Cryptarchopria Jelínek, 1975
|
publication ID |
https://doi.org/ 10.5281/zenodo.5319334 |
|
persistent identifier |
https://treatment.plazi.org/id/03BE87CC-F6EB-FF07-BAA8-FF2FFC83F9AA |
|
treatment provided by |
Felipe |
|
scientific name |
Cryptarchopria Jelínek, 1975 |
| status |
|
36. Cryptarchopria Jelínek, 1975
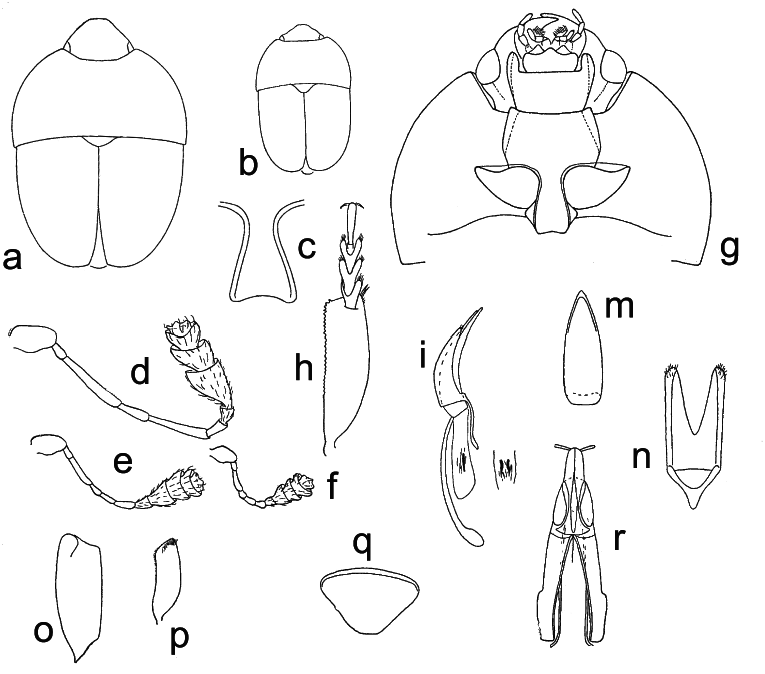
( Figs. 36 a–r View Fig )
Cryptarchopria Jelínek, 1975: 1 .
Type species. Cryptarcha infima Grouvelle, 1895: 259 (by original designation) [= Cryptarchopria infima ( Grouvelle, 1895) ].
Generic redescription and diagnosis. Inclusive species vary greatly in size (1.9–4.0 mm length), and share the following combination of characters.
Body color and pubescence: pubescence moderately long and fine, recumbent, golden, not obscuring the mostly yellowish-brown dorsal body surface, frequently with darker areas on elytra. Pubescence on lateral margins of elytra short, faintly distinct; setae on posterior margin of pronotum long and mostly bifid distad, however not observed with SEM.
Dorsal habitus: body slightly convex, more or less wide and oval ( Figs. 36a, b View Fig ); dorsal punctures on discal portion of pronotum smaller than eye facet, shallowly impressed and moderately dense; anterior margin of clypeus subtruncate anteriorly, simple, i.e. without small distinct medial bulge, lateral angles completely rounded and widely obtuse, fused with frons; frons with lateral margins moderately dilated over antennal insertions ( Fig. 36a View Fig ); circum-ocular furrows (occipital sulci) on dorsal side of head absent; eyes small-sized and moderately projecting laterally ( Figs. 36a, g View Fig ); pronotum with markedly distinct posterior angles, subrectangular to slightly acute, and slightly directed posteriorly ( Figs. 36a, g View Fig ); scutellum densely punctate on most of exposed portion; elytral punctation almost completely finely transversely strigose; elytral humeral angle obtuse, not protruding laterally; elytral humeral striae indistinct; elytral pre-sutural striae faintly visible, peculiarly fine, originating posteriorly to scutellar vertex, terminating before elytral apex, and delimiting on each elytron a faintly distinct, flat, unraised sutural area, widest at posterior third, nearly as wide as width of third antennomere; elytra apically truncately rounded in males ( Fig. 36a View Fig ), moderately and separately lobed in females; pygidium partially exposed, moderately convex, apically rounded in both sexes ( Figs. 36a, q View Fig ).
Ventral habitus: antennal furrows long and strongly raised, delimited by moderately bulged genae, nearly rectilinearly convergent posteriorly ( Fig. 36g View Fig ); mentum subpentagonal; prosternal antennal furrows on anterior margin of prosternum strongly raised, long, complete, reaching anterior margin of procoxal cavities ( Fig. 36g View Fig ); prosternal process flat, moderately wide, shallowly arcuately emarginate apically ( Fig. 36g View Fig ), subapical dilated portion 1.3–1.5× as wide as maximum width of 1 st antennomere; lateral borders of prosternal process faintly delimiting impressed furrows, distally terminating close to posterior margin ( Fig. 36g View Fig ); posterior margin of mesoventrite simple, not medially incised; scarce sexual dimorphism, impressions on metaventrite absent in both sexes; first two visible abdominal ventrites simple in males, without tufts of setae; caudal marginal lines of metacoxal cavities simple, parallel and contiguous to posterior margin of metacoxal cavities, without deep arched impression of outer ‘axillary’ portion; ‘axillary’ space on first abdominal ventrite well developed, ‘axillary’ angle widely obtuse; arched impressions on basal portion of last visible abdominal ventrite absent.
Appendages: male 1 st antennomere ~1.0× as long as width of protibiae, peculiarly wide in both sexes ( Figs. 36d–f View Fig ); 3 rd antennomere long and thin in males, allometrically developed, ~4–5× longer than wide in large males, 3× longer and much thinner than 2 nd antennomere ( Figs. 36d–e View Fig ); 4 th antennomere relatively short, 2× longer than wide, 5 th antennomere frequently allometric in males, usually long and thin, ~3–4× longer than wide; antennal club long, moderately loose, comprising last 4 or 5 antennomeres in males, 3-segmented in females, slightly wider than protibiae; labial palpi long and slender in both sexes ( Fig. 36g View Fig ), terminal segment ~2.3× as long as wide; maxillary palpi long and slender in both sexes ( Fig. 36g View Fig ), terminal segment ~3.0× as long as wide; mandibles mid-sized, arcuate, apex acuminate with small subapical tooth; tarsal claws simple, not toothed at base; tarsi of normal size and shape, 0.6–0.7× as long as corresponding tibiae; protibiae with a series of minute, fine sharp teeth on apical portion of almost rectilinear outer margins, inner margins flatly arcuate; lateral margin of meso- and metatibiae bearing a single and regular row of long thin pegs, without U-shaped sinuosity at distal third; meso- and metatibiae flat, moderately slender, markedly subtrapezoidal or axe-shaped; tarsal plates of prolegs scarcely wider in males; posterior margins of metafemora simple in males, without tubercles or projections.
Male genitalia: processes along inner side of parameres absent ( Figs. 36m –n View Fig ), with deep and wide V-shaped excision along distal margin, without deep median longitudinal desclerotization from proximal portion of tegmen extending to medial distal V-shaped excision; median lobe of aedeagus long, without lateral emargination, sharply acuminate distally, without distal minute excision or emargination; main sclerites of male internal sac small, narrow, W-shaped, moderately sclerotized, 4–5× shorter than aedeagus ( Figs. 36i–m View Fig ).
Female genitalia (ovipositor): relatively small; styli long and distinct, simple, cylindrical, not darkly pigmented, inserted at apex of contiguous gonostyloids; each gonostyloid lightly sclerotized and never pigmented distally, with a simple, non-indentate outer portion of basicoxites ( Fig. 36r View Fig ), and a single, wide, moderately pigmented and relatively more sclerotized arcuate area along outer subdistal portion of gonostyloids. ‘Central point’ of ovipositor centrally located, without proximad directed spicule.
Etymology. The generic name was derived from the combination of Cryptarcha Shuckard, 1840 , a widespread genus of Cryptarchinae , and Pria , a widespread genus of Meligethinae , which is indicative of the phylogenetic position of this taxon, i.e. closely related to Pria and allied genera, and with the body shape superficially resembling species of Cryptarcha . Gender feminine.
Biology. Members of Cryptarchopria are strictly associated with male inflorescences of forest palm species ( Arecaceae ), as analogously observed in the related Kabakovia . Cryptarchopria infima is associated with male inflorescences of the betel palm Areca catechu L. in Indonesia ( JELÍNEK 2000a); C. kabakowi is associated with male inflorescences of Arenga pinnata (Wurmb.) Merr. in Vietnam ( KIREJTSHUK & KABAKOV 1997); C. ponomarenkoi was recently discovered in northwestern Thailand (Mae Hong Son province, Pang Mapha, 1.v.2009, P. Audisio, R. Fochetti & P. Cerretti leg.) in association with male inflorescences of Caryota mitis Lour. (AUDISIO unpublished data).
Phylogenetic position. Available morphological data provide evidence of a likely sister-group relationship of Cryptarchopria to Horakia , with Kabakovia being sister to this clade ( JELÍNEK 2000a). More weakly supported relationships could also be postulated with Meligethinus and Pria . Preliminary molecular analyses with PEPCK and ITS2 nuclear genes suggest that Cryptarchopria is more closely related to Meligethes s. str. and to Pria ( LAMANNA 2009) , however no molecular data are available for Horakia and Kabakovia .
Taxonomy and geographic distribution. As previously discussed, this taxon includes three species from southeastern Asia ( KIREJTSHUK 1979b, JELÍNEK 2000a). However, undescribed species will likely be discovered on several of the endemic palm species ( HENDERSON 1986, RIFFLE 2008) occurring in southeastern Asia, from northeastern India to the Philippines and Indonesia.
Cryptarchopria infima ( Grouvelle, 1895) Indonesia: Java, Moluccas Islands
Cryptarchopria kabakowi Kirejtshuk, 1979 Vietnam
Cryptarchopria ponomarenkoi Kirejtshuk, 1989 Vietnam, N Thailand
No known copyright restrictions apply. See Agosti, D., Egloff, W., 2009. Taxonomic information exchange and copyright: the Plazi approach. BMC Research Notes 2009, 2:53 for further explanation.
|
Kingdom |
|
|
Phylum |
|
|
Class |
|
|
Order |
|
|
Family |
Cryptarchopria Jelínek, 1975
| Audisio, Paolo, Cline, Andrew Richard, Biase, Alessio De, Antonini, Gloria, Mancini, Emiliano, Trizzino, Marco, Costantini, Lorenzo, Strika, Sirio, Lamanna, Francesco & Cerretti, Pierfilippo 2009 |
Cryptarchopria Jelínek, 1975: 1
| JELINEK J. 1975: 1 |