the length of the smallest one 47 × 6 μm. From segment XVI or at the latest from XXII, in the lateral bundles only 2 chaetae, length about 80 × 4.5–7 μm.
HEAD PORE. A longitudinal slit at 0/I ( Fig. 4B View Fig ).
DORSAL PORES. From VII. Epidermal gland cells inconspicuous.
CLITELLUM. In XII–1 /2XIII, weakly developed, girdle-shaped, gland cells irregularly arranged ( Fig. 4D View Fig ), between bursal slits mostly granulocytes.
BODY WALL. Thick, about 35–60 μm, cuticle thin, about 1–2 μm when fixed.
BRAIN ( Fig. 4A View Fig ). Egg-shaped, about 170 μm long, about 1.2–1.6 times as long as wide (fixed).
OESOPHAGEAL APPENDAGES. Long, with some distal branches in V. All pharyngeal glands united dorsally and with short ventral lobes.
CHLORAGOCYTES. From V. Dorsal vessel from 11/12 (in one case in XII), with large heart-like expansions in XI–VIII, blood colourless.
CHYLUS CELLS AND MIDGUT PARS TUMIDA. Not visible (probably because the intestine was full of woodfragments and crystals (most probably calcite) in all studied specimens; Fig. 4I View Fig ).
NEPHRIDIA. Five pairs of preclitellar nephridia from 6/7 to 10/11, length ratio anteseptale: postseptale 1: 1.3–1.4 ( Fig. 4H View Fig ), adseptal origin of efferent duct.
COELOMO- MUCOCYTES. c-type: with smooth outline, elliptic, matrix pale, with well visible nucleoli, 17– 26 μm long in vivo, 13–25 μm long when fixed ( Figs 1E View Fig , 4G View Fig ).
LENTICYTES. 5–10 μm long.
SEMINAL VESICLE. In XI or X–XI.
SPERM FUNNELS. Mostly roundish, about 270–300 μm long and about 1.4–1.6 times longer than wide in vivo ( Fig. 4K View Fig ). Funnel ( Figs 4 View Fig K–L, 5A) length in fixed specimens 180–300 μm, funnel body 1.2–1.4 times longer than wide, sometimes two times as long as wide; collar about as wide as funnel body ( Figs 4L View Fig , 5A View Fig ). Length of spermatozoa not measurable in vivo, heads 110–130 μm ( Fig. 4K View Fig ), in fixed specimens, spermatozoa 150–170 μm long, sperm heads 50–70 μm. Diameter of sperm ducts 8–9 μm when fixed.
MALE COPULATORY ORGANS. 100–200 μm long, 90–180 μm wide and about 80 μm high when fixed ( Fig. 4J View Fig ), glandular body weakly developed. Bursal slits T-shaped.
SUBNEURAL GLANDS. Absent.
SPERMATHECAE ( Figs 1F View Fig , 5 View Fig B–H). Two or three large ectal glands, size variable (35–97 μm long in vivo and 40–90 μm long, fixed); ectal ducts about 310–340 μm long and 37–38 μm wide, canal 8–8.5 μm wide in vivo (when fixed, variable: 150–300 μm long, 27–40 μm wide, canal 6–8 μm), not or slightly widened entally, projecting into ampulla, ental bulbs about 70–84 μm wide when fixed.
AMPULLAE. Surrounded distally by about 10–15 sessile diverticula ( Figs 5B, F View Fig , G–H), these diverticula are 50–90 μm long and 40–90 μm wide. Sperm in a circle in lumen of ampullar distal part and also in the diverticula ( Figs 5B View Fig , G–H). Diameter of ampulla and diverticula together 180–300 μm when fixed. Proximal part of ampullae conspicuous, 100–320 μm long when fixed and fused into one common duct, which opens into oesophagus dorsally ( Figs 5 View Fig D–E). Often the length of two the proximal part is different (e.g. 130 μm and 255 μm long). 1–2 rather small mature eggs at a time.
Distribution and habitat
In Kis-kőháti shaft and Szepesi-Láner cave system, in decaying wood, Bükk Mts, Hungary.
Differential diagnosis
The new species differs from all Fridericia species described up to now by the unusual origin of the dorsal vessel before clitellar segments (mostly in XI from septum 11/12, only in one specimen in XII). Only in the following species of Fridericia does the dorsal vessel originate in XIII (intraclitellarly) or XIV, but not preclitellarly: F. parasitica Černosvitov, 1928 , F. pretoriana Stephenson, 1930 , F. monochaeta Rota, 1995 , F sousai Schmelz, 2013 and F. cusanica Schmelz, 2003 . Other differences: F. sousai and F. monochaeta are smaller (38–43 and 30–35 segments) and have only two spermathecal diverticula; F. parasitica has 40–52 segments but only 4 spermathecal diverticula; F. pretoriana has 36–51 segments and F. cusanica 32–35 segments and spermatheca without diverticula.
Results of molecular analysis
In total, 5, 4 and 6 new sequences were obtained from the studied Fridericia species in the case of ITS, CO1 and H3, respectively. Unfortunately, we failed to amplify the ITS region and CO1 gene from some specimens, which was probably due to the improper hybridization of PCR primer sequences to the extracted genomic DNA. Results of molecular analysis confirmed that Fridericia baradlana sp. nov. and Fridericia spelaeophila sp. nov. are distinct species, since based on the three studied regions, sequences acquired from the examined specimens were clearly separated in the phylogenetic trees ( Figs 6–7 View Fig View Fig ).
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
|
|
Genus |